Some biomarkers refine treatment, while others redefine it and Microsatellite Instability (MSI) is one of them. From the laboratory perspective, it represents a shift from looking at where a cancer started to understanding how it behaves at a molecular level.
What is MSI?
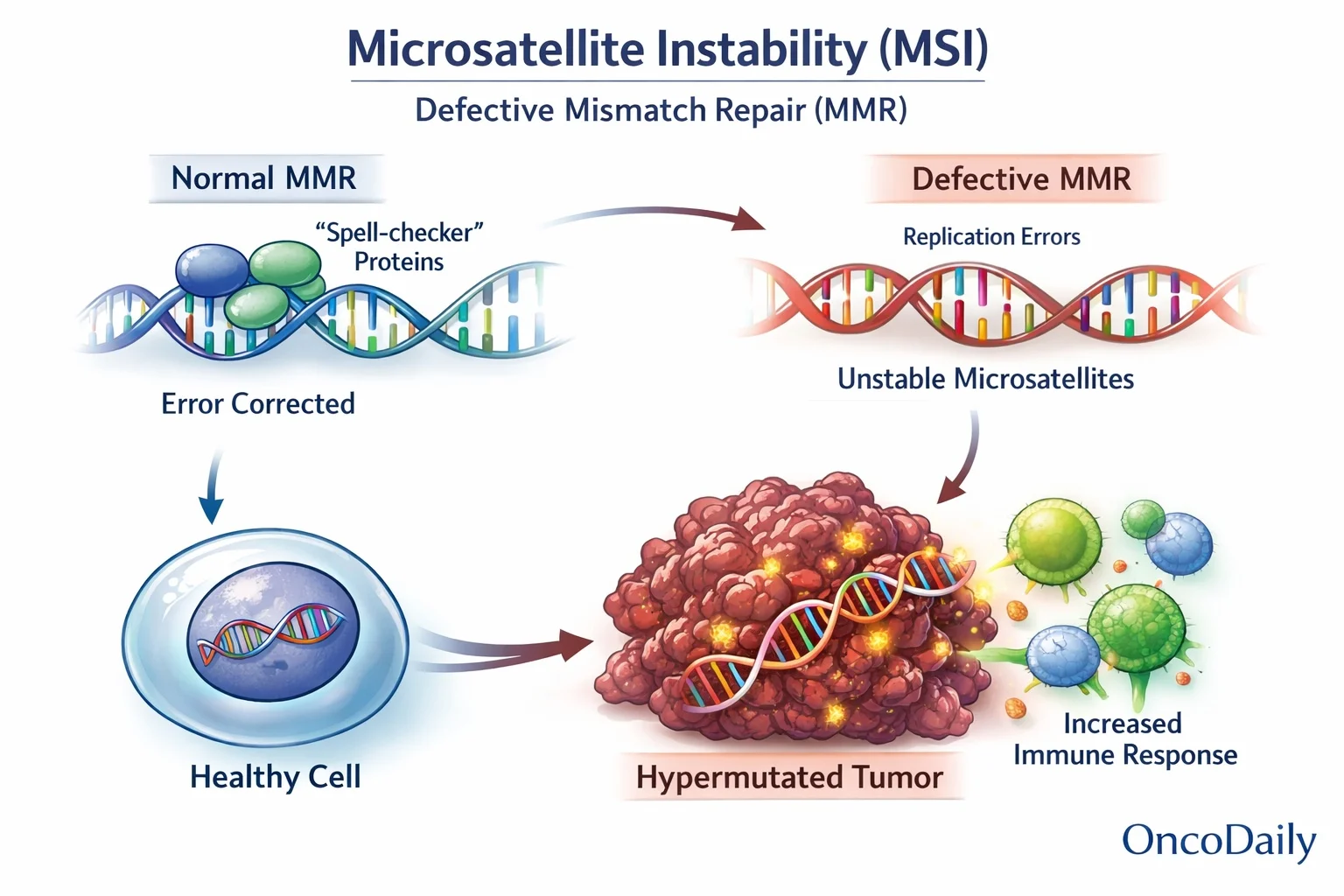
MSI is the phenotypic evidence of a defective mismatch repair (MMR) system. Under normal conditions, MMR proteins act as “spell-checkers” during DNA replication, correcting errors that occur in repetitive DNA regions (microsatellites). When this system fails, replication errors accumulate, tumors become hypermutated, and neoantigens are produced — ultimately increasing tumor visibility to the immune system.
The Lab Perspective: Our Diagnostic Approach
In practice, we don’t simply “test for MSI” — we assess the functional integrity of the genome using complementary methods.
Immunohistochemistry (IHC), often referred to as the protein map, allows us to stain tumor tissue for key MMR proteins including MLH1, MSH2, MSH6, and PMS2. Interpretation is based on nuclear staining: intact staining indicates proficient MMR (pMMR), while loss of expression suggests deficient MMR (dMMR). A critical aspect of interpretation lies in the biology of these proteins, which function as heterodimers — MLH1 pairs with PMS2, and MSH2 pairs with MSH6. As a result, loss of one protein often leads to paired loss of its partner, providing insight into the underlying defect.
In parallel, PCR-based testing serves as a genetic ruler, comparing microsatellite markers such as BAT-25 and BAT-26 between tumor and normal DNA. MSI-High status is defined by instability in two or more markers, while microsatellite stable (MSS) tumors show no instability. Increasingly, next-generation sequencing platforms integrate MSI assessment into broader genomic profiling, allowing MSI to be evaluated alongside other molecular alterations.
Laboratory Considerations
Accurate MSI testing requires careful attention to detail. Tissue fixation and processing, tumor cellularity, and assay sensitivity — as well as marker selection — all play critical roles in ensuring reliable results. These factors are not merely technical; they directly influence diagnostic accuracy and downstream clinical decisions.
Clinical Relevance and Interpretation
MSI results function as a clinical crossroads, guiding multiple aspects of patient management.
One of the most impactful applications is in immunotherapy eligibility. MSI-H tumors produce high levels of neoantigens, making them particularly responsive to immune checkpoint inhibitors such as pembrolizumab, regardless of whether the tumor originates in the colon, uterus, or stomach. This forms the basis of tumor-agnostic therapy.
At the same time, MSI testing plays a key role in Lynch syndrome screening. A dMMR/MSI-H result may indicate hereditary cancer risk, particularly in cases with loss of MSH2, MSH6, or PMS2. When MLH1 loss is observed, additional testing — such as BRAF mutation analysis or MLH1 methylation testing — is required to distinguish sporadic from hereditary cases.
Beyond this, MSI status carries both prognostic and therapeutic implications. MSI-H colorectal cancers often demonstrate a better prognosis in early-stage disease, yet they may derive reduced benefit from certain chemotherapies, including 5-FU. As a result, MSI directly influences treatment planning.
You Can Watch More on OncoDaily Youtube TV
Written by Simon Senanu, Medical Laboratory Scientist


