The rise of neoadjuvant and perioperative immunotherapy has changed the treatment landscape for resectable non-small cell lung cancer. For many patients with stage II and III disease, especially those without EGFR or ALK alterations, chemoimmunotherapy is no longer a future possibility. It is already part of current standard treatment pathways. The oncologic gains have been clear, with higher major pathologic response rates, more pathologic complete responses, and better long-term outcomes. What has remained less clear is the surgical price of that progress. Does preoperative immunotherapy make surgery more difficult, more dangerous, or less feasible in real-world thoracic practice?
A new systematic review and meta-analysis by Bertoglio and colleagues helps answer that question by pooling prospective data from 27 trials involving 2,691 patients with resectable NSCLC treated with immune checkpoint inhibitor-based regimens before surgery. The conclusion is reassuring, but not simplistic: surgery after immunotherapy-based treatment is feasible and generally safe, yet it comes with distinct perioperative challenges that require careful multidisciplinary coordination and substantial surgical experience (Bertoglio et al., 2026).
Why This Question Matters Now
The surgical implications of immunotherapy are no longer theoretical. As checkpoint inhibitors have moved into earlier-stage NSCLC, thoracic surgeons are increasingly operating in tissues altered by immune activation, inflammation, and fibrosis. That matters because technically successful lung cancer surgery depends on more than resection alone. It depends on safe hilar dissection, preservation of critical structures, judgment about approach, and management of adhesions or altered tissue planes. If preoperative systemic treatment changes those planes, then the conversation shifts from “Can we improve tumor response?” to “What does that response mean in the operating room?”
That is the central importance of this meta-analysis. It does not ask whether neoadjuvant chemoimmunotherapy improves cancer outcomes. That question has already been answered in major trials. Instead, it focuses on something equally important for daily practice: what happens when these patients actually get to surgery?
How The Meta-Analysis Was Built
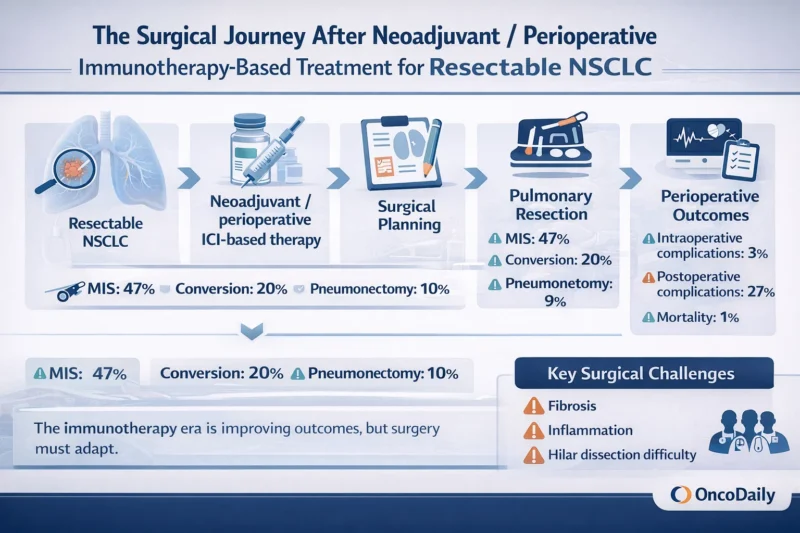
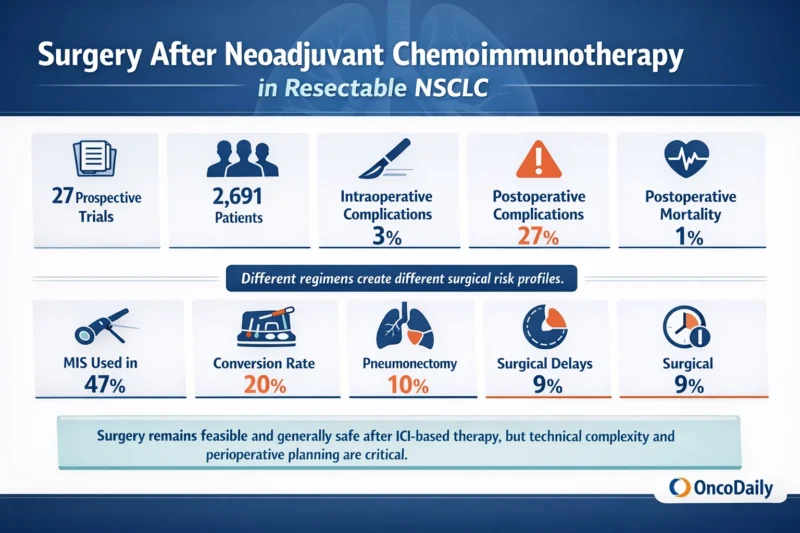
The authors performed a systematic search across PubMed, EMBASE, Scopus, Cochrane CENTRAL, and Web of Science up to January 2025, following PRISMA methodology. They included only prospective single-arm trials and randomized controlled trials that reported surgical outcomes after preoperative immune checkpoint inhibitor-based treatment, either with chemotherapy or without it. In total, 27 trials and 2,691 patients were included in the quantitative synthesis (Bertoglio et al., 2026).
This design choice is important. By focusing on prospective trial populations, the analysis avoids some of the inconsistency and selection bias that often affect retrospective surgical series. At the same time, it means the results come from relatively selected patient groups treated in structured clinical trial environments, which is helpful for internal validity but may underestimate some of the challenges seen in routine practice.
The Main Surgical Message Is Reassuring
The broad finding of the paper is that surgery after ICI-based treatment remains achievable and generally safe. The pooled event proportion for intraoperative complications was 0.03, postoperative complications 0.27, and postoperative mortality 0.01. In practical terms, that means intraoperative complications were uncommon, postoperative complications occurred in roughly one quarter of patients, and postoperative death was rare (Bertoglio et al., 2026).
These numbers do not suggest a collapse of surgical feasibility in the immunotherapy era. They suggest that surgery remains a realistic and acceptable part of multimodality treatment, even after neoadjuvant or perioperative immunotherapy-based therapy. That point matters because there has been real concern among thoracic surgeons that immune-related fibrosis and hilar inflammation could make resection substantially more hazardous. This analysis does not support that kind of alarmist view.
Still, “feasible” does not mean “easy,” and “safe” does not mean “unchanged.”
Surgery Is Still Feasible, But It Is Not Uncomplicated
One of the most relevant findings in the meta-analysis is the level of technical burden that still accompanies these operations. Minimally invasive surgery was used in 47% of resections, and the pooled conversion rate to thoracotomy was 20%. Surgical delay occurred in 9% of cases, and pneumonectomy was required in 10% (Bertoglio et al., 2026).
Those numbers tell an important story. Even if mortality remains low, surgery after neoadjuvant immunotherapy is not identical to upfront surgery. A 20% conversion rate is not trivial. It reflects the fact that preoperative treatment may change anatomy enough to make an initially planned minimally invasive approach more difficult to complete safely. Likewise, a 10% pneumonectomy rate raises questions about extent of resection and how often inflammation or local complexity may influence the operative plan.
In other words, this is not a setting for casual surgical optimism. These are operations that appear manageable, but they may demand more flexibility, more experience, and more readiness to escalate the operative approach when needed.
Chemoimmunotherapy And Immunotherapy Alone Do Not Look The Same
A particularly useful part of the study is the meta-regression comparing chemoimmunotherapy protocols with immunotherapy-only protocols. The authors found that chemoimmunotherapy was associated with higher rates of intraoperative complications and surgery omission, whereas immunotherapy-alone regimens were associated with higher postoperative mortality. No significant differences were seen in pneumonectomy rate, minimally invasive surgery use, or conversion rate between the two strategies (Bertoglio et al., 2026).
This is one of the more nuanced findings in the paper. It suggests that not all immunotherapy-based preoperative regimens shape surgical risk in the same way. Chemoimmunotherapy may create more intraoperative complexity or interfere more often with getting patients to surgery on time. Immunotherapy alone, meanwhile, may carry a different postoperative risk profile.
The paper does not overstate these findings, and it should not. Meta-regression is helpful for signal detection, but it cannot replace direct randomized comparison of surgical outcomes by regimen type. Still, the message is clinically relevant: surgeons and multidisciplinary teams should not assume that all neoadjuvant immunotherapy strategies are surgically interchangeable.
What These Results Mean For Thoracic Surgeons
For surgeons, the practical implication is not that immunotherapy makes surgery impossible. It is that surgery after immunotherapy should be approached as a different technical setting from untreated resection.
Immune-related tissue effects may complicate hilar dissection, blur anatomic planes, and increase the chance that a minimally invasive plan will need to be converted. That does not make surgery inappropriate. It means surgeons should anticipate difficulty earlier, plan more carefully, and work in settings where anesthesia, oncology, radiology, pathology, and postoperative support are tightly integrated.
This is why the authors emphasize multidisciplinary coordination and centralization. Not every resectable NSCLC case needs to be concentrated in a referral center, but once surgery follows checkpoint inhibitor-based therapy, especially in borderline resectable or more locally advanced disease, experience matters more, not less (Bertoglio et al., 2026).
The Oncologic Era Has Changed Faster Than The Surgical Culture
One of the most interesting tensions this paper exposes is that oncology has moved quickly into the perioperative immunotherapy era, while surgical evidence has lagged behind. Major trials have established pathologic and survival benefit, but the surgical literature has often consisted of smaller institutional experiences, heterogeneous definitions of complications, and mixed impressions about technical difficulty.
This meta-analysis helps stabilize that conversation. It shows that surgeons are not facing a catastrophic postoperative environment after chemoimmunotherapy. At the same time, it confirms that the technical concerns are real enough to justify caution. That balance is valuable. It protects against two extremes: excessive fear that might make surgeons hesitant to operate, and excessive enthusiasm that might underestimate the demands of these procedures.
Important Limitations
The authors appropriately acknowledge several limitations. Heterogeneity was substantial for some key outcomes, particularly postoperative complications and minimally invasive surgery use. That means pooled results should be interpreted carefully, especially when translating them into expectations for individual centers. Differences in patient selection, stage distribution, operative expertise, perioperative care pathways, and reporting standards likely all contributed to the observed variability (Bertoglio et al., 2026).
Another important limitation is that these were prospective trial populations, not broad real-world cohorts. Trial patients are generally better selected, more closely monitored, and more likely to be treated in experienced centers. As a result, routine clinical practice may include greater complexity than this analysis captures.
Still, those limitations do not erase the value of the study. They mainly define how it should be used: as a high-quality overview of surgical feasibility in the immunotherapy era, not as a rigid prediction tool for every operating room.
What This Meta-Analysis Adds To The Field
The key contribution of this paper is clarity. It tells the field that surgery after neoadjuvant or perioperative immunotherapy-based treatment in resectable NSCLC is not only possible, but generally safe in selected patients. But it also makes clear that these operations are not simply standard resections performed later in the treatment timeline. They are shaped by the biology and tissue consequences of prior systemic therapy.
That distinction matters. It means surgical planning in resectable NSCLC must now incorporate not just stage and anatomy, but treatment pathway. As more patients receive preoperative checkpoint inhibitor-based regimens, perioperative strategy becomes inseparable from surgical judgment.
Bottom Line
This meta-analysis supports an important conclusion for modern thoracic oncology: surgery after neoadjuvant or perioperative immunotherapy-based therapy for resectable NSCLC is feasible and safe, but it is not surgically neutral. Postoperative mortality remains low, intraoperative complications are uncommon, and resection remains achievable in most appropriately selected patients. But conversion, delay, omission, and technical complexity remain real concerns, especially as treatment protocols differ. The strongest practical message is not just that surgery can still be done. It is that it should increasingly be done in coordinated multidisciplinary programs with surgeons who are experienced in operating after modern systemic therapy.
You can read full abstract here