Perioperative immunotherapy has already changed the treatment landscape of resectable non-small cell lung cancer. The next challenge is more precise: understanding which patients derive the greatest benefit, and whether molecular features can help refine treatment decisions. A new exploratory analysis from the phase 3 CheckMate 77T study presented at the AACR Annual Meeting 2026 moves that conversation forward by examining event-free survival according to tumor genomic alterations and circulating tumor DNA dynamics in patients treated with perioperative nivolumab (Cascone et al., 2026).
The findings suggest that perioperative nivolumab may be particularly important in patients whose tumors harbor alterations traditionally linked to poor prognosis, including KEAP1, STK11, CDKN2A, and SMARCA4, and that ctDNA clearance before surgery, pathologic complete response, and definitive surgery emerge as some of the strongest markers associated with better event-free survival (Cascone et al., 2026).
Why This Analysis Matters
CheckMate 77T has already shown that perioperative nivolumab can provide a statistically significant and clinically meaningful event-free survival benefit compared with placebo in resectable NSCLC. That was the major therapeutic message. What this newer analysis adds is biologic depth. It asks whether the benefit of nivolumab is uniform across all patients or whether genomic context and ctDNA behavior help explain why some patients appear to benefit more than others.
That question is increasingly important in early-stage lung cancer. As immunotherapy moves into potentially curative settings, clinicians need better tools not only to escalate treatment, but also to understand response biology. A biomarker that identifies particularly high-value benefit could help frame risk discussions, refine surveillance strategies, and support more individualized perioperative care.
Study Design and Biomarker Population
In CheckMate 77T, patients with resectable stage IIA to IIIB (N2) NSCLC were randomized to receive either neoadjuvant nivolumab plus chemotherapy followed by surgery and adjuvant nivolumab, or neoadjuvant placebo plus chemotherapy followed by surgery and adjuvant placebo. The biomarker analysis reported at AACR 2026 used a median follow-up of 41.0 months and focused on event-free survival according to genomic marker status, baseline ctDNA detectability, and post-surgical minimal residual disease status (Cascone et al., 2026).
Of the 229 patients randomized to the nivolumab arm and 232 patients randomized to the placebo arm, marker-evaluable samples were available for 98 patients (43%) and 92 patients (40%), respectively. This is an important limitation, but it still provides a meaningful dataset for exploratory analysis.
Genomic Alterations Traditionally Seen as High Risk May Not Limit Benefit From Nivolumab
One of the most notable signals in the study involved tumors with single or co-alterations in KEAP1, STK11, CDKN2A, and/or SMARCA4. These alterations are often associated with poor prognosis and in some metastatic settings have been linked to relative immune resistance. In this analysis, however, the pattern was different.
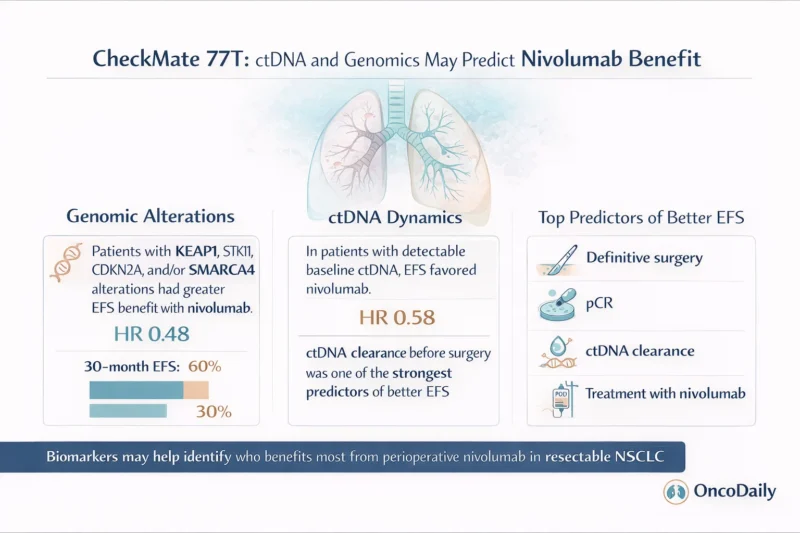
Among marker-evaluable patients, 61% of those in the nivolumab arm and 49% of those in the placebo arm had one or more of these alterations. In that subgroup, event-free survival clearly favored perioperative nivolumab, with a hazard ratio of 0.48 and 30-month event-free survival rates of 60% versus 30% for nivolumab and placebo, respectively (Cascone et al., 2026).
By contrast, in patients without these four alterations, the event-free survival hazard ratio was 0.90, suggesting much less separation between the treatment arms. Just as importantly, within the nivolumab-treated population itself, event-free survival was similar regardless of whether these genomic alterations were present, with a hazard ratio of 0.91. In the placebo arm, however, the presence of these alterations was associated with worse outcomes, with a hazard ratio of 1.77 (Cascone et al., 2026).
This is one of the most interesting aspects of the analysis. It suggests that perioperative nivolumab may reduce, or at least partly offset, the poor prognostic effect of these alterations in early-stage disease. That differs from the simpler assumption that such tumors are uniformly less sensitive to immunotherapy.
ctDNA Detectability and Clearance Add Important Clinical Context
The ctDNA results also deserve attention. Among patients with detectable ctDNA at baseline, event-free survival again favored nivolumab, with a hazard ratio of 0.58 compared with placebo (Cascone et al., 2026). Since baseline ctDNA positivity often reflects biologically active or higher-risk disease, this finding suggests that nivolumab retains meaningful activity even in patients who may begin treatment with less favorable disease biology.
Post-surgical MRD analysis showed a similar directional pattern. In patients who were MRD-negative after surgery, landmark event-free survival from definitive surgery favored nivolumab, with a hazard ratio of 0.75, although the confidence interval crossed 1.0 (Cascone et al., 2026). The numbers were too small for reliable interpretation in patients who had no detectable ctDNA at baseline or who remained MRD-positive after surgery, with 12 or fewer patients per treatment arm in those groups.
These results are therefore not definitive, but they are clinically meaningful. They support the growing view that ctDNA dynamics may become one of the most informative ways to understand treatment response in curative-intent lung cancer.
Machine Learning Highlights the Strongest Predictors of Better Outcome
The investigators also applied a random survival forest machine learning model to identify factors associated with lower risk of worse event-free survival. In that model, the strongest predictors included definitive surgery, pathologic complete response, ctDNA clearance before surgery, and treatment with nivolumab (Cascone et al., 2026).
This part of the analysis is especially valuable because it moves away from the idea that one biomarker alone will define benefit. Instead, it points to a more integrated model in which pathologic response, molecular response, and successful surgical completion all matter. That reflects how perioperative treatment actually works in practice. It is not a single-variable system. It is a treatment pathway in which biology, response, and operability are tightly connected.
A Different Immunotherapy Biology in Early-Stage Disease?
The authors interpret the findings as supporting a context-dependent model of immunotherapy sensitivity. That is an important concept. In advanced NSCLC, genomic alterations such as STK11 and KEAP1 have often been viewed through the lens of immune resistance. But the biology of early-stage disease is different. Tumor burden is lower, the microenvironment is different, and surgery remains part of the treatment plan. In that context, the same genomic alterations may not carry the same treatment implications.
This analysis does not overturn what is known from metastatic disease, but it does suggest that early-stage NSCLC may require a more nuanced framework. In some patients, a biologically aggressive genotype may actually identify a group with more to gain from perioperative nivolumab rather than less.

Limitations Must Still Be Kept in Mind
These are exploratory findings and should be interpreted as such. Only a subset of randomized patients had marker-evaluable samples. Several subgroup analyses involved relatively small numbers. The ctDNA and MRD findings, although interesting, are not yet strong enough to direct routine clinical decision-making on their own. The same is true for the machine learning outputs, which are hypothesis generating rather than immediately practice changing (Cascone et al., 2026).
Still, exploratory does not mean unimportant. In perioperative NSCLC, where biomarker-guided treatment remains underdeveloped, analyses like this are often the first step toward more refined, biologically informed care.
The Clinical Takeaway
The CheckMate 77T biomarker analysis suggests that perioperative nivolumab may be especially relevant in patients with tumors harboring KEAP1, STK11, CDKN2A, and SMARCA4 alterations, and that ctDNA clearance before surgery may emerge as one of the most useful indicators of benefit. It also reinforces the central importance of achieving definitive surgery and pathologic complete response in the overall treatment strategy (Cascone et al., 2026).
The broader OncoDaily message is that perioperative immunotherapy in resectable NSCLC is entering a more sophisticated phase. The field is moving beyond the question of whether nivolumab works and toward the more clinically meaningful question of who benefits most, and why.
You can read full abstract here