The phase 2 ICARUS-LUNG01 study adds an important translational layer to the rapidly evolving story of antibody-drug conjugates in lung cancer. Published in Cancer Cell, the study evaluated datopotamab deruxtecan (Dato-DXd) in patients with previously treated advanced non-small cell lung cancer (NSCLC), while also performing serial tumor and blood-based analyses to better understand why some tumors respond and others develop resistance (Planchard et al., 2026).
The clinical message is clear but not complete: Dato-DXd demonstrated activity and a manageable safety profile in a pretreated NSCLC population, with stronger signals in non-squamous tumors. The biological message may be even more important. Response to Dato-DXd appears to depend on more than TROP2 expression alone. TROP2 localization, ADC internalization, DNA repair activation, and immune microenvironment changes may all influence sensitivity or resistance.
© 2026 The Author(s).
Why ICARUS-LUNG01 Matters
TROP2-directed antibody-drug conjugates are becoming increasingly relevant in thoracic oncology. Dato-DXd, which links a TROP2-directed antibody to a topoisomerase I inhibitor payload, has already shown activity in advanced NSCLC and received accelerated FDA approval in 2025 for previously treated EGFR-mutated NSCLC after EGFR TKI and platinum-based therapy, based on the TROPION-Lung program (Ahn et al., 2025; Sands et al., 2025).
However, as ADCs move into broader use, one major question remains unresolved: which patients are most likely to benefit?
Unlike classic targeted therapies, ADC response is not determined only by the presence or absence of a single mutation. It may depend on target expression, target internalization, linker-payload biology, tumor DNA damage response, and immune activation. ICARUS-LUNG01 was designed to explore this complexity through a prospective clinical trial with integrated translational sampling.
Study Design and Patient Population
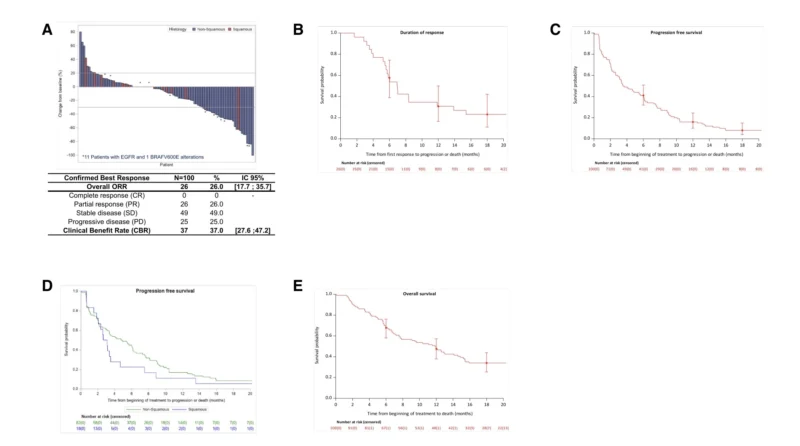
ICARUS-LUNG01 enrolled 100 patients with previously treated advanced NSCLC across 8 French sites. All patients received Dato-DXd 6.0 mg/kg intravenously every 3 weeks until disease progression or unacceptable toxicity.
This was a clinically relevant pretreated population. Most patients had non-squamous histology, which represented 82% of the cohort. Nearly all patients had received prior platinum-based chemotherapy, and 89% had previously received anti-PD-1 or anti-PD-L1 therapy. The median number of prior systemic treatment lines was 2, and 17% had received prior targeted therapy.
Tumor profiling was available for most patients. Among those with known genomic alterations, EGFR mutations, KRAS mutations, and one BRAF V600E alteration were reported. PD-L1 expression was below 1% in more than half of evaluable patients.
© 2026 The Author(s).
Clinical Activity: Stronger Signal in Non-Squamous NSCLC
Dato-DXd achieved a confirmed objective response rate of 26.0% in the overall population. The signal was clearly stronger in non-squamous tumors, where the ORR reached 30.5%, compared with 5.6% in squamous carcinoma.
Median progression-free survival was 3.6 months overall. Again, histology appeared important: median PFS was 4.8 months in non-squamous NSCLC and 2.9 months in squamous disease. Median overall survival was 11.9 months, with longer survival in non-squamous tumors than in squamous tumors.
The study also reported notable response rates in molecularly defined subgroups, although these analyses were limited by small numbers. Patients with EGFR or BRAF V600E-mutated tumors had an ORR of 50.0%, while patients with KRAS-mutated tumors had an ORR of 63.6%. These subgroup findings are hypothesis-generating and require validation in larger datasets.
Safety: Manageable, With Low Confirmed ILD
The safety profile was consistent with the known toxicity pattern of Dato-DXd. Treatment-related adverse events occurred in 88% of patients, while grade 3 or higher treatment-related adverse events occurred in 24%.
The most common treatment-related adverse events were stomatitis, nausea, alopecia, and fatigue, most of which were grade 1 or 2. Stomatitis was the most frequent toxicity, reported in 48% of patients, with grade 3 or higher events in 11%.
Interstitial lung disease remains a key safety concern with DXd-based ADCs. In ICARUS-LUNG01, 8 cases of suspected ILD were reviewed, but only one case was confirmed as Dato-DXd-related ILD by an independent adjudication committee, and it was grade 1. Three grade 5 toxicities were considered probably related to treatment, including two pneumonitis events and one hepatobiliary disorder. This makes careful monitoring essential, even when confirmed adjudicated ILD appears uncommon.
© 2026 The Author(s).
TROP2 Expression Alone Was Not Enough
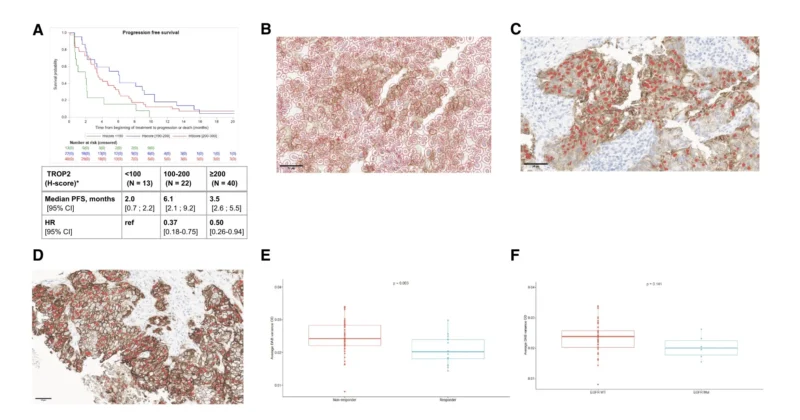
One of the most important findings from ICARUS-LUNG01 is that standard TROP2 expression by immunohistochemistry did not fully explain response.
Patients with very low TROP2 expression appeared less likely to respond, and tumors with TROP2 H-score ≥100 showed longer PFS than tumors with lower expression. However, higher TROP2 expression did not translate into a simple linear relationship with objective response.
This matters because it challenges the idea that TROP2 ADC selection can rely only on a single expression cutoff. Instead, the study suggests that where TROP2 is located inside the tumor cell may matter as much as how much TROP2 is present.
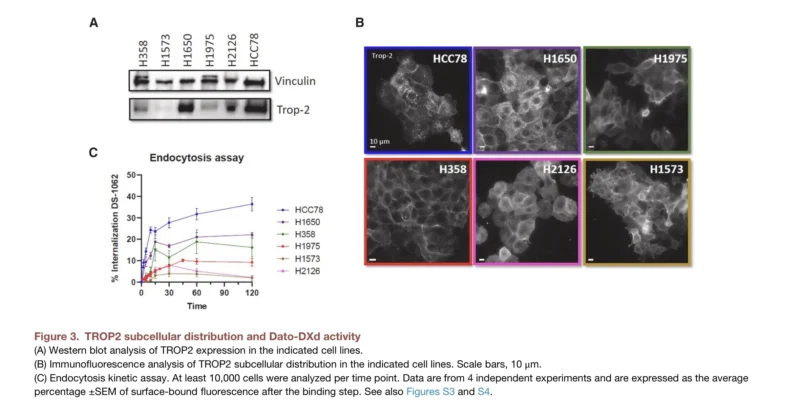
Using image-based analysis, investigators found that responders had more homogeneous cytoplasmic TROP2 staining, while non-responders showed more heterogeneous staining patterns. This may reflect better TROP2 internalization and more efficient ADC uptake. In preclinical NSCLC cell lines, cytoplasmic TROP2 localization correlated with faster Dato-DXd internalization, stronger DNA damage response, and greater drug sensitivity.
DNA Repair Activation May Drive Resistance
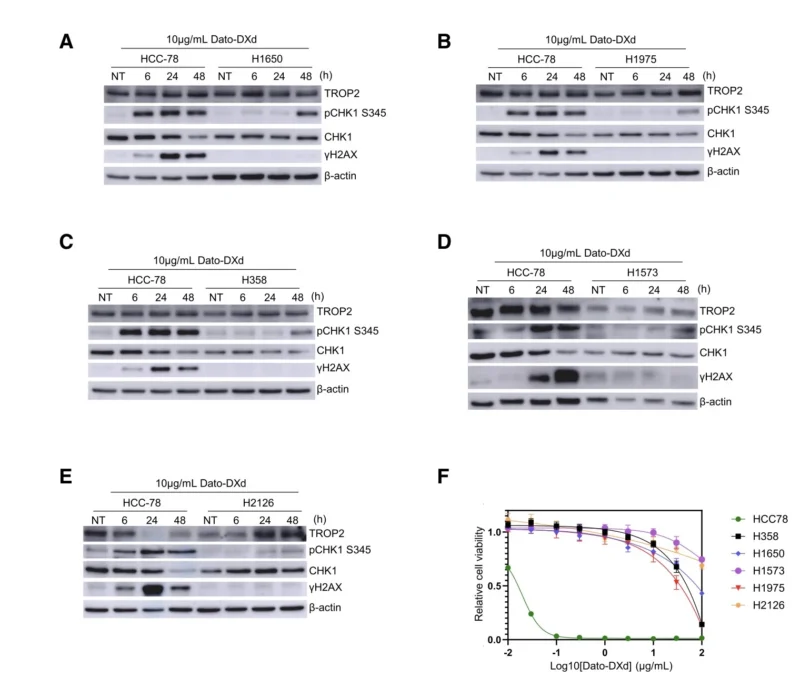
Dato-DXd delivers a topoisomerase I inhibitor payload, meaning that tumor cell killing depends partly on DNA damage. ICARUS-LUNG01 found that non-responding tumors showed activation of DNA repair pathways after treatment exposure.
This suggests a plausible resistance mechanism: tumors capable of rapidly repairing payload-induced DNA damage may be less sensitive to Dato-DXd. The study also explored SLFN11, a biomarker linked to sensitivity to DNA-damaging agents. Responding tumors showed numerically higher SLFN11-positive tumor cells, and laboratory experiments demonstrated that loss of SLFN11 markedly reduced sensitivity to Dato-DXd.
These findings position SLFN11 and DNA repair activity as potential future biomarkers for DXd-based ADC response, although clinical validation is still needed.
© 2026 The Author(s).
Immune Activation May Support Response
The study also found that response to Dato-DXd may involve the tumor immune microenvironment. Responders showed activation of immune-related pathways and increases in immune cell populations after treatment, including CD4-positive T cells, CD8-positive T cells, cytotoxic immune subsets, and CD68-positive macrophages.
This supports the broader concept that ADCs are not only chemotherapy delivery systems. By inducing DNA damage and tumor cell death, they may also trigger immune signaling pathways, including interferon-related responses. This may help explain why immune activation was associated with response in ICARUS-LUNG01.
Clinical Interpretation
ICARUS-LUNG01 confirms that Dato-DXd has clinical activity in previously treated advanced NSCLC, particularly in non-squamous disease. But its deeper contribution lies in the biomarker work.
The study suggests that ADC response in lung cancer is multidimensional. TROP2 expression, TROP2 internalization, DNA repair capacity, SLFN11 expression, and immune activation may all contribute to whether a patient benefits from Dato-DXd.
This is a critical message for the ADC era. The future of ADC therapy will likely require composite biomarkers rather than single-marker selection. Larger validation cohorts from studies such as TROPION-Lung01, TROPION-Lung05, and ongoing Dato-DXd trials will be needed before these findings can guide routine clinical decision-making.
© 2026 The Author(s).
Clinical Takeaway
ICARUS-LUNG01 shows that Dato-DXd is active and generally manageable in pretreated advanced NSCLC, with greater benefit in non-squamous tumors. More importantly, it begins to explain the biology of response and resistance.
The study suggests that tumors may respond better when TROP2 is internalized efficiently and when immune activation occurs after treatment. Resistance may be linked to poor cytoplasmic TROP2 localization and activation of DNA repair pathways.
For thoracic oncology, ICARUS-LUNG01 is not just another phase 2 ADC study. It is a step toward understanding how to use TROP2-directed ADCs more precisely.


