KRAS G12D has long been one of the most frustrating targets in thoracic oncology. It is biologically important, clearly druggable in theory, and yet patients with KRAS G12D-mutant non-small cell lung cancer have had no approved targeted therapy. That is why the updated Phase 1 data for zoldonrasib presented at the 2026 AACR Annual Meeting drew attention. In a previously treated KRAS G12D NSCLC population, the oral RAS(ON) G12D-selective inhibitor showed encouraging response rates, durable disease control, and a manageable safety profile, offering one of the clearest signals so far that this subset may finally be moving toward a targeted treatment era (Revolution Medicines, 2026; Riess et al., 2026).
Why KRAS G12D Matters in Lung Cancer
KRAS G12D is not the most common KRAS alteration in lung cancer, but it remains clinically meaningful. According to the company and abstract background, it is found in about 4% of NSCLC cases, and for these patients there is currently no approved RAS-targeted therapy. That leaves treatment largely dependent on standard systemic approaches after progression, usually immune checkpoint inhibitors, platinum chemotherapy, and later-line agents, with median overall survival in previously treated advanced NSCLC often remaining under one year (Revolution Medicines, 2026; Riess et al., 2026).
That context is important because it shapes how these results should be read. This is not a crowded space with multiple established targeted options. It is a setting of real unmet need.
What Is Zoldonrasib?
Zoldonrasib, also known as RMC-9805, is described as a potent, oral, covalent, RAS(ON) G12D-selective tri-complex inhibitor. Mechanistically, it targets the active, GTP-bound state of RAS G12D. That matters because the drug is designed specifically for the mutant active form rather than relying on broader pathway suppression. Revolution Medicines is positioning it as a selective approach for a mutation that has historically lacked a direct therapeutic option (Revolution Medicines, 2026; Riess et al., 2026).
How The Study Was Designed
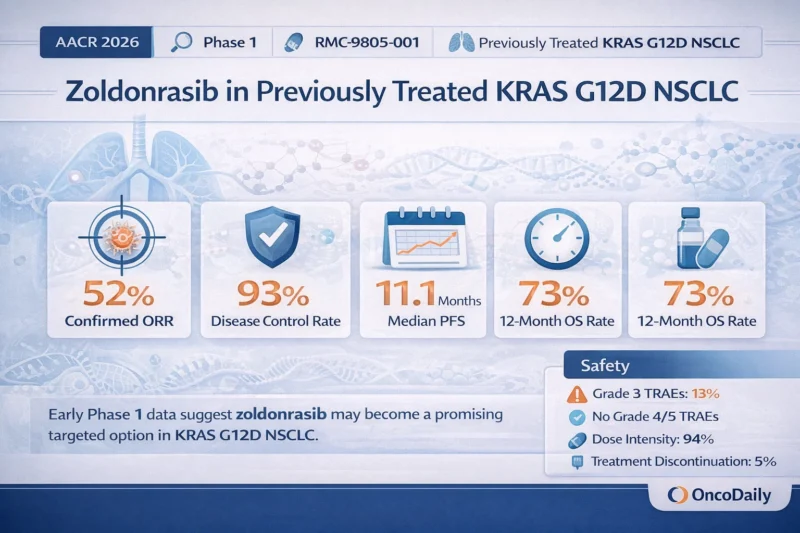
The data came from the ongoing Phase 1 study RMC-9805-001 (NCT06040541), a multicenter, open-label dose-escalation and dose-expansion trial in advanced solid tumors harboring a KRAS G12D mutation. The recommended Phase 2 dose in NSCLC was identified as 1200 mg once daily (Riess et al., 2026).
Two populations are especially important in interpreting the results. The safety population included 40 patients with KRAS G12D NSCLC treated at 1200 mg once daily. The efficacy population was more selective: it included 27 patients treated at the same dose who had received prior immune checkpoint inhibitor and platinum chemotherapy, had not received prior docetaxel, and had enough follow-up for response assessment (Revolution Medicines, 2026; Riess et al., 2026).
This distinction matters. The efficacy signal is promising, but it comes from a selected subgroup rather than the full treated cohort.
The Response Rate Is Strong for a Phase 1 Signal
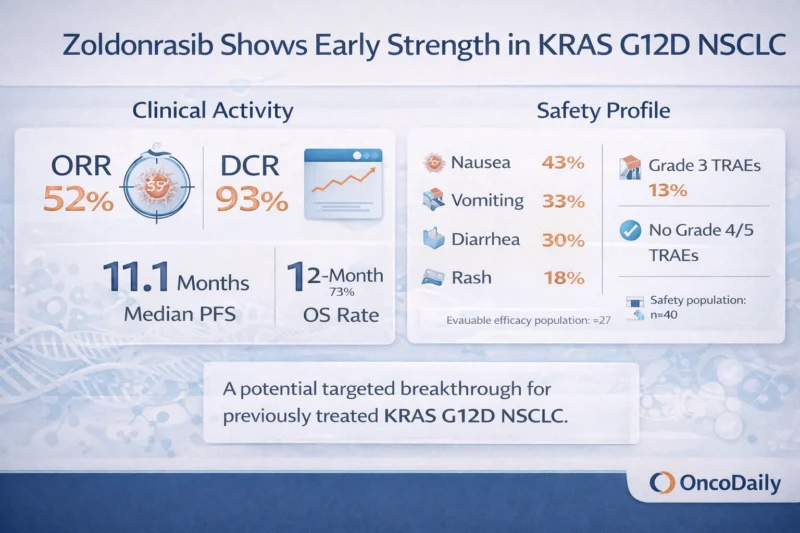
Among the 27 efficacy-evaluable patients, the confirmed objective response rate was 52% with a 95% confidence interval of 32 to 71. The disease control rate was 93% with a 95% confidence interval of 76 to 99. Median time to response was 1.4 months, suggesting the activity was not only meaningful but also relatively rapid (Revolution Medicines, 2026; Riess et al., 2026).
In a previously treated KRAS G12D NSCLC setting, those numbers stand out. They do not establish a new standard on their own, but they clearly justify further development. In practical terms, a response rate above 50% in this population signals that zoldonrasib is doing more than producing occasional tumor shrinkage. It is demonstrating repeatable antitumor activity in a mutation-defined group that has lacked a targeted option.
Durability Also Looks Encouraging
Response rate alone is not enough. What gives these data more weight is the early durability signal. Median duration of response was not yet estimable, with the lower bound of the 95% confidence interval at 8.3 months. Median progression-free survival was 11.1 months with a 95% confidence interval of 5.3 months to not estimable. Overall survival data were still immature, but median overall survival had not yet been reached, and the estimated 12-month overall survival rate was 73% (Revolution Medicines, 2026; Riess et al., 2026).
For a Phase 1 study, those are meaningful outcomes. They do not yet offer the certainty of a randomized trial, but they do suggest that the responses are not fleeting. The progression-free survival figure is especially notable in a previously treated population where historical options often have limited durability.
Safety Profile: Encouraging, But Still Needs Context
The safety results are one of the more attractive aspects of the dataset. In the 40-patient safety population, treatment-related adverse events occurring in at least 15% of patients were nausea (43%), vomiting (33%), diarrhea (30%), and rash (18%). Most treatment-related adverse events were Grade 1, with fewer Grade 2 events and Grade 3 treatment-related adverse events in 13% of patients. No Grade 4 or Grade 5 treatment-related adverse events were observed. Treatment-related discontinuation occurred in 5%, dose interruption in 15%, dose reduction in 3%, and the mean relative dose intensity remained high at 94% (Revolution Medicines, 2026; Riess et al., 2026).
That profile is favorable for an early-phase targeted therapy in a heavily pretreated setting. Still, it is worth being precise. “Favorable” does not mean toxicity-free. Gastrointestinal adverse events were common, and tolerability always looks cleaner in smaller, intensively monitored early-phase cohorts than it may in broader use. Even so, the absence of Grade 4 or 5 treatment-related adverse events and the low rate of dose modification are both encouraging signs.
Why These Data Matter
The importance of these results lies in what they represent more than in what they already prove. They do not prove that zoldonrasib should become standard treatment tomorrow. They do show that KRAS G12D NSCLC is beginning to look like a disease subtype that may finally support a mutation-matched strategy.
That is a major development. KRAS-targeted therapy in lung cancer has already changed practice for KRAS G12C. The question has been whether other KRAS alleles could follow with equally rational, allele-specific approaches. Zoldonrasib is one of the strongest early answers yet for G12D.
Important Caveats
There are several reasons not to overstate the findings. First, this is still a Phase 1 study, not a randomized registrational comparison. Second, the efficacy analysis was performed in a small selected subgroup of 27 patients, not the full treated NSCLC population. Third, the data currently come from a conference abstract and company press release, both valuable sources for early signals but not substitutes for a mature peer-reviewed full report (Revolution Medicines, 2026; Riess et al., 2026).
This means the results should be interpreted as highly promising early evidence, not final proof. The confidence intervals are still wide, and survival outcomes remain immature.
What Comes Next
The development path is already expanding. According to the abstract and company materials, zoldonrasib is being evaluated not only as monotherapy in previously treated disease but also in combination with standard-of-care regimens in treatment-naive patients (Revolution Medicines, 2026; Riess et al., 2026). That next step is important because targeted therapies become truly practice-changing when they move from late-line rescue into earlier lines where they can reshape the treatment sequence.
If the current signal holds up in larger cohorts and later-phase studies, zoldonrasib could become one of the most important precision oncology developments for KRAS G12D-driven lung cancer.
Bottom Line
The updated AACR 2026 data position zoldonrasib as one of the most credible emerging targeted therapies for previously treated KRAS G12D NSCLC. In a selected subgroup, the drug delivered a 52% confirmed objective response rate, 93% disease control rate, 11.1-month median progression-free survival, and a generally manageable safety profile with few severe treatment-related toxicities (Revolution Medicines, 2026; Riess et al., 2026). These are still early data, and caution is appropriate. But for a population that has had no approved targeted therapy, the message is clear: KRAS G12D NSCLC may no longer be standing still.
Read Full Abstract Here