KRAS G12C NSCLC has entered the era of targeted therapy, but the benefit of currently available inhibitors remains limited by adaptive resistance. In clinical practice, sotorasib and fulzerasib have both demonstrated activity in previously treated KRAS G12C-mutant NSCLC, yet responses remain incomplete and progression-free survival is still relatively short. In the study explored one of the most clinically relevant resistance mechanisms in this setting: EGFR-driven feedback reactivation of RAS-MAPK signaling. Their work tested whether combining cetuximab with the KRAS G12C inhibitors fulzerasib or sotorasib could improve antitumor activity in preclinical NSCLC models.
The rationale is compelling. KRAS G12C inhibitors bind the inactive GDP-bound form of KRAS, but upstream EGFR signaling can shift KRAS back toward its active GTP-bound state, restoring downstream signaling and weakening drug activity. This mechanism has already been implicated in resistance to KRAS G12C inhibition in colorectal cancer and increasingly in lung cancer as well. The present study asked whether EGFR inhibition with cetuximab could blunt that escape pathway and enhance the effect of fulzerasib or sotorasib in KRAS G12C NSCLC cells.
Why This Question Matters Clinically
The clinical relevance of this problem is clear. As reviewed in the paper, sotorasib monotherapy produced response rates around 37% to 41% and median progression-free survival in the range of 5.6 to 6.8 months in previously treated KRAS G12C NSCLC, while fulzerasib achieved an objective response rate of 49.1% and median progression-free survival of 9.7 months in a phase II study (Skoulidis et al., 2021; de Langen et al., 2023; Zhou et al., 2024; Olmo-González et al., 2026). These are important advances, but the benefits are neither universal nor durable. The authors therefore focused on whether EGFR cotreatment could deepen and extend response, and whether biomarkers such as MIG6, MRAS, YAP1, ASS1, or fumarate might help explain differential sensitivity.
Study Design and Experimental Approach
The investigators evaluated several KRAS G12C-mutant cell lines, with particular mechanistic emphasis on H358, H23, and H2030 NSCLC cells. These models differ biologically and in drug sensitivity, making them useful for understanding heterogeneity of response. Fulzerasib and sotorasib were tested alone and in combination with cetuximab under conditions designed to activate EGFR signaling, including serum starvation plus exogenous human EGF.
Cell viability was assessed using MTT assays, and synergy was measured using the Chou-Talalay combination index. Western blot analyses examined EGFR pathway signaling, ERK and AKT reactivation, and the expression of resistance-related proteins including MRAS, YAP1, MIG6, and ASS1. Fumarate levels were measured as a possible metabolic correlate. The authors also conducted in vivo experiments using H358 xenografts in nude mice treated with fulzerasib, cetuximab, or the combination.

Key Findings in Cell Viability and Synergy
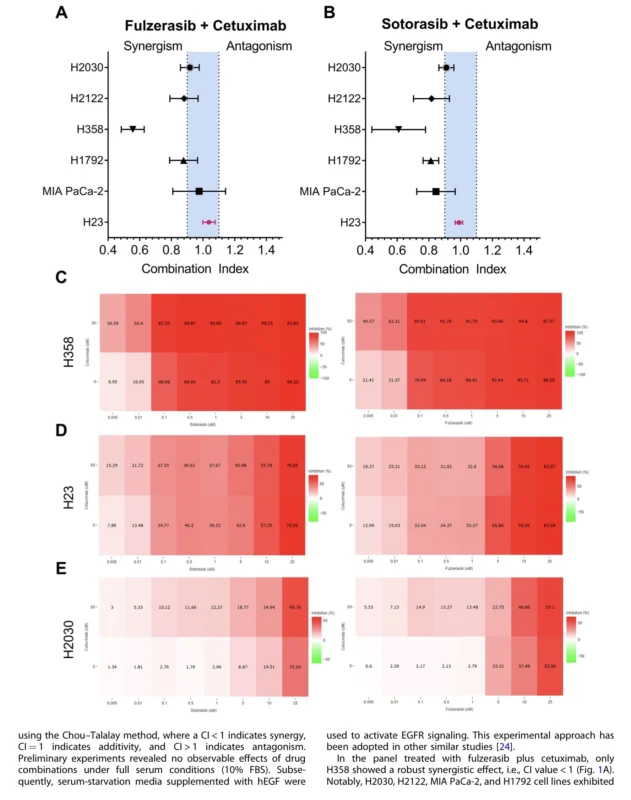
The study showed that fulzerasib and sotorasib both inhibited KRAS G12C-mutant cell growth, but their effects varied substantially across cell lines. H358 cells were highly sensitive to both agents, with IC50 values below 5 nM for sotorasib and 6.3 nM for fulzerasib in full serum conditions. H23 cells were less sensitive, and H2030 cells were largely resistant, particularly to sotorasib and fulzerasib under some conditions.
When cetuximab was added, the strongest synergistic effect was observed in H358 cells. In this model, both fulzerasib plus cetuximab and sotorasib plus cetuximab enhanced inhibition of cell proliferation beyond what was achieved with KRAS G12C inhibition alone. In H23 cells, the effect was additive rather than strongly synergistic. In H2030 cells, the addition of cetuximab increased inhibitory activity but did not reproduce the robust synergy seen in H358. These results suggest that not all KRAS G12C NSCLC models are equally dependent on EGFR feedback signaling, and that cetuximab-based cotreatment may be especially beneficial in a biologically defined subset.
Mechanistic Insights: ERK, AKT, MRAS, and YAP1
A major contribution of this paper is the mechanistic analysis of pathway reactivation. Fulzerasib and sotorasib both suppressed signaling initially, but rebound activation of ERK was observed over time, particularly at 48 to 72 hours. In H358 cells, cetuximab delayed this ERK reactivation, supporting the idea that EGFR signaling contributes directly to resistance. Similar delay was observed in H23 and H2030 cells, although the overall biological consequences differed.
The combination also affected other pathways. In H358 cells, fulzerasib plus cetuximab suppressed AKT phosphorylation, MRAS, and activated YAP1 more effectively than monotherapy. This is notable because MRAS and YAP1 have emerged as potential mediators of adaptive resistance to KRAS G12C inhibition. Previous work has linked MRAS-SHOC2-PP1C signaling and YAP1 activation to ERK pathway rebound and KRAS inhibitor resistance, and this study strengthens that link in lung cancer models (Adachi et al., 2023; Olmo-González et al., 2026).
The less sensitive H2030 model behaved differently. In these cells, YAP1 and MRAS were not fully suppressed by cotreatment and in some settings were more strongly induced, especially with sotorasib-based combinations. This suggests that some KRAS G12C NSCLC tumors may be relatively KRAS-independent or may use bypass programs that are not fully blocked by EGFR inhibition alone.

MIG6, ASS1, and Fumarate as Potential Biomarkers
The authors also examined several potential biomarkers. MIG6, a known negative regulator of EGFR, was consistently downregulated after treatment with fulzerasib or sotorasib, both alone and in combination with cetuximab, across all three cell lines. Cetuximab alone did not reduce MIG6. This finding is interesting because reduced MIG6 can release EGFR signaling and facilitate bypass activation. However, since MIG6 fell in both sensitive and resistant models, it may not by itself distinguish tumors that will respond best to EGFR cotreatment.
ASS1 and fumarate offered another metabolic angle. ASS1 was low at baseline in H23 and H2030 cells but increased with KRAS G12C inhibition, with or without cetuximab. Fumarate levels also rose over time, especially by 72 hours, in all three cell lines. The biological meaning of this pattern remains uncertain, but the authors suggest these metabolic alterations could be explored in future translational work as markers of therapeutic response or adaptation.
In Vivo Evidence Strengthens the Signal
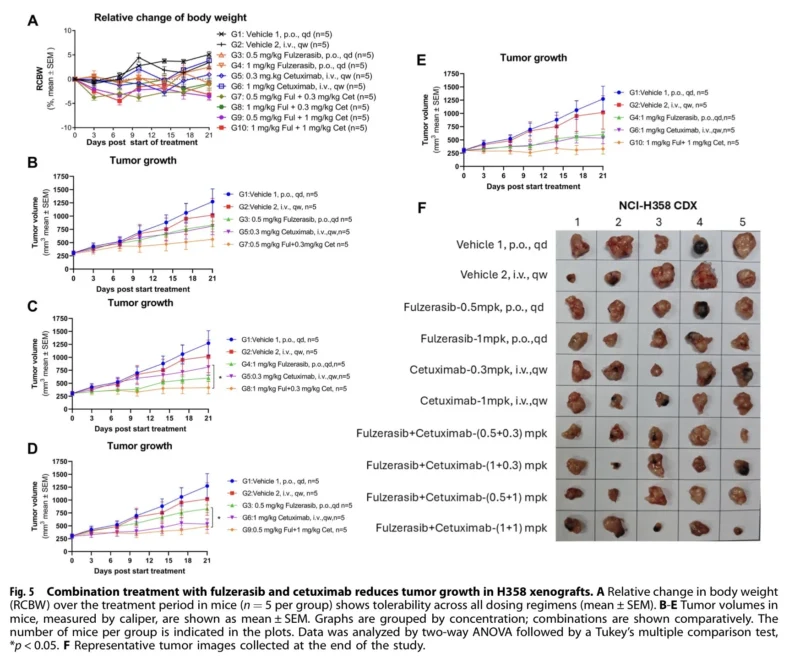
The in vivo xenograft experiments provide some of the most compelling data in the paper. In mice bearing subcutaneous H358 tumors, fulzerasib and cetuximab each inhibited tumor growth as single agents, but the combination produced the strongest effect. At the highest tested concentrations, tumor growth inhibition reached 97%. Importantly, no major body weight loss or treatment-related mortality was observed, suggesting acceptable tolerability in this model.
These findings support the in vitro observations and reinforce the concept that cetuximab can enhance KRAS G12C inhibitor activity in selected lung cancer models.
Clinical Interpretation
The most important takeaway from this paper is not that every KRAS G12C NSCLC should automatically receive EGFR blockade, but rather that a biologically defined subset may derive substantial benefit. The H358 model appears to represent tumors in which EGFR feedback is a dominant escape mechanism and in which cetuximab can restore deeper pathway suppression. Other models, particularly H2030, appear more complex and may require additional strategies beyond EGFR cotreatment, potentially including YAP-targeted approaches.
This nuance matters because the findings mirror early clinical observations. As the authors note, first-line fulzerasib plus cetuximab has already shown promising activity in KRAS G12C NSCLC, with a reported response rate of 68.9% and median progression-free survival of 12.5 months in the KROCUS study, although mature survival data are still pending (Majem et al., 2025; Olmo-González et al., 2026). The present preclinical work provides a mechanistic framework for why such combinations may work and why patient selection will be important.
Conclusion
Olmo-González and colleagues provide strong preclinical evidence that cetuximab can enhance the antitumor activity of fulzerasib and sotorasib in selected KRAS G12C NSCLC models. The most robust effect was seen in H358 cells, where cotreatment suppressed ERK, AKT, MRAS, and YAP1 signaling and translated into major tumor growth inhibition in vivo. In less sensitive models such as H23 and H2030, the benefit was more limited, highlighting the biological heterogeneity of KRAS G12C lung cancer.
The study strengthens the case for EGFR-KRAS cotreatment in NSCLC and identifies a set of candidate biomarkers, including MRAS, YAP1, MIG6, ASS1, and fumarate, that may help refine patient selection in future translational and clinical studies. More broadly, it underscores a central lesson of targeted therapy: blocking the driver oncogene is often not enough, and understanding feedback circuitry is essential to extending benefit.
You can read full article here