Early-stage non-small cell lung cancer (NSCLC) is increasingly being diagnosed at a time when cure is still possible, driven in part by broader use of low-dose CT screening and greater awareness of early pulmonary lesions. Yet a meaningful proportion of these patients are not candidates for surgery because of severe chronic obstructive pulmonary disease, cardiovascular comorbidity, frailty, or poor pulmonary reserve. For this population, the central question is no longer whether curative-intent treatment should be offered, but which local therapy can provide the best balance of tumor control, safety, and preservation of lung function.
A new retrospective series published in the Journal of Vascular and Interventional Radiology adds important data to this discussion. Rehman and colleagues reported outcomes from what they describe as the largest single-institution cohort of medically inoperable Stage IA NSCLC treated with percutaneous cryoablation, evaluating 176 consecutive patients treated between July 2020 and July 2025 (Rehman et al., 2026). Their findings suggest that CT-guided cryoablation can achieve high local control and excellent short- to mid-term survival, particularly in tumors smaller than 2 cm, while maintaining an acceptable procedural safety profile.
Why This Question Matters
Surgical lobectomy remains the standard treatment for operable Stage I NSCLC, and stereotactic body radiation therapy (SBRT) is widely used for medically inoperable patients (NCCN, 2025; Schneider et al., 2018). However, the rise in screen-detected lung cancers has brought more medically fragile patients into the curative-intent setting. Many of these patients have advanced smoking-related lung disease or other comorbid conditions that make surgery high risk or technically inappropriate (Gould et al., 2017; Palma et al., 2010).
Thermal ablation, including cryoablation, has therefore emerged as a potentially important option. According to the current NCCN guidelines cited by the authors, ablative approaches may be considered in selected medically inoperable Stage I NSCLC, oligometastatic disease, or unresectable local recurrence (NCCN, 2025). Still, widespread adoption has been limited by the relative scarcity of large contemporary datasets, especially compared with the evidence base supporting surgery and SBRT.
That is where this new study becomes relevant.
Study Design and Patient Population
This was a single-center retrospective analysis of 176 consecutive patients with biopsy-proven Stage IA NSCLC treated with percutaneous cryoablation. Eligible patients had tumors 3 cm or smaller, were medically inoperable or declined surgery, and had an ECOG performance status below 2. Notably, all patients had severe airflow limitation, defined as Global Initiative for Chronic Obstructive Lung Disease stage 3 or higher, underscoring how medically fragile this population was (Rehman et al., 2026).
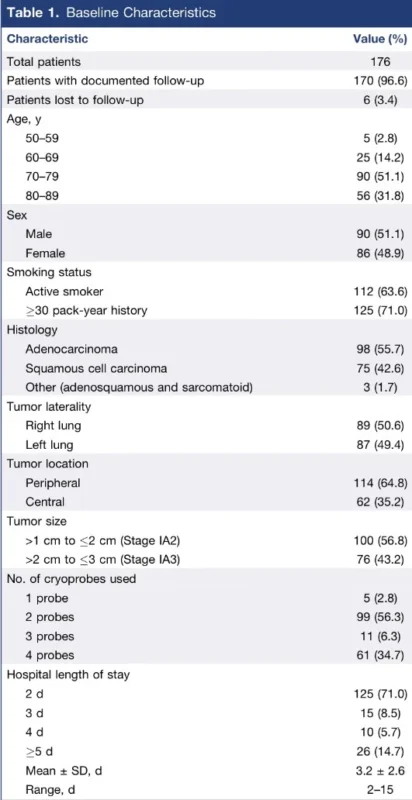
Of the 176 treated patients, 170 had complete follow-up data available for survival analysis. The cohort was elderly and high risk. More than 82% were 70 years or older, 71% had at least a 30 pack-year smoking history, and the great majority had significant pulmonary impairment. Histologically, adenocarcinoma accounted for 55.7% of tumors and squamous cell carcinoma for 42.6%. Most lesions were peripheral, though more than one third were centrally located. Slightly more than half were Stage IA2 tumors, measuring between 1.1 and 2.0 cm, while 43.2% were Stage IA3 tumors, measuring between 2.1 and 3.0 cm (Rehman et al., 2026).
This is important because it reflects a real-world group of patients who often fall outside the comfort zone of surgical management but still require definitive treatment.
How The Cryoablation Was Performed
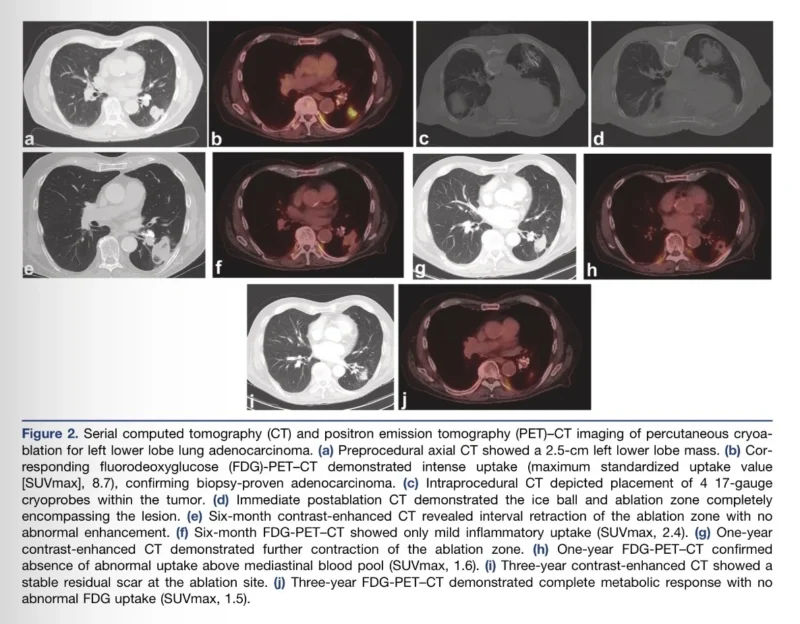
All procedures were done under general anesthesia and CT guidance by one thoracic interventional radiologist. The team used a standardized triple freeze-thaw protocol: a 3-minute freeze followed by passive thaw, then a 7-minute freeze and passive thaw, and finally a 10-minute freeze followed by active thaw. The number of cryoprobes varied according to lesion size and geometry, with one to five probes used, though most patients were treated with two or four probes. The mean probe density was 1.61 probes per centimeter of tumor for the 17-gauge systems used in most cases (Rehman et al., 2026).
The goal was complete tumor coverage with an approximately 1 cm margin, monitored in real time through imaging of the ice ball. Follow-up was rigorous, including contrast-enhanced CT at one month, repeated CT imaging at regular intervals, and PET every six months for the first three years (Rehman et al., 2026).
This procedural standardization is one of the strengths of the study, because cryoablation outcomes can be heavily influenced by technique, probe positioning, and freeze-thaw design.
Local Control: Strong Results, Especially in Smaller Tumors
The primary endpoint was local progression-free survival (LPFS), and the results were encouraging. Among the 170 patients with complete follow-up, the 1-year LPFS rate was 91.8%, and the 3-year LPFS rate was 89.4%. The overall local progression rate across the study period was 10.6% (Rehman et al., 2026).
These are clinically meaningful numbers in a medically inoperable population. They suggest that cryoablation is not merely palliative local treatment, but a potentially durable local therapy for carefully selected Stage IA NSCLC.
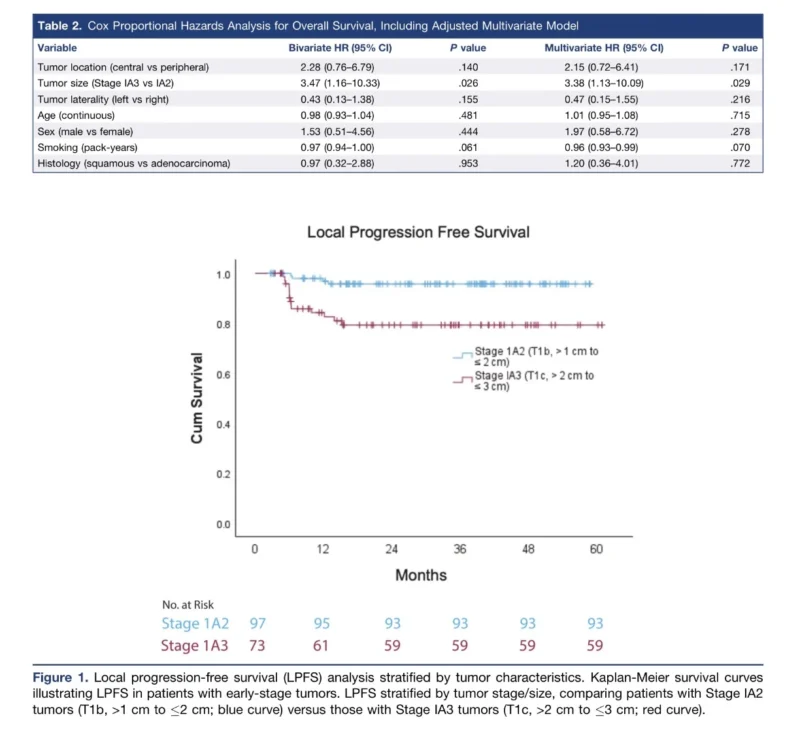
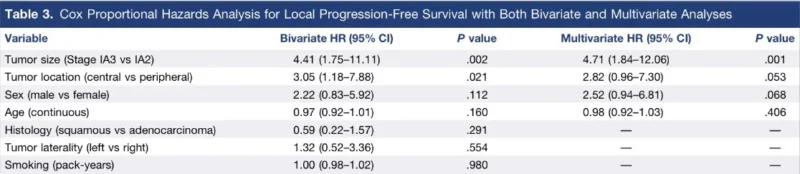
Tumor size was the most important determinant of outcome. Stage IA3 tumors, measuring more than 2 cm up to 3 cm, had substantially worse local control than IA2 tumors. On multivariable analysis, tumor size remained the strongest independent predictor of local progression, with Stage IA3 lesions carrying an adjusted hazard ratio of 4.71 compared with IA2 tumors (P = .001) (Rehman et al., 2026).
Kaplan–Meier estimates reflected this clearly. Stage IA2 tumors had a 3-year LPFS of 95.9%, while Stage IA3 tumors had a 3-year LPFS of 80.8%.
Central tumor location also appeared relevant. In raw terms, central lesions had higher recurrence than peripheral tumors, 19.4% versus 5.1%, although after adjustment this became a strong trend rather than a formally significant predictor, with an adjusted hazard ratio of 2.82 and a P value of .053. Even if it narrowly missed conventional statistical significance, the signal is clinically plausible and consistent with the technical challenges of ablating central tumors.
Overall Survival: Better Than Many Would Expect
Overall survival outcomes were also notable. Among the 170 patients with follow-up, only 13 deaths were recorded during the study period. Overall survival was 100% at one year and 94.7% at three years. Again, tumor size mattered. While one-year survival was 100% for both IA2 and IA3 tumors, three-year survival was 100% in IA2 disease compared with 92.1% in IA3 tumors. On multivariable analysis, Stage IA3 tumor size was the only significant independent predictor of worse overall survival, with an adjusted hazard ratio of 3.38 (P = .029) (Rehman et al., 2026).
These survival outcomes compare favorably with prior ablation studies and appear particularly strong for a cohort defined by medical inoperability. As the authors note, this group included many older patients with heavy smoking histories and severe pulmonary dysfunction, all factors that would generally predict worse outcomes (Rehman et al., 2026).
How Does Cryoablation Compare With Prior Ablation Literature?
One of the most useful aspects of the paper is that the authors place their findings in context. They compare their results with prior cryoablation and radiofrequency ablation series.
Moore and colleagues previously reported five-year survival after cryoablation in medically inoperable Stage I NSCLC, with a smaller cohort and a 5-year recurrence-free survival of 87.9% (Moore et al., 2015). Rehman and colleagues note that their own 3-year LPFS of 89.4% compares favorably, especially given the larger and more contemporary cohort (Rehman et al., 2026).
The study also compares favorably with historical radiofrequency ablation data. The American College of Surgeons Oncology Group Z4033 trial reported two-year local recurrence-free rates that were substantially worse than those seen here, and the French multicenter RFA study by Palussière et al. also produced lower local control at one and three years than what was observed in this cryoablation series (Dupuy et al., 2015; Palussière et al., 2018; Rehman et al., 2026).
The authors reasonably suggest that technical factors may contribute to this difference, including probe density, freeze-thaw design, and real-time ice ball visualization. They also cite a recent meta-analysis indicating that triple freeze-thaw protocols may improve local control compared with double-freeze strategies (Sarshoghi et al., 2026).
How Does It Compare With Surgery and SBRT?
Direct comparisons remain difficult because this was not a randomized trial, and the patient populations are inherently different. Still, the discussion provides an informative benchmark.
Compared with sublobar surgical resection in older high-risk populations, the three-year overall survival seen with cryoablation appears strong, especially considering how medically compromised these patients were (Liu et al., 2014; Rehman et al., 2026). Compared with SBRT, the reported local control rates are also in the same general range. The authors note that one- and three-year LPFS rates of 91.8% and 89.4% align with common SBRT benchmarks in medically inoperable Stage I NSCLC, while three-year OS appears numerically higher than in landmark SBRT trials such as RTOG 0236 and JCOG0403, though cross-trial comparison should be cautious (Timmerman et al., 2010; Nagata et al., 2015; Rehman et al., 2026).
The real distinction may lie in toxicity and retreatment flexibility. SBRT carries risks such as delayed radiation pneumonitis and fibrosis, whereas cryoablation complications are usually immediate and procedure-related. In addition, cryoablation can be repeated more easily in selected cases, which may matter in patients with local recurrence.
Salvage and Repeat Cryoablation
This is another practical strength of the study. Among patients who developed local progression, salvage treatment included repeat cryoablation, radiation, or chemoradiation. Repeat cryoablation was performed in 12 patients and appeared feasible, with a secondary 12-month LPFS of 83.34% after the repeat procedure. Importantly, overall survival did not differ significantly between patients treated once and those undergoing repeat ablation, suggesting that salvage cryoablation may preserve long-term outcomes when recurrence is detected early (Rehman et al., 2026).
That flexibility may be one of the underappreciated advantages of percutaneous ablation.
Safety: Acceptable, But Not Trivial
No discussion of lung ablation is complete without addressing safety. The most common complication was pneumothorax requiring chest tube placement, which occurred in 17.6% of patients. Five patients required a second chest tube. Central tumors significantly increased the likelihood of needing chest tube drainage, with an odds ratio of 4.95 (P = .007) (Rehman et al., 2026).
Hemothorax occurred in 2.3%, and pleural effusions in 4.5%, while pneumonia was rare. There were no air emboli, no bronchopleural fistulas, no grade 4 or 5 events, and no procedure-related deaths. No deaths occurred within two months of the procedure (Rehman et al., 2026).
For a population with severe lung disease and poor surgical fitness, that safety profile is reassuring. It is not complication-free, but it appears acceptable and predictable. Most importantly, the complications were manageable, and the absence of catastrophic events supports the role of cryoablation in experienced hands.
What Are The Main Limitations?
The authors are careful not to overstate their findings. This was a retrospective, single-operator, single-institution study, and therefore subject to selection bias and unmeasured confounding. There was no randomized comparison with SBRT, surgery, or other ablation techniques. PET-based recurrence assessment may also make comparisons with older CT-based series imperfect. And as with any local therapy study outside of surgery, systematic nodal staging remains less robust than in resection-based pathways (Rehman et al., 2026).
These are real limitations. The study does not prove that cryoablation is superior to SBRT or surgery. It does, however, make a strong case that cryoablation deserves serious consideration as a primary curative-intent option in carefully selected medically inoperable Stage IA NSCLC.
What Is The Clinical Takeaway?
The most important message is not that cryoablation should replace every existing standard. It is that for a specific group of patients, particularly those with medically inoperable Stage IA tumors smaller than 2 cm, CT-guided cryoablation can provide very strong local control and excellent survival, with a manageable procedural risk profile.
The data also suggest a more nuanced view of patient selection. Smaller tumors did best. Central tumors remain more challenging. Technical standardization appears to matter. And repeat ablation may offer an effective salvage strategy in selected recurrences.
In a lung cancer landscape increasingly shaped by screening, earlier detection, and multimodality personalization, these are important findings.
Conclusion
Rehman and colleagues provide the largest reported single-institution experience of percutaneous cryoablation in medically inoperable Stage IA NSCLC, and the results are impressive. The study found 1-year and 3-year local progression-free survival rates of 91.8% and 89.4%, with overall survival of 100% at one year and 94.7% at three years. Tumor size emerged as the key prognostic factor, with lesions above 2 cm carrying a substantially higher risk of local progression and death (Rehman et al., 2026).
The procedure was not without complications, especially pneumothorax, but it showed no grade 4 or 5 events and no procedure-related mortality. For medically inoperable patients with Stage IA NSCLC, especially those with tumors under 2 cm, these results strongly support cryoablation as a lung-preserving, potentially curative local treatment option.
More prospective, multicenter comparative studies are clearly needed. But after this series, the role of cryoablation in early-stage inoperable NSCLC looks harder to dismiss and more important to define.
You can read full article here