A growing body of real-world evidence suggests that not all patients receiving standard-dose immunotherapy achieve the same systemic drug exposure. A new multicenter study published explored whether early pembrolizumab plasma level could serve as a prognostic biomarker in metastatic non-small cell lung cancer (NSCLC), potentially helping identify patients less likely to benefit from treatment.
The study, led by Cristina Martínez-Toledo and colleagues, analyzed 133 patients with metastatic NSCLC treated with first-line pembrolizumab either as monotherapy or in combination with chemotherapy across 12 centers in Spain. The investigators focused on pembrolizumab trough plasma levels measured after the first treatment cycle (C1 trough), evaluating their association with survival outcomes, tumor burden, albumin levels, and PD-1 receptor availability on T cells.
Why Pembrolizumab Exposure Matters
Pembrolizumab has transformed the treatment landscape of advanced NSCLC, particularly for patients with PD-L1-positive tumors. However, despite standardized dosing strategies, clinical outcomes remain highly variable. While pivotal clinical trials suggested a relatively flat exposure-response relationship once PD-1 receptor saturation is achieved, real-world populations may experience broader pharmacokinetic variability due to factors such as body weight, cachexia, systemic inflammation, and tumor burden.
This study aimed to determine whether early pembrolizumab exposure reflects more than pharmacokinetics alone and whether it could act as a clinically relevant biomarker.
Study Design and Methods
The analysis included patients from the BLI-O project, a multicenter observational initiative evaluating immunotherapy biomarkers in NSCLC. Eligible patients had metastatic disease, ECOG performance status 0–1, and received standard pembrolizumab-based first-line therapy.
Pembrolizumab plasma concentrations were measured immediately before cycle 2 using ELISA assays. Patients were divided into two groups based on a cutoff of 10 µg/mL:
- High exposure: ≥10 µg/mL
- Low exposure: <10 µg/mL
The investigators also assessed:
- Tumor burden using tumor size, T-stage, and metastatic spread
- Baseline albumin levels
- Free PD-1 expression on circulating T cells using flow cytometry
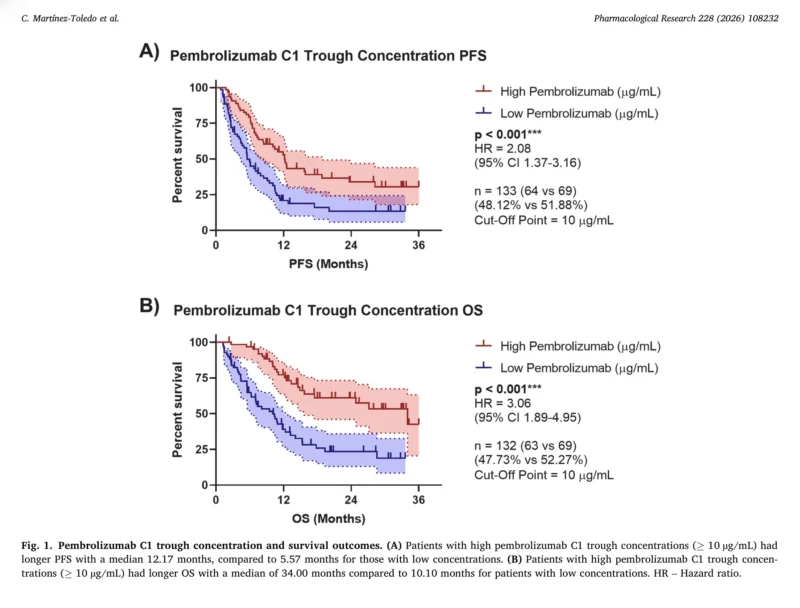
Low Pembrolizumab Levels Were Linked to Worse Outcomes
The most important finding of the study was the strong association between low pembrolizumab exposure after the first cycle and inferior survival outcomes.
Patients with low pembrolizumab trough levels had:
- More than double the risk of disease progression (PFS HR 2.08; p<0.001)
- More than triple the risk of death (OS HR 3.06; p<0.001)
Median progression-free survival was:
- 12.17 months in patients with high pembrolizumab levels
- 5.57 months in patients with low levels
Median overall survival was:
- 34.0 months in the high-exposure group
- 10.1 months in the low-exposure group
The survival differences remained consistent across multiple subgroup analyses, including treatment type, PD-L1 status, ECOG performance status, and smoking history.
Importantly, pembrolizumab trough concentration remained independently associated with progression-free survival in multivariate analysis alongside ECOG performance status and PD-L1 positivity.
For overall survival, pembrolizumab exposure was the only variable retaining independent prognostic significance in the multivariate model.
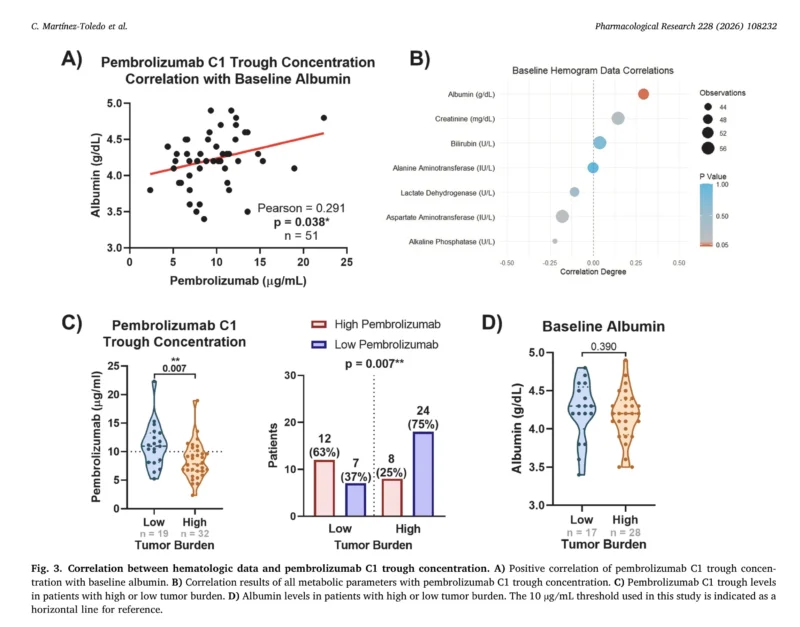
Tumor Burden and Albumin Influenced Drug Exposure
The investigators also explored factors associated with lower pembrolizumab levels.
Patients with higher tumor burden had significantly lower pembrolizumab exposure:
- Median trough concentration: 7.87 µg/mL in high tumor burden patients
- Median trough concentration: 10.98 µg/mL in low tumor burden patients
Similarly, lower baseline albumin levels correlated with reduced pembrolizumab exposure.
These findings support the hypothesis that systemic catabolic activity, cancer cachexia, and altered FcRn-mediated antibody recycling may increase pembrolizumab clearance in certain patients.
The authors suggest that high tumor burden may not only reflect aggressive disease biology but may also directly influence monoclonal antibody pharmacokinetics.
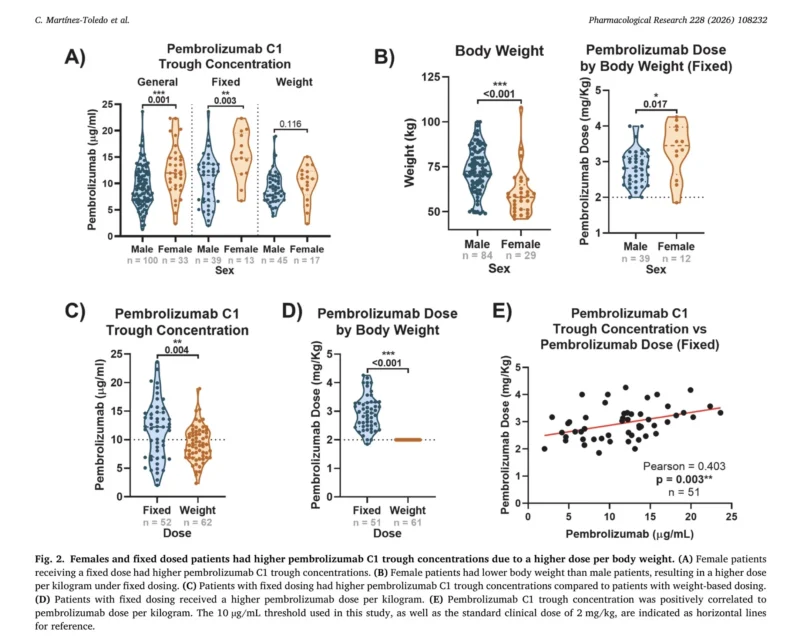
Fixed Dosing Produced Higher Drug Exposure
Another notable finding involved dosing strategy.
Patients receiving fixed-dose pembrolizumab (200 mg every 3 weeks) had significantly higher trough concentrations than those receiving weight-based dosing (2 mg/kg).
Female patients also demonstrated higher pembrolizumab exposure, largely explained by lower body weight and consequently higher relative dose per kilogram.
However, neither sex nor dosing strategy independently affected survival outcomes, suggesting that actual drug exposure, rather than the dosing approach itself, may be the clinically relevant factor.
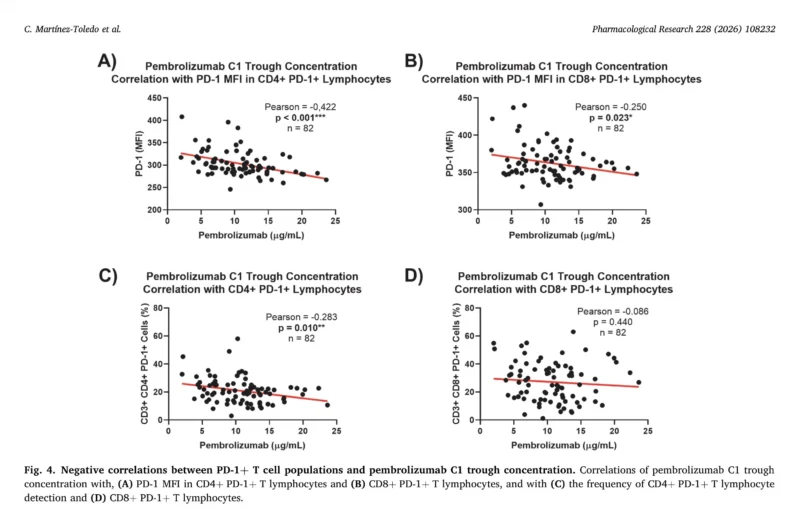
Lower Drug Levels Were Associated With Incomplete PD-1 Blockade
One of the most biologically interesting aspects of the study involved flow cytometry analysis of circulating immune cells.
The investigators found that lower pembrolizumab plasma concentrations correlated with higher detectable free PD-1 expression on CD4+ and CD8+ T cells. Since the assay detects unbound PD-1 receptors, this suggests that patients with lower drug exposure may experience incomplete receptor blockade.
Specifically:
- CD4+ PD-1 MFI negatively correlated with pembrolizumab levels (r = –0.422; p<0.001)
- CD8+ PD-1 MFI also negatively correlated with drug exposure (r = –0.250; p=0.023)
These findings provide pharmacodynamic evidence supporting a biologically plausible exposure-response relationship.
Why This Study Is Important
The study challenges the assumption that pembrolizumab exposure alone can determine response and outcome in patients with different baseline characteristics.
While randomized trials previously suggested PD-1 saturation at standard doses, this real-world analysis indicates that some patients may still experience insufficient systemic exposure during treatment regardless of the cumulative dose.
The findings also align with earlier studies showing associations between pembrolizumab clearance and poor outcomes in NSCLC and melanoma.
If validated prospectively, early pembrolizumab trough measurement could potentially help:
- Identify patients at high risk of treatment failure
- Guide individualized dosing strategies
- Improve pharmacodynamic monitoring
- Support therapeutic drug monitoring approaches in immunotherapy
Limitations of the Study
The authors acknowledge several important limitations.
The study was retrospective and observational, with a relatively small sample size. Pembrolizumab concentrations were measured at a single time point, limiting full pharmacokinetic characterization. Additionally, no external validation cohort was available.
The investigators also note that low drug exposure may partly reflect aggressive disease biology rather than being directly causal.
Nevertheless, the consistency of findings across subgroups and the biological correlations with PD-1 occupancy strengthen the clinical relevance of the results.
Conclusion
This multicenter real-world study suggests that low pembrolizumab plasma levels after the first treatment cycle are associated with poorer progression-free and overall survival in metastatic NSCLC.
The data support the possibility that early pembrolizumab trough concentration could serve as a clinically useful prognostic biomarker, potentially enabling more individualized immunotherapy strategies in the future.
Further prospective studies will be needed to determine whether therapeutic drug monitoring and dose optimization can improve outcomes for patients with low pembrolizumab exposure.
Read full article here