KRAS G12C-mutant non-small cell lung cancer has become one of the clearest examples of progress and frustration in modern thoracic oncology. The development of KRAS G12C inhibitors such as sotorasib and adagrasib proved that KRAS could be therapeutically targeted after decades of being considered “undruggable.” Yet the clinical benefit of these agents has remained limited by incomplete responses, adaptive signaling, and acquired resistance. Most patients eventually progress, and the depth of response is often less durable than what has been achieved with targeted therapies in EGFR-mutant or ALK-positive NSCLC (Skoulidis et al., 2021; de Langen et al., 2023; Jänne et al., 2022).
A new preclinical study explores a biologically grounded strategy to deepen and extend responses to KRAS G12C inhibition: combining sotorasib with trastuzumab deruxtecan, the HER2-directed antibody-drug conjugate better known as T-DXd. The study evaluated this combination in KRAS G12C-mutant NSCLC xenograft models and performed exploratory HER2 profiling in human NSCLC samples. The findings suggest that sotorasib may create a therapeutic vulnerability by inducing adaptive HER2 upregulation, allowing T-DXd to enhance tumor regression in models that are either treatment-naïve or relapsed on sotorasib (Ozakinci et al., 2026).
Why HER2 Matters After KRAS G12C Inhibition
Resistance to KRAS G12C inhibitors is biologically complex. Tumors can reactivate downstream MAPK signaling, engage bypass pathways, acquire secondary mutations, or evolve through heterogeneous adaptive states. Among these bypass mechanisms, ERBB/HER family signaling has emerged as an important route of persistence after KRAS inhibition. HER2, encoded by ERBB2, can support signaling adaptation and help tumor cells survive pharmacologic KRAS suppression (Solanki et al., 2021; Ho et al., 2021; Kruspig et al., 2018).
This makes HER2 an attractive therapeutic target, but not necessarily in the traditional sense of HER2 addiction. T-DXd does not require HER2 to be the sole oncogenic driver. It requires sufficient HER2 protein expression on the tumor cell surface for antibody binding, internalization, and payload delivery. Because T-DXd carries a topoisomerase I inhibitor payload and can produce bystander cytotoxicity, it may be particularly useful in tumors with heterogeneous HER2 expression. That feature is relevant in NSCLC, where HER2 expression may be variable, dynamic, and difficult to classify using scoring systems developed for breast or gastroesophageal cancer (Modi et al., 2022; Ozakinci et al., 2026).
A Combination Designed to Exploit Adaptive HER2 Upregulation
The investigators first tested the combination in H358 xenografts, a commonly used KRAS G12C-mutant NSCLC model. At baseline, these tumors showed abundant membranous HER2 expression. Sotorasib alone produced marked tumor regression, but tumors relapsed after 10 to 12 weeks of continuous treatment, reflecting the clinical problem of acquired resistance. Importantly, tumors progressing on sotorasib showed further HER2 upregulation, detectable as early as 48 hours after treatment and sustained during therapy (Ozakinci et al., 2026).
T-DXd alone did not induce regression in treatment-naïve H358 xenografts. The combination, however, produced a much deeper response, with approximately 1,000-fold tumor volume reduction and tumor control maintained for at least 16 weeks during treatment. This is the central biological message of the paper: T-DXd was not simply acting as an independent monotherapy. Its major effect emerged when paired with KRAS G12C inhibition, suggesting that sotorasib may help create or amplify HER2-directed ADC sensitivity (Ozakinci et al., 2026).
Activity Was Preserved in Sotorasib-Relapsed Models
One of the most clinically relevant parts of the study was the evaluation of sotorasib-relapsed xenograft models. The investigators continued sotorasib in the original H358 cohort until tumors regrew to approximately pre-treatment volumes. When T-DXd was added to ongoing sotorasib, tumors regressed again and remained controlled for an additional 23 weeks of treatment (Ozakinci et al., 2026).
The authors then created two independent sotorasib-relapsed models, SR1 and SR2, derived from tumors that had progressed during continuous sotorasib exposure. These models retained high HER2 expression, which increased further after sotorasib exposure. Sotorasib monotherapy produced only partial and less durable responses, while T-DXd alone again failed to induce meaningful regression. In contrast, the sotorasib plus T-DXd combination restored deep and durable regression, similar to the responses seen in treatment-naïve xenografts (Ozakinci et al., 2026).
This finding is important because it positions the combination not only as a possible upfront intensification strategy but also as a potential post-KRAS-inhibitor approach. That is where the unmet need is especially high. Many patients with KRAS G12C-mutant NSCLC now receive immunotherapy and chemotherapy earlier in the disease course, and KRAS G12C inhibitors are often used in later lines. A strategy that remains active after sotorasib relapse would be clinically meaningful if it translates into human benefit.
Broad Activity Across Additional KRAS G12C Models
The investigators also tested the combination across eight additional KRAS G12C-mutant xenograft models. As expected, responses to sotorasib alone were heterogeneous. None of the additional models showed the same strong sensitivity seen in H358, and several had only transient tumor stabilization or modest regression before progression. T-DXd monotherapy inhibited tumor growth in several models but generally did not induce regression (Ozakinci et al., 2026).
The combination was more active. Sotorasib plus T-DXd induced regression in 7 of 8 additional KRAS G12C-mutant xenograft models. The authors did not observe overt toxicity or weight loss in these preclinical experiments. This broader activity supports the hypothesis that combining KRAS inhibition with HER2-directed ADC therapy may overcome some of the heterogeneity that limits single-agent KRAS G12C inhibition (Ozakinci et al., 2026).
At the same time, the findings remain preclinical. Xenograft models are useful for testing drug activity and tumor-intrinsic mechanisms, but they do not fully capture immune effects, human pharmacology, long-term toxicity, or the clinical complexity of heavily pretreated NSCLC.
HER2 Expression in Human NSCLC Samples
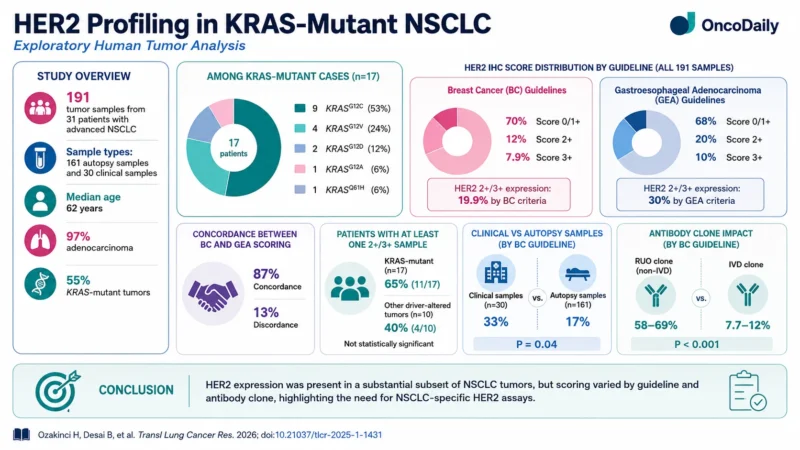
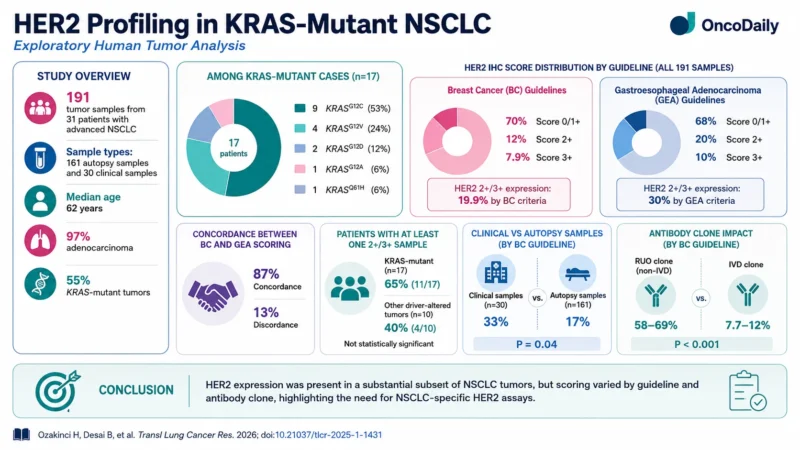
To support clinical translation, the authors also examined HER2 expression in 191 clinical and rapid autopsy tumor samples from 31 patients with advanced NSCLC. Most patients had adenocarcinoma, and 55% had KRAS-mutant disease. No HER2 mutations or amplifications were detected, meaning the analysis focused on HER2 protein expression rather than ERBB2 genomic alteration (Ozakinci et al., 2026).
HER2 scoring varied depending on whether breast cancer or gastroesophageal adenocarcinoma interpretation guidelines were used. Across all samples, HER2 IHC 2+/3+ expression was identified in 19.9% of samples using breast cancer guidelines and 30% using gastroesophageal guidelines. Concordance between the two scoring systems was 87%, but 13% of samples were discordant. HER2 2+/3+ expression was numerically more frequent in KRAS-mutant tumors than in tumors with other oncogenic drivers, although this difference was not statistically significant (Ozakinci et al., 2026).
These data suggest that HER2 expression is present in a meaningful subset of KRAS-mutant NSCLC, but they also expose a major biomarker challenge. HER2 testing in NSCLC is not yet optimized for selecting patients for HER2-directed ADC combinations. Current interpretation systems were built for other tumor types, and this study shows that applying them to NSCLC can lead to discordant results.
The Biomarker Problem Is Not Minor
The HER2 biomarker question may become the most important translational issue for this strategy. The study found that HER2 scoring was influenced by the guideline system used, the sample type, and even the antibody clone. Clinical samples showed higher HER2 expression than autopsy samples by breast cancer scoring, while a research-use-only HER2 clone identified far higher HER2 2+/3+ positivity than the FDA-approved 4B5 clone (Ozakinci et al., 2026).
This matters because biomarker classification could determine trial eligibility and eventual treatment access. If NSCLC-specific HER2 thresholds are not developed, some patients may be incorrectly classified as HER2-negative or HER2-positive. The authors therefore emphasize the need for assay harmonization, optimized scoring, and prospective validation in NSCLC rather than relying on breast or gastroesophageal frameworks.
The xenograft data also suggest that categorical HER2 IHC scoring may not fully predict response depth. HER2 expression may be necessary for T-DXd activity, but it may not be sufficient on its own. Adaptive HER2 upregulation after sotorasib, intratumoral heterogeneity, payload sensitivity, internalization efficiency, and bypass signaling may all influence response.
Clinical Translation Is Already Underway
The preclinical results provided the rationale for a first-in-human phase I/II clinical trial evaluating sotorasib plus T-DXd in previously treated KRAS G12C-mutant NSCLC. The study is registered as NCT07012031 and is designed to evaluate safety and efficacy in patients who have already been exposed to standard therapies, including prior KRAS G12C inhibition (Ozakinci et al., 2026).
This trial will be important for several reasons. First, it will determine whether the strong xenograft activity translates into meaningful clinical responses. Second, it will clarify safety. Sotorasib is most commonly associated with hepatotoxicity, while T-DXd carries a known risk of interstitial lung disease and pneumonitis. These toxicities are not mechanistically overlapping, but the combination still requires careful prospective monitoring. Third, the trial includes exploratory HER2 analyses that may help define whether baseline HER2 expression, treatment-induced HER2 modulation, or other biomarkers can guide patient selection.
Clinical Meaning
This study offers a thoughtful example of how resistance biology can be turned into a therapeutic opportunity. Instead of treating HER2 upregulation after KRAS inhibition only as a resistance mechanism, the investigators propose using it as a targetable vulnerability. If sotorasib pushes tumor cells toward greater HER2 dependence or HER2 surface expression, T-DXd may convert that adaptation into a therapeutic liability.
The concept is attractive because it does not require HER2 mutation or HER2 amplification. It focuses on dynamic HER2 protein expression and ADC payload delivery. In a disease where resistance to KRAS G12C inhibitors is common and heterogeneous, this could represent a more flexible approach than targeting one specific resistance mutation.
However, the interpretation must remain cautious. These are preclinical data with exploratory human HER2 profiling. The combination is promising, but not yet clinically proven. The real test will be whether patients with KRAS G12C-mutant NSCLC can achieve deeper and more durable responses without unacceptable toxicity.
Conclusion
Ozakinci, Desai, and colleagues provide compelling preclinical evidence that T-DXd can enhance the activity of sotorasib in KRAS G12C-mutant NSCLC, including in models that relapse during sotorasib exposure. The study supports a biologically rational combination strategy based on adaptive HER2 upregulation and HER2-directed ADC delivery.
At the same time, the work highlights a critical biomarker gap. HER2 expression appears sufficiently common in KRAS-mutant NSCLC to justify clinical investigation, but current IHC interpretation systems are not optimized for this disease. As the field moves toward trials of sotorasib plus T-DXd, NSCLC-specific HER2 testing standards will be essential.
For now, this is a strong preclinical signal with clear translational momentum. If validated clinically, it could open a new strategy for overcoming the limited depth and durability of response seen with KRAS G12C inhibitor monotherapy.