The question of whether fixed-dose or weight-based pembrolizumab offers better value and comparable efficacy has remained clinically relevant since immune checkpoint inhibitors entered routine oncology practice. Although fixed dosing has become standard in many countries because of convenience and pharmacokinetic modeling, concerns persist regarding overtreatment in lighter patients, drug waste, and financial sustainability, particularly in resource-limited healthcare systems.
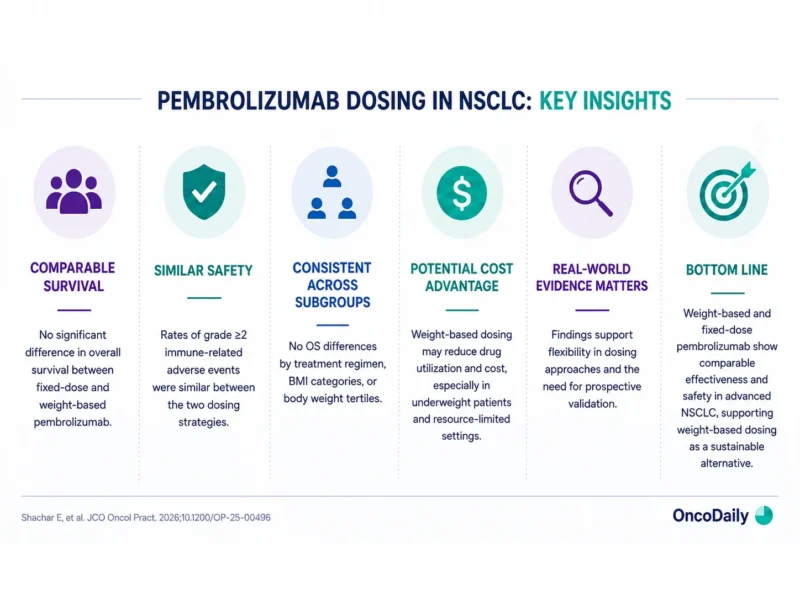
A new real-world study published in the Journal of Clinical Oncology Oncology Practice evaluated whether fixed-dose pembrolizumab and weight-based pembrolizumab achieve comparable outcomes in patients with advanced non–small cell lung cancer (NSCLC). The investigators found no significant differences in overall survival or immune-related toxicity between the two dosing approaches, supporting the possibility that weight-based dosing may remain a clinically reasonable and economically attractive alternative in selected settings.
Why Pembrolizumab Dosing Matters in NSCLC
Pembrolizumab, an anti–PD-1 immune checkpoint inhibitor, has become a foundational treatment for advanced NSCLC, both as monotherapy and in combination with platinum-based chemotherapy. The currently approved standard regimen in many settings is a fixed dose of 200 mg every three weeks.
The shift from weight-based dosing to fixed dosing was largely driven by pharmacokinetic simulations suggesting similar drug exposure across body sizes. However, real-world populations often differ substantially from the highly selected cohorts included in registration trials. Questions remain about whether fixed dosing may expose lower-weight patients to unnecessarily high drug amounts without improving efficacy.
These concerns are especially important in countries where immunotherapy costs significantly limit treatment access. Even modest reductions in drug utilization could have major implications for healthcare systems and patient affordability.
Study Design and Patient Population
The study, led by Eliya Shachar and colleagues, analyzed a retrospective cohort of 414 patients with advanced NSCLC treated with pembrolizumab between 2016 and 2021 at a single institution.
Patients received either:
- Fixed-dose pembrolizumab: 200 mg every three weeks
- Weight-based pembrolizumab: 2 mg/kg every three weeks
Among the total cohort:
- 339 patients (81.9%) received fixed dosing
- 75 patients (18.1%) received weight-based dosing
The primary endpoint was overall survival (OS), while secondary endpoints included immune-related adverse events (irAEs).
To minimize confounding and baseline imbalance, investigators used inverse probability of treatment weighting (IPTW), followed by multivariable Cox regression analyses and subgroup evaluations.

Comparable Survival Outcomes Between Dosing Strategies
The analysis demonstrated no statistically significant difference in overall survival between the two dosing approaches.
Median OS was:
- 18.4 months with fixed-dose pembrolizumab
- 23.2 months with weight-based pembrolizumab
The hazard ratio was 0.73 (95% CI, 0.49–1.10; P = .11), indicating no significant survival advantage for either strategy.
Importantly, the lack of survival difference remained consistent across multiple subgroup analyses.
When patients were stratified according to treatment regimen:
- ICI monotherapy
- ICI combined with platinum-doublet chemotherapy
no meaningful OS differences emerged between fixed-dose and weight-based approaches.
Similarly, within the weight-based dosing cohort, survival outcomes remained consistent across:
- Different body mass index (BMI) categories
- Body weight tertiles
These findings suggest that lower-weight patients did not experience inferior outcomes when treated with weight-adjusted pembrolizumab dosing.
Safety Outcomes Were Also Similar
Immune-related adverse events are a central concern during checkpoint inhibitor treatment, particularly when discussing dose intensity and drug exposure.
In this study, rates of grade 2 or higher immune-related adverse events did not significantly differ between the two groups (P = .52).
This observation supports the possibility that reducing pembrolizumab exposure through weight-based dosing does not appear to compromise safety or increase toxicity risk.
The investigators also noted that, in multivariable analyses, survival outcomes were more strongly influenced by:
- ECOG performance status
- Number of treatment cycles received
rather than by pembrolizumab dosing strategy itself.
The Economic and Global Oncology Implications
One of the most important aspects of this analysis is its potential relevance to healthcare sustainability.
Checkpoint inhibitors remain among the most expensive systemic therapies in oncology. Fixed-dose regimens may substantially increase total drug utilization in underweight patients, particularly in Asian, Middle Eastern, and lower-income populations where average body weight is often lower than in Western registration cohorts.
The authors suggest that weight-based pembrolizumab dosing could represent a cost-effective alternative without compromising efficacy.
This issue is particularly relevant in global oncology settings where:
- Drug reimbursement remains limited
- Healthcare budgets are constrained
- Access to immunotherapy remains unequal
In many regions, the financial burden associated with checkpoint inhibitors directly affects whether patients can receive treatment at all.
Although simplified administration and reduced pharmacy complexity favor fixed dosing operationally, the economic advantages of weight-based dosing may justify reconsideration in selected healthcare systems.
Real-World Evidence Adds Important Context
Randomized clinical trials often provide the foundation for dosing strategies, but real-world analyses remain valuable because they include more heterogeneous patient populations.
This cohort reflects routine clinical practice and therefore offers insight into how pembrolizumab dosing performs outside tightly controlled trial environments.
The findings align with previous pharmacokinetic analyses suggesting a relatively flat exposure-response relationship for pembrolizumab across clinically relevant dosing ranges.
In other words, beyond a certain threshold, increasing pembrolizumab exposure may not meaningfully improve antitumor activity.
That biological principle may partially explain why lower drug exposure with weight-based dosing did not appear to reduce efficacy in this study.
Important Limitations of the Analysis
Despite the clinically relevant findings, the study has important limitations.
First, the analysis was retrospective and conducted at a single center, introducing the possibility of residual confounding despite IPTW adjustment.
Second, the weight-based cohort was substantially smaller than the fixed-dose cohort, potentially limiting statistical power.
Third, treatment selection was not randomized, meaning clinicians may have preferentially selected one strategy over another based on patient characteristics not fully captured in the dataset.
Additionally, the study focused on real-world effectiveness and survival rather than pharmacokinetic measurements or biomarker-driven analyses.
The authors therefore appropriately emphasize that prospective validation would be needed before broad policy changes regarding pembrolizumab dosing could be recommended.
What This Means for Clinical Practice
The study does not challenge the current standard use of fixed-dose pembrolizumab in NSCLC. However, it raises an increasingly important discussion regarding value-based oncology care.
As immunotherapy use expands globally and treatment durations continue to increase, dosing optimization may become an important strategy for improving sustainability without compromising outcomes.
The findings suggest that weight-based pembrolizumab dosing may remain a clinically acceptable option in selected circumstances, particularly:
- Underweight patients
- Resource-limited healthcare systems
- Countries with restricted reimbursement access
- Institutions prioritizing drug conservation strategies
At the same time, prospective comparative studies would still be necessary before any major guideline shifts occur.
Conclusion
This real-world analysis found that fixed-dose and weight-based pembrolizumab produced comparable survival outcomes and similar immune-related toxicity rates in patients with advanced NSCLC.
Although fixed dosing remains the dominant global standard, the findings suggest that weight-based pembrolizumab may offer a potentially cost-effective alternative without compromising clinical effectiveness, especially in lower-weight populations and resource-constrained settings.
As oncology increasingly moves toward balancing innovation with sustainability, studies like this highlight the growing importance of treatment optimization strategies alongside drug development itself.