For years, HER2-mutant non-small-cell lung cancer sat in an uncomfortable place in thoracic oncology. It was clearly an oncogene-driven disease, but unlike EGFR-mutant or ALK-rearranged NSCLC, it did not have a clean first-line targeted treatment pathway. Patients were generally treated with chemotherapy, sometimes with immunotherapy, despite the fact that this approach often produced limited durability and offered little of the precision that had already transformed other molecular lung cancer subtypes. That gap is what makes the new Beamion LUNG-1 data so important.
In the first-line cohort of the phase 1a–1b Beamion LUNG-1 trial, zongertinib produced a confirmed objective response rate of 76%, with a median duration of response of 15.2 months and a median progression-free survival of 14.4 months in previously untreated patients with advanced or metastatic HER2-mutant nonsquamous NSCLC (Heymach et al., 2026). Just as importantly, the drug maintained a relatively favorable toxicity profile, with treatment-related grade 3 or higher adverse events in 19% of patients. In a disease where first-line targeted options have been missing, those numbers immediately place zongertinib among the most relevant new developments in lung cancer this year.
Why HER2-Mutant NSCLC Has Been So Difficult to Treat
HER2 mutations occur in approximately 2% to 4% of NSCLC and most often involve the tyrosine kinase domain. Although this represents a relatively small molecular subgroup, it is a clinically important one. HER2-mutant NSCLC is known to behave aggressively and is associated with a substantial incidence of brain metastases. Yet despite years of progress in other oncogene-driven NSCLCs, this subgroup has lagged behind. As the authors note, until recently the only approved HER2-directed therapy in lung cancer was trastuzumab deruxtecan for previously treated disease, an important advance but one associated with intravenous administration and potentially serious toxic effects, including interstitial lung disease (Heymach et al., 2026).
That left first-line therapy in an unsatisfactory position. Standard treatment generally meant chemotherapy with or without immunotherapy, even though published real-world and clinical trial data suggest modest benefit in HER2-mutant disease, with median progression-free survival often below 7 months (Heymach et al., 2026). In practical terms, patients with HER2-mutant NSCLC were being managed more like those without a clear targetable driver, despite clearly having one.
Copyright © 2026 Massachusetts Medical Society.
What Makes Zongertinib Different
Zongertinib is an oral, irreversible tyrosine kinase inhibitor that selectively inhibits HER2 while sparing wild-type EGFR. That design feature is not a small detail. It is central to the drug’s appeal. One of the recurring problems with earlier HER family inhibitors has been EGFR-related toxicity, especially rash and diarrhea. By sparing wild-type EGFR, zongertinib was designed to maintain HER2 activity while reducing the off-target toxic effects that often limit tolerability with broader inhibitors (Heymach et al., 2026).
That selectivity already looked promising in previously treated patients, where earlier Beamion LUNG-1 cohorts showed durable activity with relatively few high-grade EGFR-related toxic effects. The new question was whether the same profile would hold in untreated patients, where expectations are much higher. A first-line targeted therapy does not just need to work. It needs to work well enough, long enough, and cleanly enough to justify replacing chemotherapy at the very start of treatment.
Beamion LUNG-1 Cohort 2: A First-Line Population That Matters
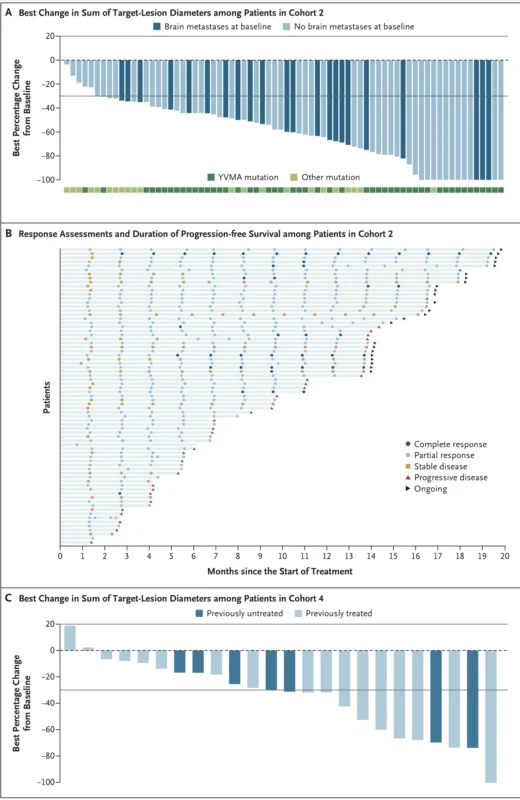
Cohort 2 of Beamion LUNG-1 included 74 previously untreated patients with advanced or metastatic nonsquamous HER2-mutant NSCLC who received zongertinib at the selected dose of 120 mg once daily. The population looked clinically relevant rather than artificially narrow. Fifty-eight percent of patients were 65 years of age or older, half were women, 45% were non-Asian, and 35% had a history of tobacco exposure. Brain metastases were already present in 30% of patients at baseline, though these were stable and asymptomatic in this cohort. The most common HER2 alteration was the A775_G776insYVMA mutation, present in 68% of patients (Heymach et al., 2026).
That baseline profile matters because it suggests the efficacy signal is not limited to an unusually favorable subgroup. This was a genuine first-line advanced NSCLC population, including older patients and patients with central nervous system involvement, which is exactly where a new targeted therapy has to prove itself.
The Response Rate Immediately Gets Attention
At the primary analysis, the confirmed objective response rate was 70%, already enough to reject the prespecified null hypothesis. With longer follow-up, that rate rose to 76%, including complete responses in 11% of patients and partial responses in 65%. The median time to objective response was just 1.4 months, which means responses were not only frequent but rapid (Heymach et al., 2026).
This is the kind of early tumor control clinicians want in first-line targeted therapy. It is not enough for a drug to show eventual activity if patients are left waiting months for disease shrinkage. The fact that zongertinib produced quick and substantial tumor reduction strengthens the case for its frontline use. The median best percentage change from baseline in the sum of target-lesion diameters was −59%, which reflects a meaningful depth of response rather than borderline radiographic improvement.
Just as important, the activity was not confined to one molecular subgroup. Patients with the common A775_G776insYVMA mutation had a confirmed response rate of 84%, while those with other HER2 tyrosine kinase domain mutations still had a response rate of 58%. That difference suggests mutation subtype may matter biologically, but it does not undermine the overall signal. Zongertinib appears active across HER2-mutant disease, while being especially strong in the most common insertion subtype (Heymach et al., 2026).
Durability Looks Strong Enough for the Front Line
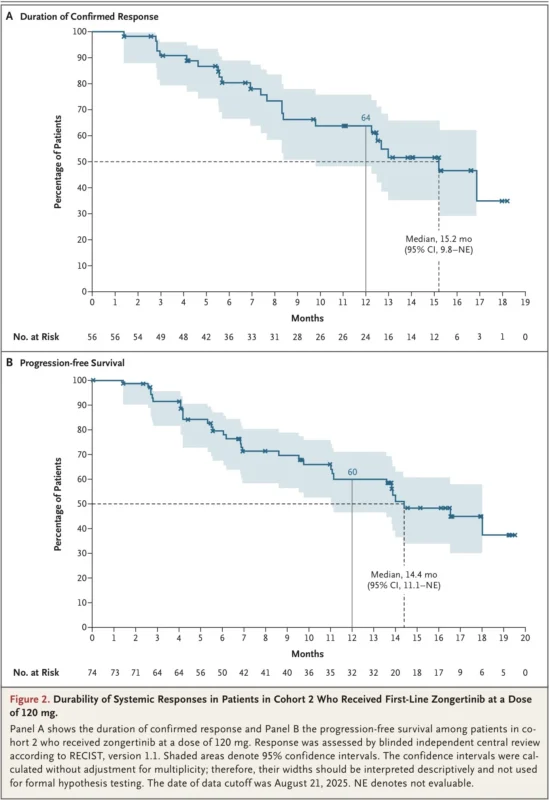
Response rate can open the conversation, but durability determines whether the drug belongs in first line. Here, zongertinib looks convincing. The median duration of response was 15.2 months, and the median progression-free survival was 14.4 months. In the context of HER2-mutant NSCLC, where first-line chemotherapy-based strategies have historically delivered much shorter control, that is a meaningful jump (Heymach et al., 2026).
This matters because the first-line setting is where the highest bar exists. A previously treated population will often accept less durable control if toxicity is manageable and alternatives are poor. First-line therapy is different. It sets the tone for the entire treatment course. The Beamion LUNG-1 data suggest zongertinib can do that credibly. It offers response rates and progression-free survival that look more like the kinds of benefits expected from targeted therapy in other driver-defined NSCLCs, even if the absolute numbers are not yet at the level seen in the most established EGFR or ALK settings.
Brain Metastases Remain a Critical Part of the Story
Any serious first-line HER2-targeted drug in NSCLC must address the brain. HER2-mutant lung cancer has a notable tendency toward central nervous system involvement, and clinicians have repeatedly seen how effective systemic therapy outside the brain can still leave an important therapeutic gap if intracranial activity is weak.
Cohort 2 included only patients with stable, asymptomatic brain metastases, and within that group the confirmed systemic objective response rate was 77%. But Beamion LUNG-1 also included exploratory cohort 4, specifically designed to examine patients with active brain metastases, including both previously untreated and previously treated patients. In that cohort, the confirmed intracranial objective response rate was 47% according to Response Assessment in Neuro-Oncology Brain Metastases criteria. Among the 23 patients who had not received prior brain radiotherapy, the intracranial response rate increased to 57% (Heymach et al., 2026).
Those numbers are especially relevant because active brain metastases are often underrepresented or excluded in targeted therapy trials. Even though cohort 4 was exploratory and smaller, it gives early evidence that zongertinib is not merely a systemic drug with incidental CNS exposure. It appears to have real intracranial activity, which strengthens its practical value in HER2-mutant disease.
Copyright © 2026 Massachusetts Medical Society.
Safety Profile: Probably One of the Most Important Reasons This Drug Matters
The other reason this dataset feels important is tolerability. In cohort 2, adverse events of any grade occurred in 99% of patients, which is expected in oncology trials, but treatment-related grade 3 or higher adverse events occurred in 19%. Dose reductions occurred in 16%, and treatment discontinuation due to adverse events occurred in 9% (Heymach et al., 2026).
The most common treatment-related toxicity was diarrhea, occurring in 55% of patients, but only 3% experienced grade 3 diarrhea. Rash occurred in 24%, and none of those cases were grade 3 or higher. This low incidence of severe EGFR-type toxic effects is consistent with the drug’s design as a HER2-selective inhibitor that spares wild-type EGFR. That is not just a mechanistic success. It is a clinically meaningful one.
There were also important toxicities that deserve attention. Increased alanine aminotransferase and decreased ejection fraction each accounted for 4% of grade 3 or higher treatment-related adverse events. Two grade 2 cases of interstitial lung disease or pneumonitis were reported and resolved with management. So the safety profile is favorable, but not trivial. Cardiac monitoring remains important, and clinicians will still need to stay alert to pulmonary toxicity. Even so, this looks clearly more manageable than the risk profile many clinicians associate with antibody-drug conjugates in the same molecular space.
How This Fits Into the Current HER2 Landscape
The Beamion LUNG-1 results immediately raise a practical question: where does zongertinib fit relative to trastuzumab deruxtecan and other emerging HER2 agents?
Trastuzumab deruxtecan changed the treatment landscape for previously treated HER2-mutant NSCLC, but it is an antibody-drug conjugate with intravenous delivery and a toxicity profile that includes serious interstitial lung disease risk. Zongertinib offers something different. It is an oral tyrosine kinase inhibitor, designed for chronic first-line administration, with durable activity and a lower burden of severe EGFR-related toxicity. That alone makes it attractive for frontline use.
The study also mentions sevabertinib, another HER2-directed tyrosine kinase inhibitor that has shown preliminary first-line activity. In that context, zongertinib’s response rate of 76%, median duration of response of 15.2 months, and median progression-free survival of 14.4 months, along with a 19% incidence of treatment-related grade 3 or higher adverse events, position it very competitively within the developing HER2 TKI field (Heymach et al., 2026).
What This Could Change in Practice
The real significance of this paper is not that it adds another active drug to a crowded space. HER2-mutant NSCLC is not a crowded space. The significance is that it may finally allow clinicians to treat this disease the way they already treat other oncogene-driven lung cancers: with targeted therapy upfront, rather than reserving HER2-directed treatment for later.
If these data are confirmed in phase 3 testing, the treatment logic for HER2-mutant NSCLC may become much cleaner. First-line zongertinib could become the preferred starting point, with antibody-drug conjugates such as trastuzumab deruxtecan potentially moving later in sequence. That would represent a major conceptual shift. Instead of giving chemotherapy first and saving HER2-directed treatment for relapse, clinicians could finally begin HER2-mutant NSCLC with a HER2-specific oral inhibitor.
That matters not only for efficacy, but for patient experience. An oral targeted therapy with rapid responses and manageable toxicity is a very different first treatment from platinum-based chemotherapy with or without immunotherapy. For patients, that difference is tangible from the first week of therapy.
Limits of the Study
The study still has clear limitations. It is open-label and lacks a standard-care comparator arm. The brain metastases cohort is exploratory and relatively small. Confidence intervals were not adjusted for multiplicity and should be interpreted descriptively. And as always, cross-trial comparisons with chemotherapy, antibody-drug conjugates, or other TKIs must be made cautiously (Heymach et al., 2026).
That said, none of those limitations cancel the central point. A first-line response rate of 76%, with median progression-free survival of 14.4 months and relatively favorable tolerability, is not a marginal result that depends on statistical decoration. It is a clinically strong signal.
Bottom Line
Zongertinib is the clearest sign yet that HER2-mutant NSCLC is finally entering the targeted therapy era in the first-line setting. In previously untreated advanced disease, it produced rapid, deep, and durable responses, including meaningful activity in patients with brain metastases, while maintaining a toxicity profile that appears considerably more manageable than many clinicians might expect from HER2-directed therapy. The most important message is simple: HER2-mutant NSCLC may no longer have to begin with chemotherapy. With zongertinib, it may finally begin with a targeted drug designed for exactly this disease (Heymach et al., 2026).
You can read full abstract here