This week in OncoDaily Immuno-Oncology, expert perspectives highlighted how the field continues to evolve through deeper biological insights and increasingly sophisticated therapeutic strategies. Discussions ranged from emerging logic-gated CAR-T technologies, reflecting the growing integration of computer science into immunotherapy engineering, to new data suggesting that metabolic states such as fasting may influence immunotherapy responsiveness, underscoring the expanding role of immunometabolism in treatment optimization.
Major clinical conversations centered on how neoadjuvant chemo-immunotherapy is reshaping multidisciplinary decision-making, particularly in resectable lung cancer where immune-mediated downstaging and ctDNA dynamics are challenging traditional surgical paradigms. Additional insights explored immune escape mechanisms during early metastasis, including glucocorticoid receptor signaling that enables tumor cells to evade cytotoxic immune responses, as well as evolving evidence supporting microbiome modulation as a determinant of checkpoint inhibitor efficacy.
Precision oncology also remained a central theme, with updated biomarker-driven frameworks guiding treatment selection in gastroesophageal cancers, alongside emerging perioperative strategies combining immunotherapy with chemotherapy to improve surgical conversion in challenging settings such as borderline resectable oral cavity cancer. Meanwhile, advances in cytokine-based immunotherapy—including regulatory developments around IL-15–driven immune activation strategies such as ANKTIVA—highlighted renewed interest in immune stimulation approaches that complement checkpoint blockade.
Collectively, this week’s highlights reflect an immuno-oncology landscape increasingly defined not only by therapeutic innovation, but by mechanistic precision, multidisciplinary integration, and the translation of complex biological insights into clinically actionable cancer treatments.

You Can Also Read 10 Must-Read Posts in Immuno-Oncology This Week
This Week’s Expert Highlights in Immuno-Oncology
Thomas Ichim (President and Chief Scientific Officer of Immorta Bio Inc):
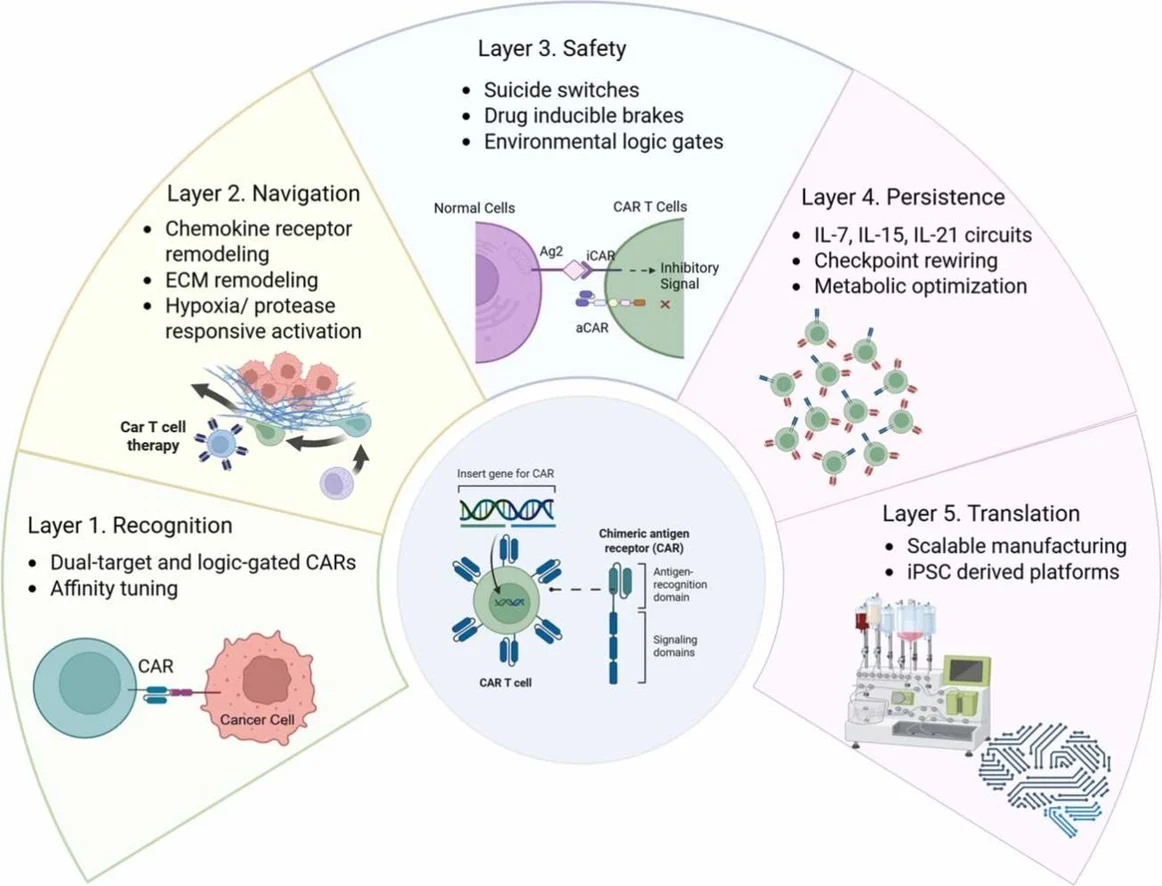
“I love how they are applying computer sciences to cancer immunotherapy
LOGIC GATED CAR-T !!!”
John Tedesco (Thoracic Surgeon at Norton Thoracic Institute):
“Neoadjuvant chemo-immunotherapy is no longer just an oncology story—it’s a surgical one. As trials continue to show deep pathologic responses and improved survival in resectable NSCLC, the question for surgeons is shifting from “Can I resect this?” to “Does surgery still add value here and when?” Traditional anatomic definitions of resectability are being challenged by immune-mediated downstaging, ctDNA clearance, and biologic response that imaging doesn’t always capture.
For surgeons, this means earlier involvement, tighter collaboration with medical oncology, and a bigger role in defining who truly benefits from an operation versus alternative curative pathways. The OR is no longer the finish line: it’s one decision point in a biologically driven continuum of care.
Curious to see how this reshapes MDT discussions and surgical candidacy over the next few years.”
 Yüksel Ürün ( Medical Oncology Professor at Ankara University School of Medicine):
Yüksel Ürün ( Medical Oncology Professor at Ankara University School of Medicine):
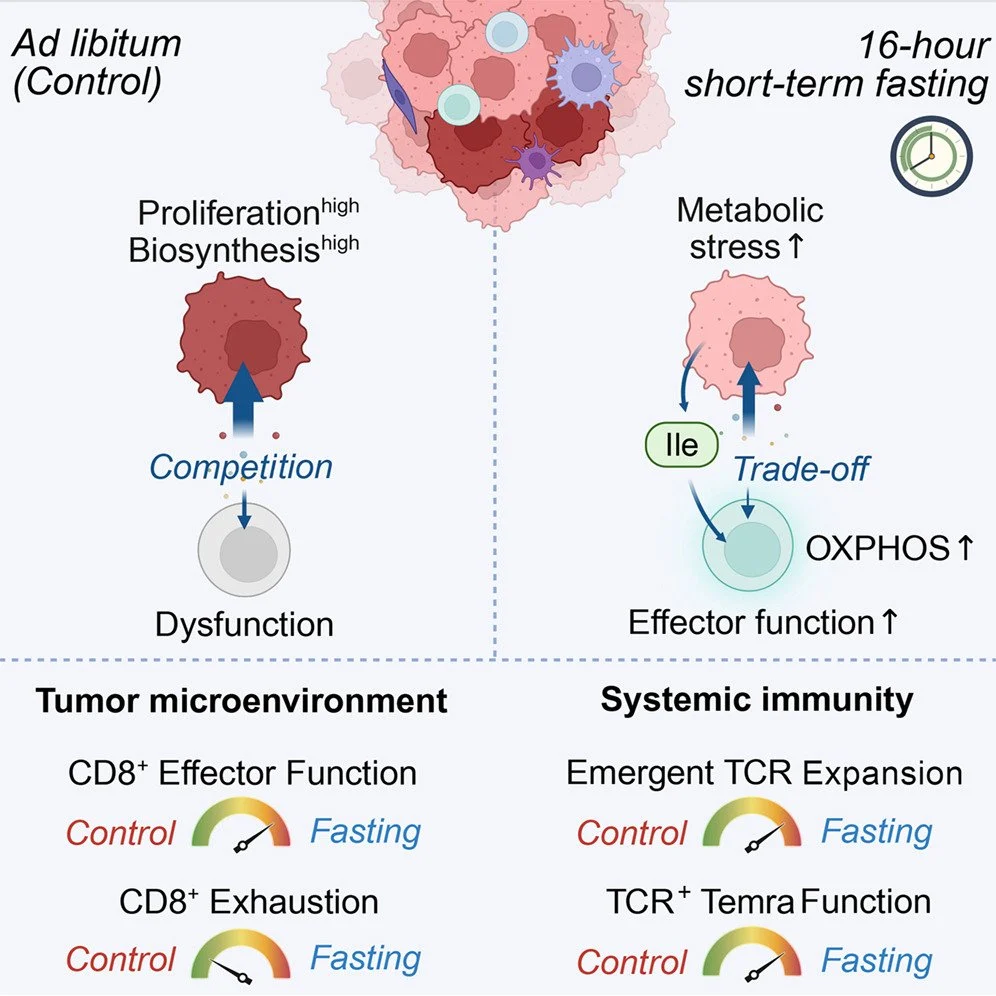
“16-h fasting optimizes cancer immunotherapy in mice and humans!”
Tara Chand Gupta (Medical oncologist at Bhagwan Mahaveer Cancer Hospital and Research Centre):
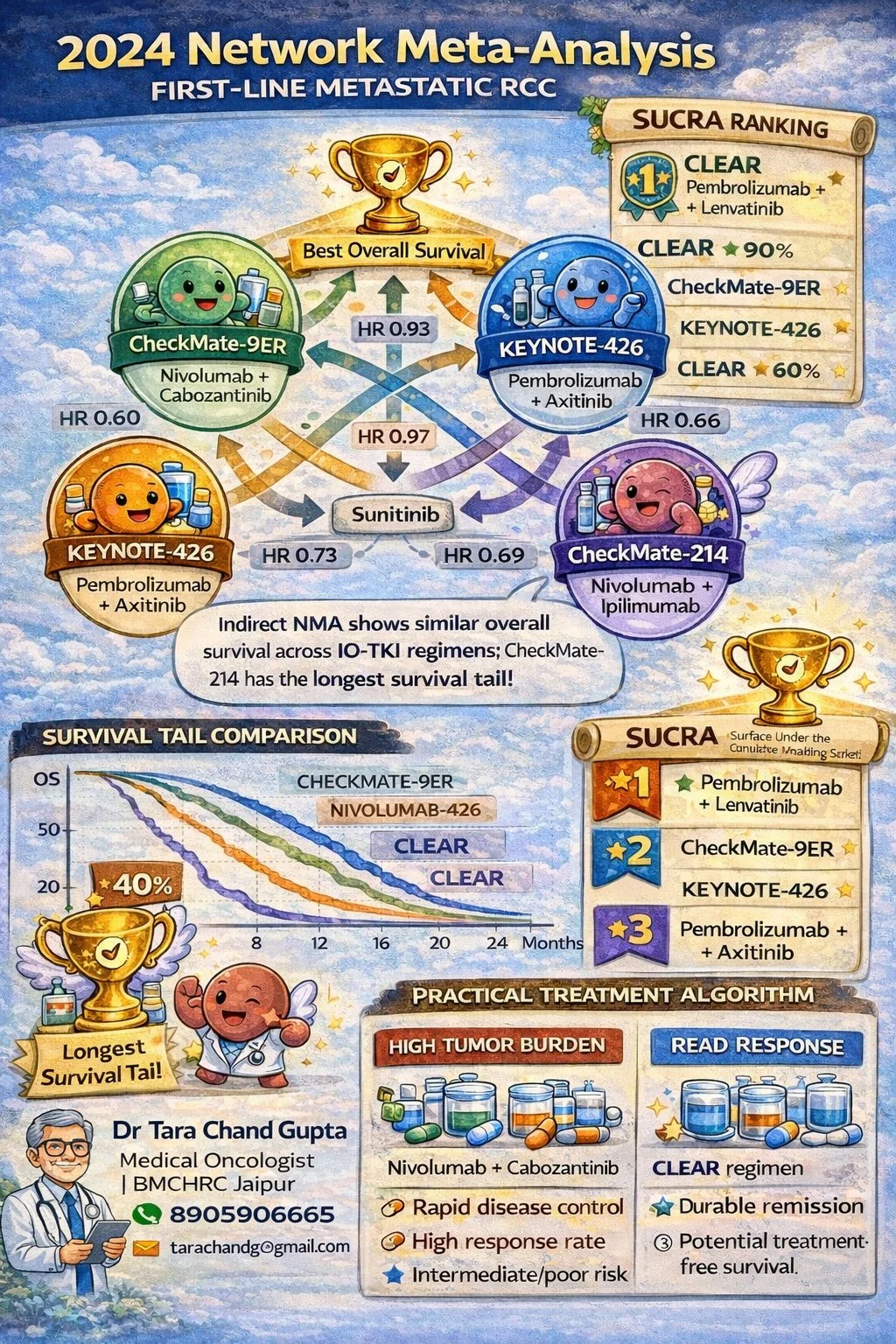
“2024 Network Meta-Analysis in metastatic kidney cancer compared major first-line treatments. Modern therapies like immunotherapy + targeted drugs have improved survival significantly. Choosing the right treatment needs expert evaluation.”
Patrick Hwu (President and CEO of Moffitt Cancer Center):
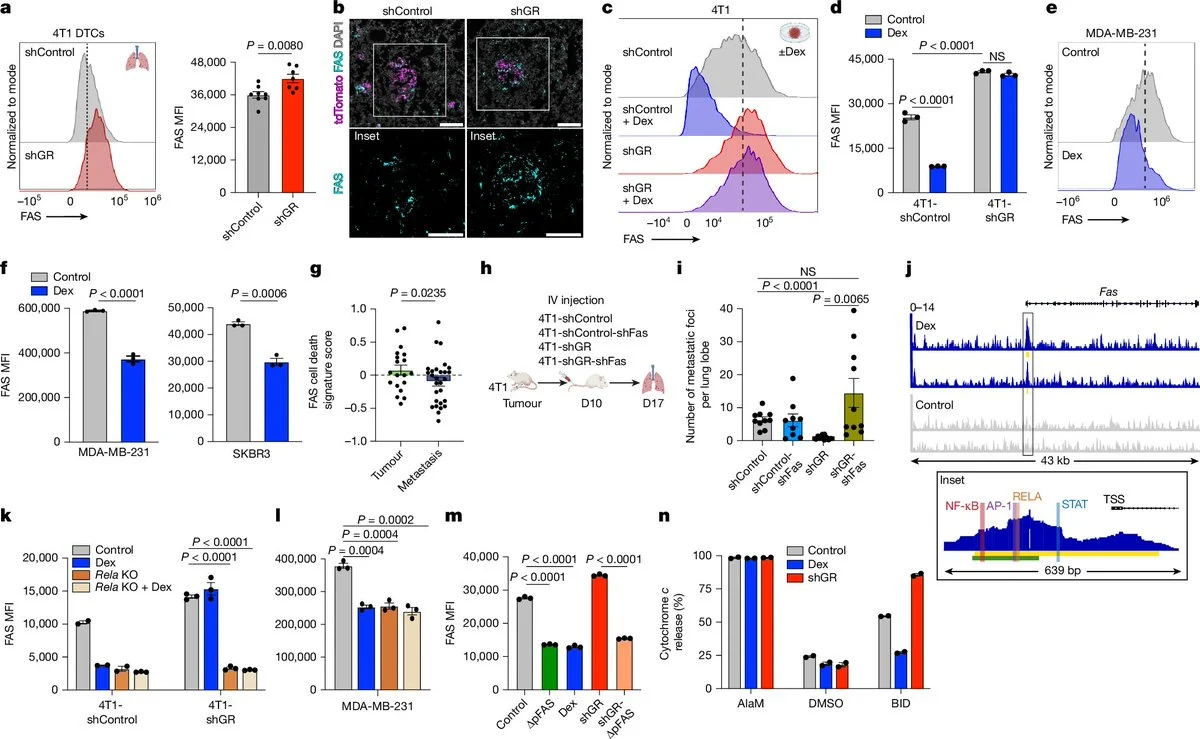
” Why do some cancer cells survive the immune system’s attack when they first spread to new organs?
A new study in Nature shows metastatic cancer cells can exploit stress-hormone signaling to evade immune destruction. Researchers found that activation of the glucocorticoid receptor (GR) helps tumor cells resist killing by T cells and natural killer cells during the earliest stages of metastasis.
GR signaling suppresses the FAS–FASL pathway, one of the key ways immune cells trigger cancer-cell death.
When researchers blocked GR signaling and combined it with immunotherapy in vivo, metastatic tumors were reduced and survival improved.
The takeaway: Immune escape may begin very early in metastasis. Targeting stress-hormone signaling alongside immunotherapy could help eliminate metastatic “seeds” before new tumors take hold.”
Mario Balsa (Medical Oncology Resident at Catalan Institute of Oncology):
” Immunotherapy and targeted therapy for advanced gastroesophageal cancer – ASCO Guideline update!!
Testing early is key to guide first-line decisions:
- PD-L1 ≥1 → ChT + IO recommended
- MSI-H/dMMR → IO ± ChT
- HER2+ → trastuzumab + ChT ± pembrolizumab (PD-L1 ≥1)
- CLDN18.2+ → zolbetuximab + ChT
The guideline is clear: treat the tumor…and of course follow the markers ”
Rishabh Jain (Medical Oncologist at AIIMS):
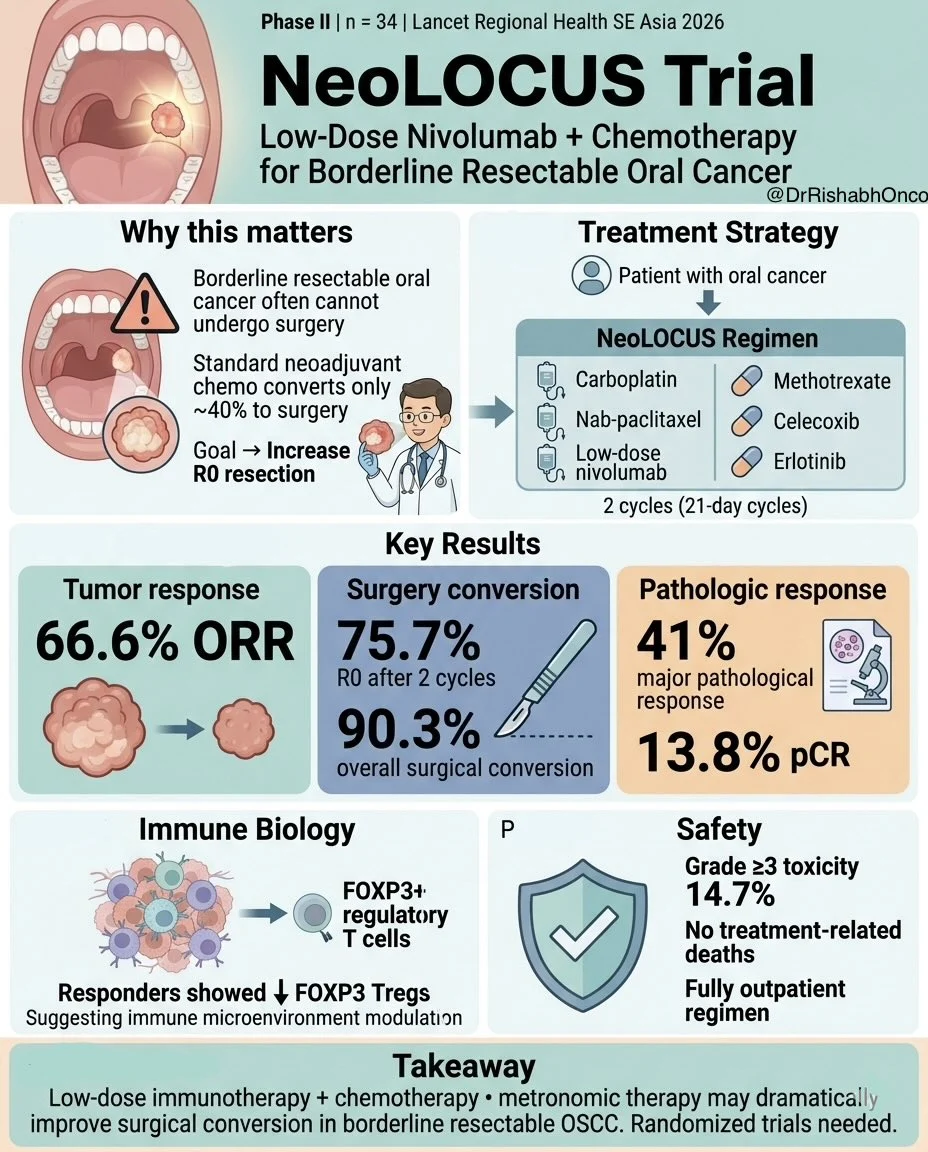
” Borderline resectable oral cavity cancer is a surgical dilemma. Can chemo-immunotherapy increase operability?
A new Phase II trial suggests yes.
NeoLOCUS trial (Lancet Regional Health SE Asia)
Regimen:
- Carboplatin + nab-paclitaxel + low-dose nivolumab
- oral metronomic therapy (methotrexate + celecoxib + erlotinib)
Population
Borderline resectable OSCC
n = 34
Key results
- ORR: 66.6%
- R0 resection after 2 cycles: 75.7%
- Overall surgical conversion: 90.3%
- Major pathological response: 41%
- pCR: 13.8%
Interesting biology
Good responders showed reduction in FOXP3+ Tregs, suggesting immune-microenvironment modulation.
Safety
- Grade ≥3 toxicity: 14.7%
- No treatment-related deaths.
Why this matters Borderline OSCC often fails surgery after standard NACT. This low-dose IO + metronomic + chemo strategy may offer an affordable outpatient approach in LMIC settings.
But this is single-arm Phase II → randomized validation needed. ”
Johan E.T. van Hylckama Vlieg (Co-founder and scientific advisor at Sidekick Bio):
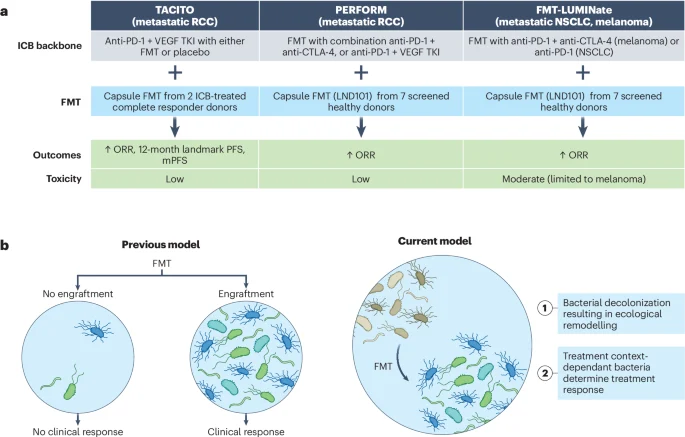
“Recent insights from three fecal microbial transplant (FMT) trials reveal that microbiome interventions can enhance cancer immunotherapy outcomes. The responsiveness to immune checkpoint blockade (ICB) is linked to the overall configuration of the microbiome rather than solely relying on individual beneficial taxa, which has been the prevailing hypothesis. Notably, the decolonization of adverse taxa seems to be a key factor contributing to poor responses. ”
Asger Meldgaard Frank (PhD Student in Immunotechnology at Lund University):
“Here we go!
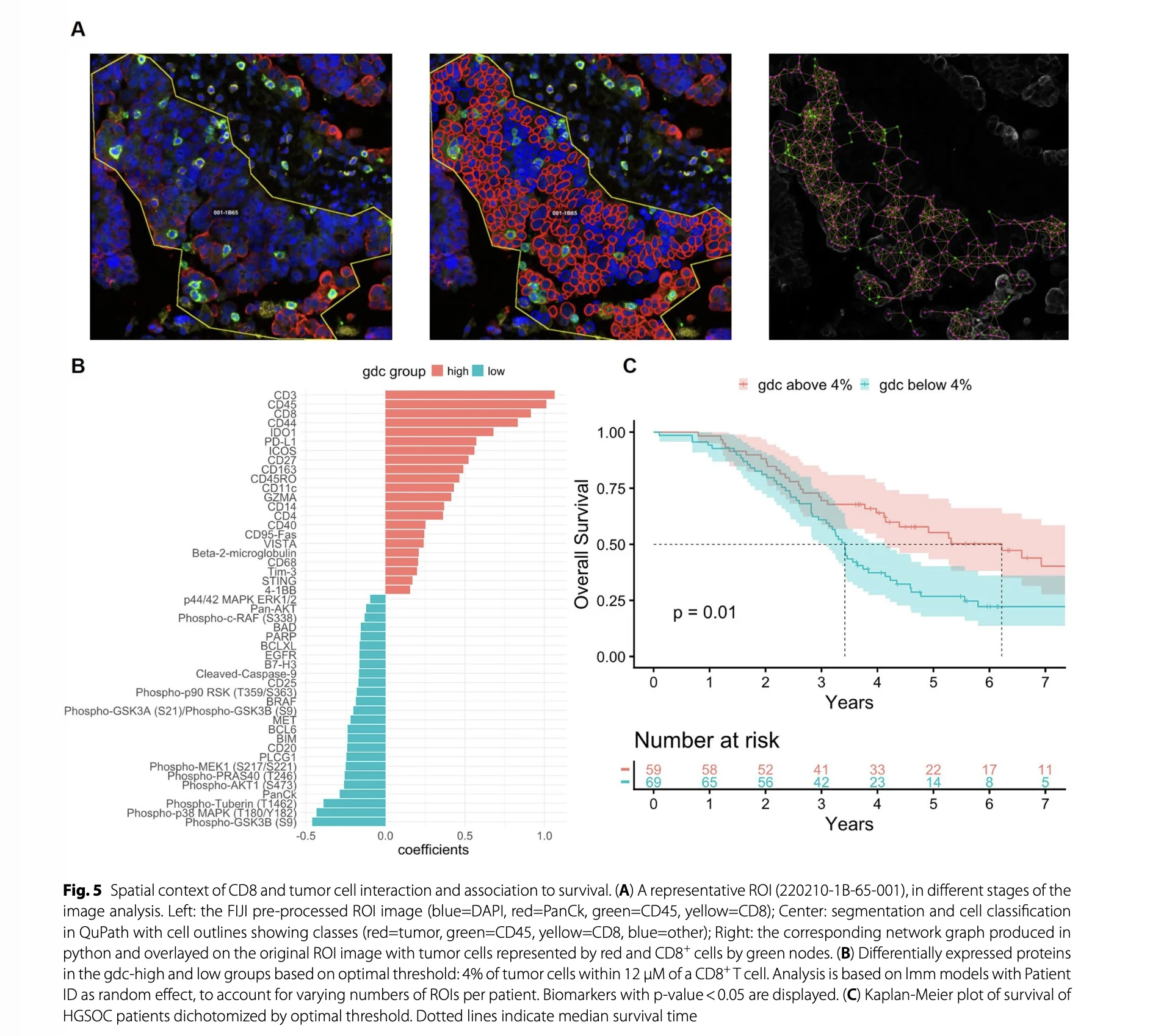
Our paper “Distinct immune microenvironments in ovarian cancer subtypes indicate potential for immunotherapies” is now out wherever you get your articles
Ovarian cancer is not just one disease. Different histotypes behave very differently, and we think that should be reflected in how we treat them. In this work, we used spatial proteomics (GeoMx) to map the tumor immune microenvironment across ovarian carcinoma subtypes, combining tumor-immune spatial scoring with intra-tumoral expression of key markers.
The door to a more tailored immunotherapy strategies based on different ovarian cancer histotypes, tumor-immune spatial scoring and intra-tumoral expression of key markers is now open.
This work only exists because of brilliant collaborators and, most importantly, patients who donated parts of themselves for research. That generosity never gets taken for granted.”
Jack Shuang Hou (Executive, Diagnostic Technology Firm – Regulatory & Business Development):
” ImmunityBio, IncResubmits sBLA to FDA for ANKTIVA + BCG in BCG-Unresponsive Bladder Cancer with Papillary Disease
A new regulatory step could expand treatment options for patients with non-muscle invasive bladder cancer (NMIBC). ImmunityBio, Inc. announced that the U.S. FDA has acknowledged receipt of its resubmitted supplemental Biologics License Application (sBLA) for ANKTIVA® (nogapendekin alfa inbakicept-pmln) plus BCG in BCG-unresponsive NMIBC with papillary tumors.
The resubmission follows multiple meetings with the FDA, during which the agency requested additional data. After reviewing the updated information in February 2026, the company provided updated efficacy and long-term follow-up data, leading to the formal resubmission of the sBLA. Notably, no new clinical trials were requested by the FDA.
Clinical Evidence Supporting the Filing: The application is supported by results from the Phase 2/3 QUILT 3.032 study (Cohort B) involving 80 patients with high-grade papillary-only NMIBC.
Key outcomes:
- 12-month disease-free survival: 58.2%
- 36-month disease-specific survival: 96%
- 12-month progression-free survival: 94.9%
- 36-month progression-free survival: 83.1%
Importantly, bladder preservation remained high, with:
- 92.2% cystectomy-free survival at 12 months
- 81.8% at 36 months
These results suggest a durable bladder-sparing treatment approach for patients who otherwise may require radical cystectomy.
About ANKTIVA
ANKTIVA is a first-in-class IL-15 superagonist fusion complex designed to activate:
- Natural killer (NK) cells
- CD8+ cytotoxic T cells
- Memory T-cell responses
The therapy was FDA-approved in 2024 for BCG-unresponsive NMIBC with carcinoma in situ (CIS), with or without papillary tumors.
Leadership Perspective
SOON SHIONG PATRICK, Founder, Executive Chairman, and Global Chief Scientific and Medical Officer of ImmunityBio, Inc., emphasized that IL-15 has long been recognized as a powerful immunotherapy pathway, with ANKTIVA designed to restore immune activity against tumor cells.
Why This Matters: Approximately 60,000 patients are diagnosed with NMIBC annually in the United States, with ~70% presenting with papillary disease.
For patients who fail BCG therapy, treatment options remain limited and often lead to radical bladder removal.
My Takeaways
- Cytokine-based immunotherapy is making a comeback.
IL-15–driven immune activation represents a different strategy from checkpoint inhibitors. - Bladder-preserving therapies are becoming a major clinical priority.
Avoiding cystectomy dramatically improves patient quality of life. - Long-term follow-up data are increasingly important for regulatory decisions.”