For many patients without targetable driver alterations, Pembrolizumab in Advanced NSCLC has made two years of immune checkpoint blockade a familiar treatment milestone. That time point is built into major pembrolizumab trials, reflected in routine clinical practice, and often accepted as the natural stopping point when patients remain progression-free and clinically well. What has remained much less certain is what happens after treatment stops in the real world. Do most patients relapse quickly? How often is immunotherapy restarted? And what treatments are actually used once pembrolizumab is discontinued?
A new nationwide French cohort study published in JAMA Oncology offers one of the clearest answers so far. Using administrative health data from the French National Health Insurance database, investigators examined outcomes after discontinuation of Pembrolizumab in Advanced NSCLC among patients who completed approximately two years of frontline treatment. The results are reassuring, but they also show that immunotherapy rechallenge is uncommon in routine practice and that radiotherapy often becomes the next step in selected long-term responders.
A Study Focused on a Very Specific Clinical Moment
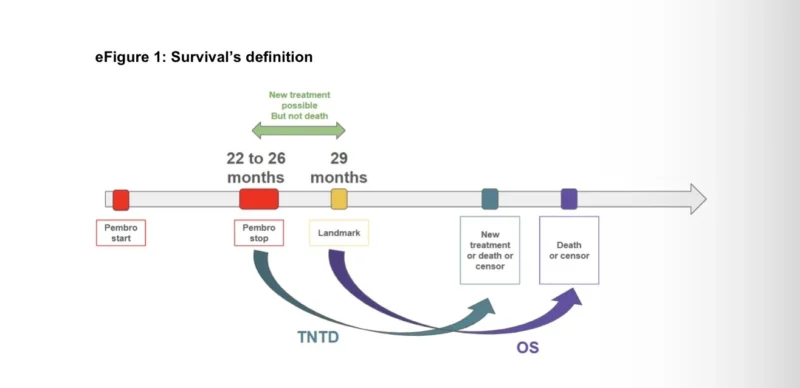
This was not a broad analysis of all patients treated with pembrolizumab. It focused on a highly selected group: long-term survivors who had already done well enough to receive 22 to 26 months of treatment and remain alive at a predefined landmark of 29 months after treatment initiation.
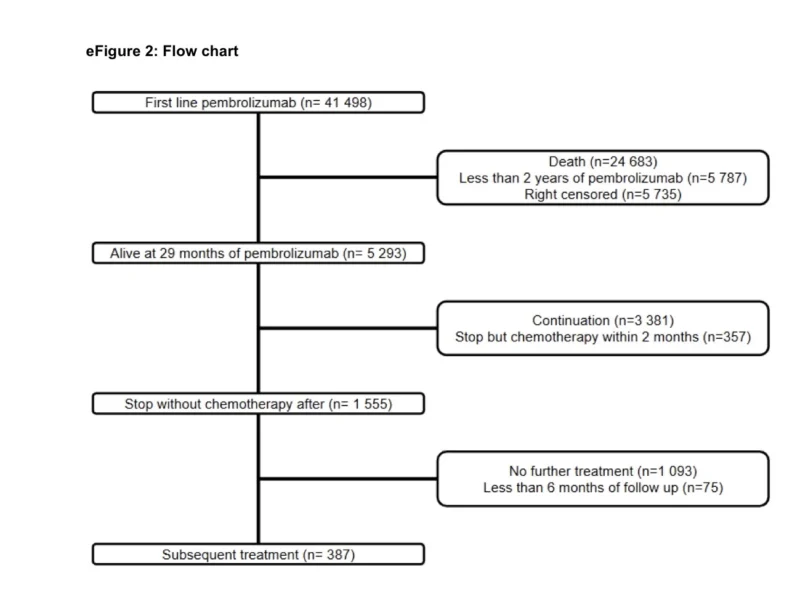
From a total of 41,498 patients initially treated with frontline pembrolizumab in the advanced setting between 2015 and 2022, 5,293 were alive at the landmark time and had completed two years of therapy. Among them, 1,555 discontinued pembrolizumab at two years, and 1,480 had at least six months of follow-up after discontinuation and were included in the main analysis. The median age was 63 years, 36.3% were women, and 41.6% had received pembrolizumab monotherapy rather than pembrolizumab plus chemotherapy.
That design matters. This is not a study about what happens to the average patient with advanced NSCLC on immunotherapy. It is a study about what happens to the subgroup who reaches the two-year finish line and then stops.
© 2026 American Medical Associatio
Survival After Stopping Pembrolizumab Was High
The central message of the study is simple: many of these patients continued to do well long after pembrolizumab ended.
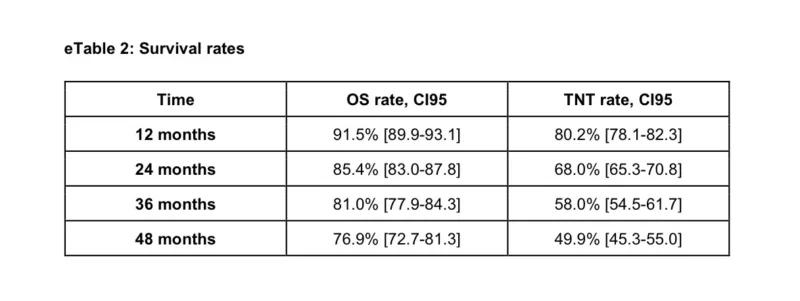
After a median follow-up of 16 months after the landmark, the overall survival rate at 48 months was 76.9% (95% CI, 72.7%–81.3%). That is a striking number in advanced NSCLC, even if it reflects a selected population of long-term responders. It suggests that for many patients who remain alive and stable after two years of pembrolizumab, stopping treatment does not immediately translate into clinical decline.
The study also reported time to next treatment or death, measured from pembrolizumab discontinuation. At 48 months, that rate was 49.9% (95% CI, 45.3%–55.0%). In other words, about half of patients had still not required new systemic treatment or died by that point. This is not the same as progression-free survival, because the database did not include radiologic progression data, but it still provides a clinically meaningful signal: many patients remained off treatment for a substantial period after pembrolizumab stopped.
Most Patients Did Not Need Another Treatment Soon
Perhaps one of the most clinically useful findings is how many patients did not require any additional therapy in the observed period.
Of the 1,480 patients included in the cohort, 1,015 did not receive a new treatment during follow-up. In this group, outcomes remained favorable. Overall survival was 88.4% at 24 months after the landmark and 81.6% at 48 months. That matters because it supports a practice pattern many clinicians have adopted with some uncertainty: stopping pembrolizumab at two years and observing closely rather than automatically continuing treatment beyond the original fixed duration.
This does not prove that stopping is superior to continuing, but it does support the idea that treatment discontinuation at two years is not necessarily followed by rapid relapse in most long-term responders.
What Happened to the Patients Who Needed More Treatment?
Among the 1,480 patients in the study, 387 patients, or 26.1%, received a subsequent therapy after pembrolizumab discontinuation. The first treatment used after stopping pembrolizumab was most often chemotherapy or radiotherapy. Chemotherapy was given first in 200 patients (51.7%), radiotherapy in 183 patients (47.3%), and immunotherapy in only 4 patients (1.0%).
That pattern is important. It shows that when patients eventually needed another intervention, clinicians were far more likely to use local treatment or conventional systemic therapy than to restart checkpoint inhibition.
The radiotherapy signal is especially interesting. In retrospective practice, radiotherapy is often used for oligoprogressive disease or limited symptomatic relapse in patients who previously derived prolonged benefit from immunotherapy. This study cannot fully characterize the radiologic pattern of relapse, but the frequency of radiotherapy strongly suggests that many of these patients were not experiencing diffuse, explosive progression. Instead, at least some appear to have had more limited sites of recurrence or progression that were thought appropriate for local treatment.
© 2026 American Medical Associatio
Immunotherapy Rechallenge Was Rare
One of the most notable findings in the paper is how uncommon immunotherapy rechallenge was.
Although only 4 patients received immunotherapy as the first treatment after pembrolizumab discontinuation, the authors also looked across all subsequent treatment lines. Altogether, just 19 patients received a second course of immune checkpoint blockade. The most frequently used drug in that setting was atezolizumab.
This is a striking real-world contrast to how often rechallenge is discussed in theory. In clinical conversation, restarting immunotherapy after relapse at the two-year mark can seem like an intuitive option, especially if the initial response was deep and durable. But in this national cohort, it was clearly not the dominant strategy.
That does not necessarily mean rechallenge is ineffective. It may reflect clinician caution, lack of strong evidence, patient selection, reimbursement patterns, or preference for other therapies depending on the pattern of relapse. It may also reflect uncertainty about whether progression after a fixed-duration course should be treated biologically as “sensitive” disease.
Outcomes After Retreatment Favored Radiotherapy
The study also described survival after the start of retreatment. Although these comparisons are observational and should be interpreted carefully, they are still clinically informative.
Twelve months after initiation of a new treatment, overall survival was 87.0% (95% CI, 81.6%–92.7%) for patients receiving radiotherapy, 69.9% (95% CI, 61.1%–80.0%) for those receiving chemotherapy, and 61.4% (95% CI, 41.3%–91.4%) for the small group receiving immunotherapy rechallenge.
Radiotherapy therefore appeared associated with the most favorable subsequent outcomes, though this almost certainly reflects patient selection. Patients referred for radiotherapy after long-term pembrolizumab benefit were likely those with limited progression, slower disease biology, or better functional reserve. Even so, the finding is clinically meaningful. It reinforces the idea that local therapy has an important role in selected patients who relapse after stopping immunotherapy at two years.
Chemotherapy remained the most common systemic option and produced more modest outcomes, with 24-month survival of 42.6% and 36-month survival of 33.1% after treatment initiation. These figures remind us that once broader relapse occurs after a durable pembrolizumab response, outcomes become less favorable and treatment decisions more challenging.
Which Patients Were More Likely to Need Another Treatment?
The study also explored characteristics associated with the group that either received a new treatment or died, compared with those who remained alive without retreatment.
Patients in the new treatment or death group were more often male, older, and more likely to have received pembrolizumab monotherapy rather than pembrolizumab plus chemotherapy. They also more often had prior radiotherapy before discontinuation and higher rates of some comorbidities or background medications, including diabetes, antiplatelet agents, lipid-lowering drugs, and beta-blockers.
These associations are interesting, but they should be interpreted cautiously. Without radiologic disease burden, PD-L1 status, performance status, or smoking history, it is hard to know whether these variables are true biological signals or simply markers of treatment selection and baseline frailty. Still, the finding that pembrolizumab monotherapy was more common in the group that later required retreatment may reflect differences in underlying disease biology or in the patient population originally selected for monotherapy.
Why This Study Matters
This study answers a question many clinicians face but few datasets have addressed well: what does the post-two-year pembrolizumab period actually look like in practice?
The answer is reassuring. Many patients who complete two years of pembrolizumab remain alive years later, and most do not immediately need another treatment. Among those who do, chemotherapy and radiotherapy are the dominant next steps, while immunotherapy rechallenge remains uncommon.
That matters because the two-year stopping point has always been supported more by trial design than by strong real-world post-discontinuation data. This paper does not settle the debate over whether some patients should continue longer, but it does support the safety and feasibility of stopping at two years in carefully selected long-term responders.
It also highlights something increasingly important in thoracic oncology: for patients who do well on immunotherapy for a long time, relapse is not always diffuse or catastrophic. In a meaningful subgroup, progression may be limited enough that local treatment becomes a rational and effective bridge.
© 2026 American Medical Associatio
The Limits of the Data
The authors are careful to acknowledge the study’s limitations, and they are important.
First, the database did not contain radiologic progression data, so time to next treatment or death could not be treated as equivalent to progression-free survival. Some patients may have relapsed without needing immediate treatment, while others may have started treatment for reasons not fully captured in the database.
Second, this was a landmark-selected cohort of long-term responders. The results cannot be generalized to all patients with advanced NSCLC treated with pembrolizumab. They apply specifically to those who completed around two years of therapy and were still alive at 29 months.
Third, some retreatment events may have reflected second primary cancers rather than recurrence of the original NSCLC. Residual immortal time bias may also have influenced the estimates. And because the database lacked detailed clinical information, the study could not examine the role of toxicity, PD-L1 expression, or radiologic pattern of relapse in shaping discontinuation and outcomes.
Even with those limitations, the scale of the cohort remains a major strength. To date, this appears to be the largest national real-world analysis focused specifically on patients who completed two years of pembrolizumab in advanced NSCLC.
The Clinical Takeaway
For clinicians, this study offers a message that is both practical and reassuring. If a patient with advanced NSCLC reaches two years of pembrolizumab and remains well, stopping treatment is not equivalent to giving up control of the disease. Many of these patients remain alive and off treatment for years afterward.
When relapse does occur, radiotherapy appears to play an important role, likely in patients with oligoprogressive disease. Chemotherapy remains the most common systemic next step. Immunotherapy rechallenge, by contrast, remains rare in routine care, suggesting that this strategy is still far from standard.
Conclusion
This nationwide French cohort study adds valuable real-world evidence to a question that has become increasingly relevant in the immunotherapy era. Among 1,480 long-term survivors with advanced NSCLC who completed approximately two years of pembrolizumab, survival after discontinuation was high. At 48 months, overall survival was 76.9%, and only about one quarter of patients required a subsequent treatment. When retreatment was needed, chemotherapy and radiotherapy dominated, while immunotherapy rechallenge was uncommon.
The study does not end the debate over fixed-duration immunotherapy, but it strengthens the case that many patients who do well through two years of pembrolizumab can safely stop and remain under observation, with favorable long-term outcomes.