This week’s expert highlights reflect a field rapidly advancing through biological precision and therapeutic innovation.
From immune evasion in metastasis and host immune fitness (thymic health) as a pan-cancer biomarker, to next-generation engineering approaches such as masked T-cell engagers and in vivo CAR-T development — the focus is shifting toward smarter, safer immunotherapy design.
Clinical insights reinforced how checkpoint inhibitors continue to redefine standards of care, particularly in HCC, renal cancer, and prostate cancer, while ctDNA and biomarker-driven strategies are increasingly guiding treatment decisions.
Emerging data also pointed to the growing importance of:
- Personalized mRNA vaccines and durable immune memory
- Microenvironment and immune escape mechanisms
- Host factors and lifestyle modulators, including cannabinoids and immune interactions
Meanwhile, advances in nanovaccine platforms and immune system engineering highlight the expanding interface between immunology, biotechnology, and precision oncology.
10 Must-Read Posts in Immuno-Oncology This Week
This Week’s Expert Highlights in Immuno-Oncology
Nicholas DeVito (Assistant Professor of Medicine at the Division of Medical Oncology at Duke University,My lab at Duke Cancer Institute):
“is dedicated to understanding immune evasion during metastasis in colorectal cancer (CRC) and the development of biomarker-selected immunotherapies. We utilize novel immunotherapeutics in advanced orthotopic mouse models, human organoids, and early phase clinical trials to achieve these goals – creating a positive, focused feedback loop centered on radically transforming the first- line standard of care in CRC using our immune sytem.
Learn more at our website to see our recent progress and a vision of how together, we can (and will) overcome this disease with rational implementation of immunotherapies.”
Erman Akkus (Medical Oncology Fellow at Ankara University):
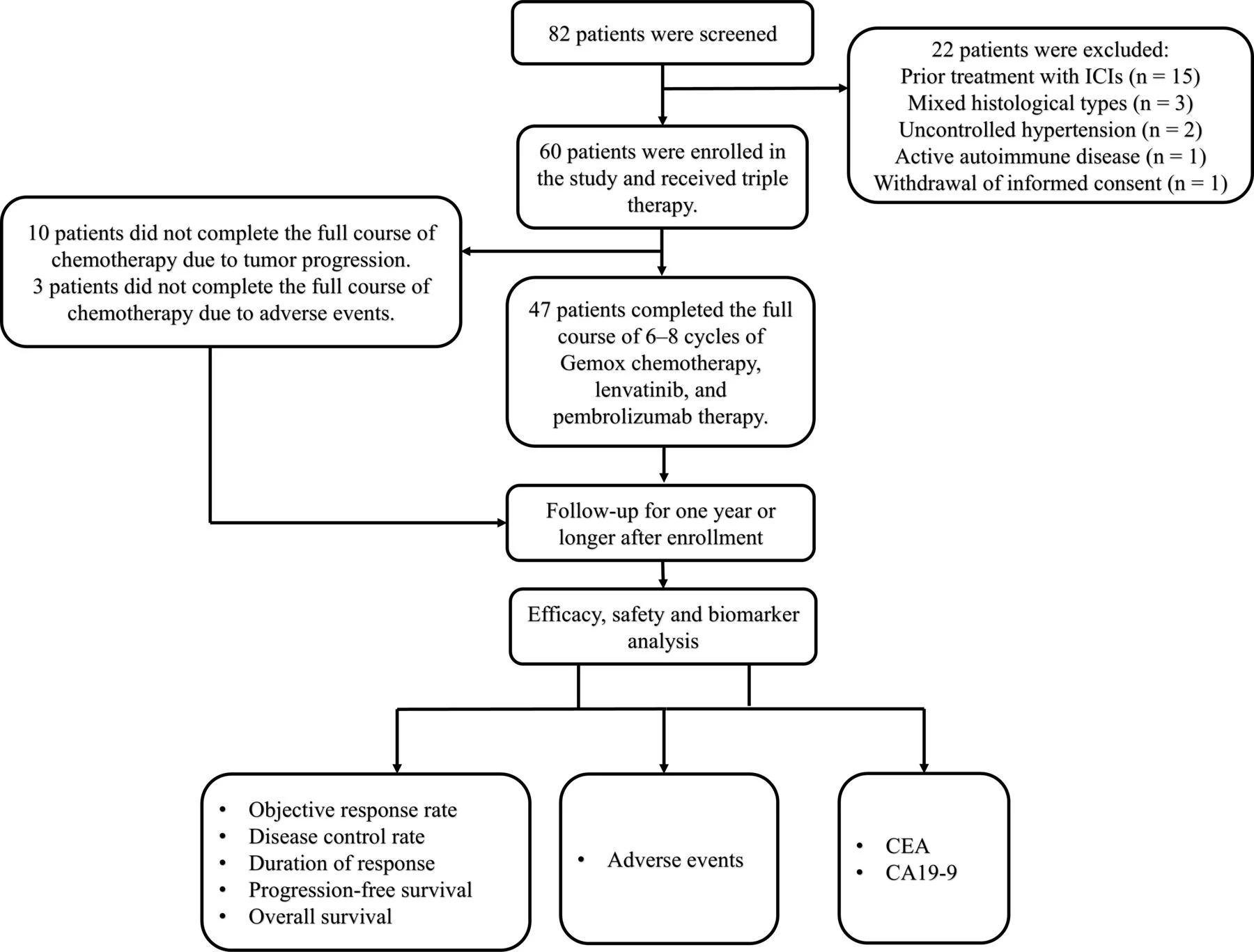
“Efficacy and safety of pembrolizumab, lenvatinib, and reduced-dose gemcitabine/oxaliplatin as initial treatment for advanced biliary tract cancer: a multicenter, single-arm, prospective, phase II study | Journal for ImmunoTherapy of Cancer”
Parag Roy (Medical Oncologist at Tata Main Hospital):
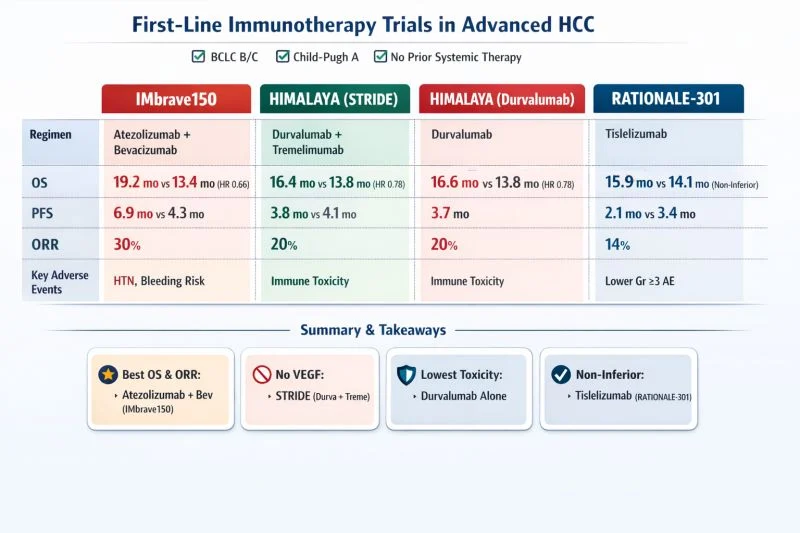
“First-Line Immunotherapy in Advanced HCC: Key Phase III Trials
Management of advanced hepatocellular carcinoma (HCC) has rapidly evolved with the introduction of immune checkpoint inhibitors. Four pivotal phase III trials have shaped current practice.
IMbrave150
Population • Unresectable / metastatic HCC
• BCLC B or C not amenable to locoregional therapy
• Child-Pugh A
• No prior systemic therapy
Arms Atezolizumab + Bevacizumab vs Sorafenib
Key Results
• OS: 19.2 vs 13.4 months (HR 0.66)
• PFS: 6.9 vs 4.3 months
• ORR: ~30%
Key AE Hypertension, proteinuria, bleeding risk (VEGF related)
HIMALAYA Trial (STRIDE regimen)
Population • Unresectable HCC
• BCLC B/C
• Child-Pugh A
Arms STRIDE (Single Tremelimumab + Durvalumab) vs Sorafenib
Key Results
• OS: 16.4 vs 13.8 months (HR 0.78)
• PFS: ~3.8 months
• ORR: ~20%
Key AE Immune-related toxicity (hepatitis, endocrine)
Clinical Advantage No VEGF inhibition → useful in varices / bleeding risk
HIMALAYA Trial (Durvalumab Arm)
Population Same as above
Arms Durvalumab vs Sorafenib
Key Results
• OS: 16.6 vs 13.8 months (Non-inferior)
• PFS: ~3.7 months
• ORR: ~17%
Key AE Lowest toxicity among the regimens.
RATIONALE‑301
Population • Advanced unresectable HCC
• BCLC B/C
• Child-Pugh A
Arms Tislelizumab vs Sorafenib
Key Results
• OS: 15.9 vs 14.1 months (Non-inferior)
• PFS: 2.1 vs 3.4 months
• ORR: ~14%
Key AE Lower grade ≥3 toxicity compared with sorafenib.
Practical Take-Home Messages
- Atezolizumab + Bevacizumab remains the preferred first-line therapy for eligible patients due to the highest OS, PFS, and response rate.
- STRIDE (Durvalumab + Tremelimumab) is a strong alternative when anti-VEGF therapy is contraindicated (e.g., untreated varices, bleeding risk).
- Durvalumab monotherapy offers a well-tolerated option for frail patients.
- Tislelizumab provides another PD-1 monotherapy option with non-inferior survival vs sorafenib.
Clinical takeaway: Patient selection based on Child-Pugh status, BCLC stage, bleeding risk, and performance status is crucial in choosing the optimal first-line immunotherapy strategy for advanced HCC.”
Francesca Jackson-Spence (Clinical Research Fellow in Genitourinary oncology at Barts Cancer Institute):
“Proud to share our latest publication in the Journal of Clinical Oncology:
CALYPSO: Final Results of Savolitinib and Durvalumab Combination in Metastatic Papillary Renal Cancer.
CALYPSO is a Queen Mary University of London-sponsored study that was one of the first trials I worked on as a sub-investigator when I joined Prof Tom Powles team in 2019. I have subsequently had the opportunity to lead the biomarker work.
Our findings show encouraging activity for savolitinib plus durvalumab in MET-driven papillary renal cancer, and suggest that ctDNA may be a useful tool in this disease.
This work has truly been a team effort, and I’m especially grateful to my mentors, collaborators, and colleagues across Queen Mary, Barts, Foundation Medicine, and AstraZeneca for their support.
Read the article here”

Mohamed Haitham Ayad (Pharmaceutical Development Advisor and Managing Director at Pharmaceutical Innovation Advisory (Freelance)):
“New Immunotherapy Drug Shows Promise in Prostate Cancer Trial:
- A new immunotherapy drug, VIR-5500, a “masked T-cell engager,” has shown early promise in a clinical trial for advanced prostate cancer.
- In the trial, 82% of patients receiving the highest doses saw reductions in PSA levels, and nearly half experienced tumor shrinkage.
- This masked approach aims to reduce the severe inflammatory side effects associated with T-cell engagers by activating the drug only within tumors.
- The trial’s finding of only mild inflammatory side effects in most patients suggests the masking technology is effective in enhancing safety.
- While further research and peer review are needed, these initial results offer significant hope for treating difficult-to-treat cancers.”
Janne Ruotsalainen (Senior Research Scientist at CureVac):
“ New paper in Nature
Bernatz, S., Prudente, V., Pai, S. et al. Thymic health and immunotherapy outcomes in patients with cancer. Nature (2026).
Using deep‑learning analysis of routine CT scans, the authors show that thymic health—a proxy for host immune competence—is strongly associated with outcomes to immune checkpoint inhibitors across multiple cancer types.
Rather than focusing solely on tumour‑intrinsic biomarkers (such as PD‑L1 or TMB), this work highlights the prognostic relevance of host immune status, with implications for patient stratification, treatment timing, and precision immuno‑oncology.”

Marco Donia (Professor at University of Copenhagen):
“𝐏𝐞𝐫𝐬𝐨𝐧𝐚𝐥𝐢𝐳𝐞𝐝 𝐦𝐑𝐍𝐀 𝐯𝐚𝐜𝐜𝐢𝐧𝐞 𝐢𝐧 𝐞𝐚𝐫𝐥𝐲 𝐓𝐍𝐁𝐂: 𝐞𝐧𝐜𝐨𝐮𝐫𝐚𝐠𝐢𝐧𝐠 𝐝𝐮𝐫𝐚𝐛𝐥𝐞 𝐢𝐦𝐦𝐮𝐧𝐢𝐭𝐲, 𝐜𝐥𝐚𝐫𝐢𝐟𝐲𝐢𝐧𝐠 𝐫𝐞𝐥𝐚𝐩𝐬𝐞 𝐛𝐢𝐨𝐥𝐨𝐠𝐲
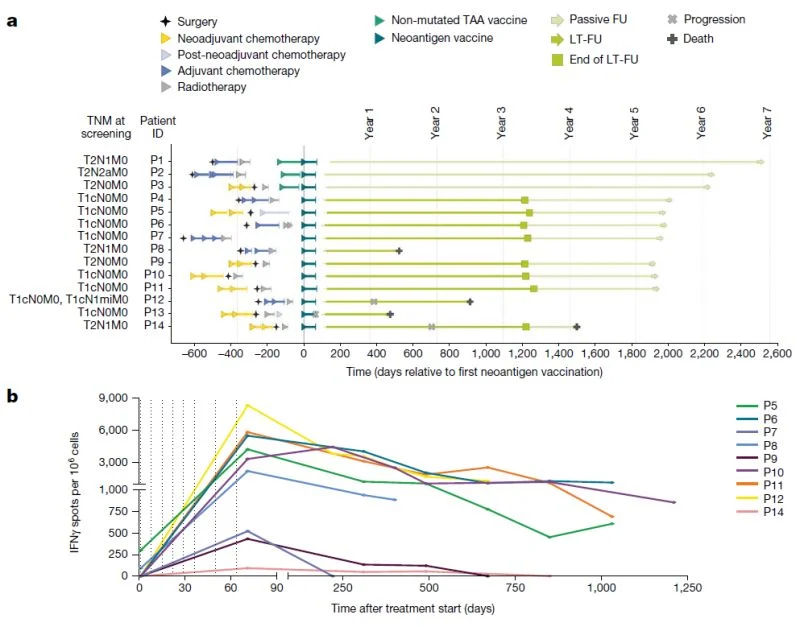
In this first-in-human phase I trial, a personalized neoantigen mRNA vaccine was evaluated in 14 patients with surgically removed early Triple Negative Breast Cancer (TNBC) after standard therapy. The approach was feasible and generally well tolerated, manufacturing took a mean of 69 days, and all 14 evaluable patients developed vaccine-induced T-cell responses, most against multiple neoantigens. Importantly, this was a study of feasibility, safety and mechanism of action, not proof of clinical benefit.
🗝 Key Points:
- 𝐒𝐭𝐫𝐨𝐧𝐠 𝐈𝐦𝐦𝐮𝐧𝐞 𝐒𝐢𝐠𝐧𝐚𝐥:
All 14 evaluable patients developed neoantigen-specific T-cell responses, and in most cases these responses were directed against multiple neoantigens - 𝐃𝐮𝐫𝐚𝐛𝐥𝐞 𝐈𝐦𝐮𝐧𝐞 𝐒𝐢𝐠𝐧𝐚𝐥:
Vaccine-induced T cells remained detectable for years and showed features of both effector function and long-lived memory - 𝐇𝐢𝐠𝐡𝐥𝐲 𝐈𝐧𝐟𝐨𝐫𝐦𝐚𝐭𝐢𝐯𝐞 𝐑𝐞𝐥𝐚𝐩𝐬𝐞𝐬:
Three relapses helped define possible resistance mechanisms: weak immune response in one case, with a complete response to anti-PD-1 after relapse, loss of antigen presentation in another, and genetically distinct recurrent disease in a third.
Take-Home:
- Important proof-of-concept study for individualized mRNA vaccination in early TNBC.
- While this study cannot establish clinical benefit, it clearly shows feasibility and durable immunogenicity.
- Successful immunotherapy is not only about generating T-cell immunity, but also about understanding tumour escape.
Clinical Implications:
- Personalized cancer vaccines may become relevant in high-risk early breast cancer, where preventing relapse is the key goal.
- Combination approaches (e.g. with other immunotherapies) may be needed earlier, especially if resistance mechanisms such as impaired antigen presentation emerge.
- For the cancer-vaccine field, this paper is a very important next step, but it is not conclusive.
Figure from “
Justin Kander (Patient-Reported Outcomes Specialist at E.R.I.C. Cancer Playbook):
“There is still substantial uncertainty about the risk/benefit profile of combining cannabinoids with immunotherapy. However, it’s encouraging that emerging evidence suggests safety, as well as potential anticancer synergy between cannabinoids and the immune system. Although the data is encouraging, special care must be taken if cannabinoids are used alongside immunotherapy because of some research suggesting impaired efficacy.
Human Research
A March 2025 study comparing patients on immune checkpoint inhibitors who did or did not use cannabis found no difference in overall survival, and even improved immune-related progression-free survival among cannabis users.
Preclinically, there’s been some really fascinating stuff. All of these studies are from November 2025 alone.
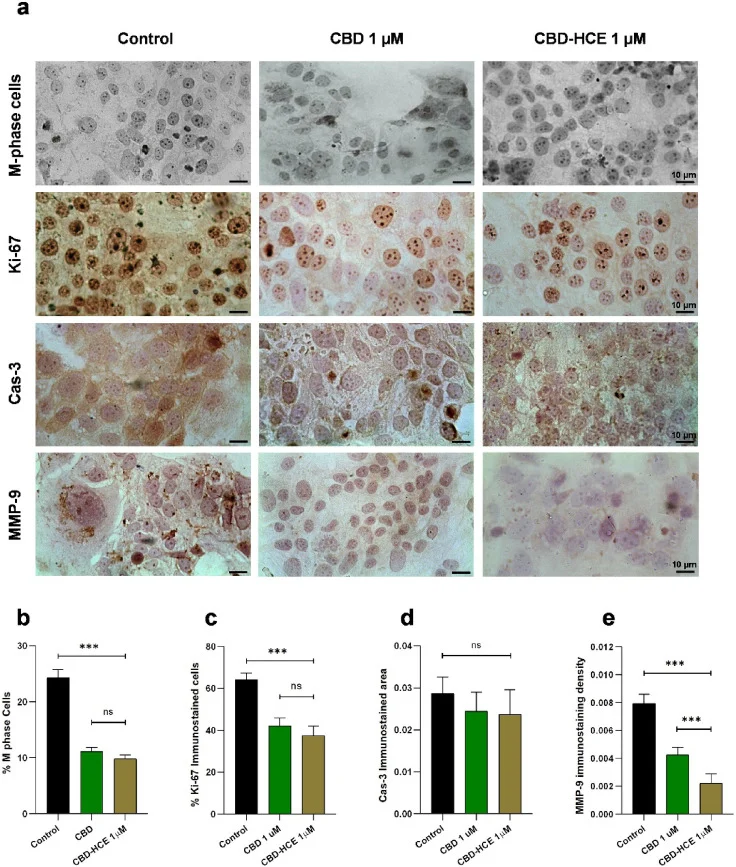
- CBDV reduced immunosuppressive activity of myeloid-derived suppressor cells and was associated with increased CD8+ T-cell activity, “thereby enhancing the immune system’s response against cancer”
- CBD enhanced the ability of natural killer cells to attack triple-negative breast cancer cells
- CBD downregulated a protein called HLA-G on choriocarcinoma cells, helping reverse immune evasion
With further research, the proper doses of specific cannabinoids may be able to work with immunotherapy to better treat resistant cancers.”
Michel Frank Ferrazo (Purchased Materials Technician at ABL Antibióticos do Brasil Ltda):
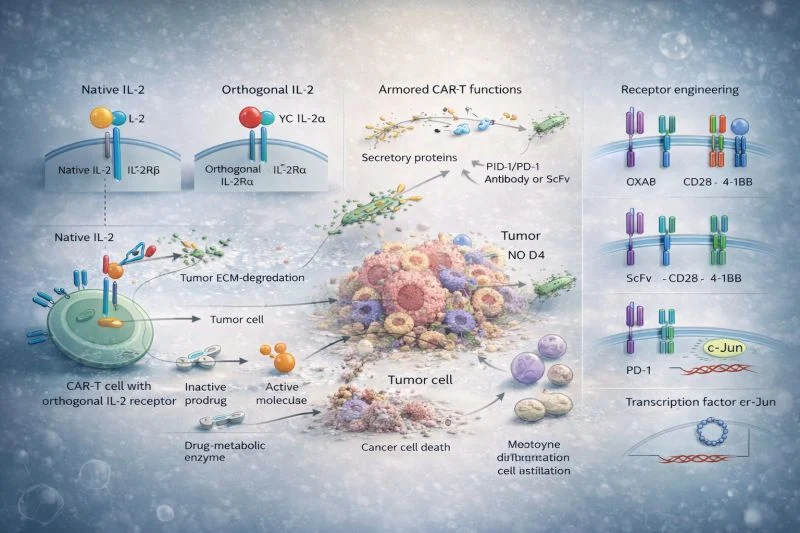
” A Nature News and Views discusses a preclinical strategy from Nyberg et al. to generate CAR T cells directly in vivo in mice, rather than through conventional ex vivo manufacturing.
- The system is described as a two-part editing approach: an adeno-associated virus (AAV) carrying CAR-encoding DNA flanked by TRAC homologous sequences, and an enveloped delivery vehicle (EDV) carrying Cas9 plus a guide RNA directed to TRAC. In the schematic, the EDV targets CD3 on T cells, while the evolved AAV preferentially enters T cells through CD7.
- According to the article, this design enabled site-specific insertion of the CAR sequence into TRAC, with CAR insertion and expression reported as restricted to CD4 and CD8 T cells, and negligible insertion in the other tested cell types.
- In immunodeficient mice reconstituted with human immune cells, the engineered CAR T cells reached about 5–20% of the T-cell pool within 2 weeks, alongside depletion of CD19-expressing B cells.
- In mouse models of B-cell leukaemia, the article states that this strategy cured all mice with leukaemia at the tested doses, whereas the laboratory-modified CAR T-cell comparator and a targeted lentiviral comparator did not.
- The scope was also extended in the article to CAR delivery against myeloma and sarcoma, including evidence of long-lived immune memory against a tumour.
- Important caution: this is still preclinical mouse evidence, and the commentary explicitly notes the need to study unintended editing outside T cells or outside TRAC, test models with cancer-associated immune dysregulation, and clarify how in vivo engineering would function without conventional lymphodepletion.
A useful development to follow as in vivo immune-cell engineering moves toward more stringent translational testing.”
Azam Bakhshandeh (Assistant Librarian at Alma Mater Studiorum Università di Bologna):
“Sharing one publication from my research journey in International Immunopharmacology.
This review focused on nanovaccine design for cancer immunotherapy, particularly antigen–adjuvant complex formation as a promising strategy to improve vaccine delivery and immune activation.
Contributing to this work was an important experience in exploring interdisciplinary biomedical research at the intersection of immunology, nanotechnology, and therapeutic innovation.”




