The phase 3 CAMPASS trial adds important evidence to the growing body of research exploring whether immune checkpoint inhibition combined with anti-angiogenic therapy can outperform single-agent immunotherapy in the first-line treatment of advanced non-small-cell lung cancer (NSCLC).
In this blinded, randomized, controlled phase 3 study conducted across 79 centers in China, investigators compared benmelstobart plus anlotinib with pembrolizumab alone in patients with previously untreated, driver gene–negative, PD-L1–positive advanced NSCLC. The trial met its primary endpoint, showing a statistically significant improvement in progression-free survival with the combination strategy.
These results position benmelstobart plus anlotinib as a potentially meaningful first-line option, particularly in a population for whom chemotherapy-free immunotherapy-based approaches remain clinically attractive but often suboptimal in durability.
Background
Pembrolizumab monotherapy is a well-established standard for patients with PD-L1–positive advanced NSCLC, especially for those with PD-L1 tumor proportion score (TPS) ≥50%, and is also used in selected patients with PD-L1 TPS 1–49% who are not candidates for chemotherapy. However, despite its regulatory and clinical success, the efficacy ceiling of single-agent immunotherapy remains clear. In KEYNOTE-042, median progression-free survival and overall survival with pembrolizumab monotherapy remained limited, underscoring the need for strategies capable of amplifying the benefit of immune checkpoint blockade.
One such strategy is dual targeting of immune escape and angiogenesis. Anti-angiogenic therapy may enhance immunotherapy activity through several biologically plausible mechanisms, including vascular normalization, reduction of tumor hypoxia, improved immune-cell trafficking, and reversal of VEGF-driven immunosuppressionwithin the tumor microenvironment. These effects may increase the ability of checkpoint inhibitors to generate durable tumor control.
This rationale has already been tested in other phase 3 trials. LEAP-007, evaluating pembrolizumab plus lenvatinib, showed improved progression-free survival over pembrolizumab alone but failed to establish an overall survival advantage and raised concerns regarding toxicity. HARMONi-2, testing ivonescimab against pembrolizumab, also demonstrated progression-free survival benefit and reinforced interest in this class of combinations. CAMPASS now contributes a third major phase 3 dataset to this evolving therapeutic space.
Study Design
CAMPASS enrolled patients aged 18–75 years with stage IIIB–IV squamous or non-squamous NSCLC, PD-L1 TPS ≥1%, ECOG performance status 0–1, and no prior systemic therapy for advanced disease. Patients with known EGFR, ALK, or ROS1 alterations were excluded, except that comprehensive molecular testing was not mandatory for all squamous cases.
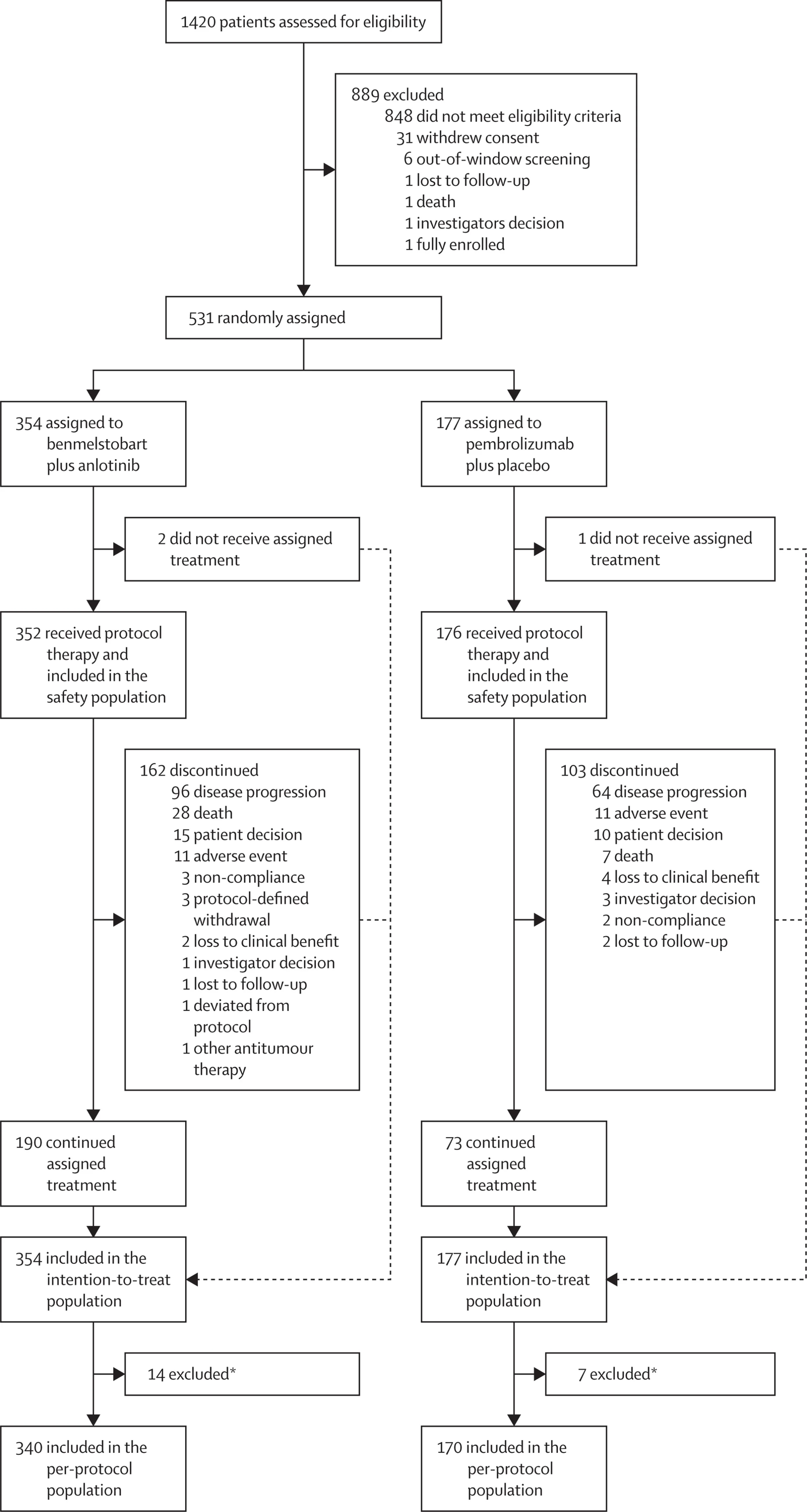
A total of 531 patients were randomly assigned in a 2:1 ratio to receive either:
- Benmelstobart 1200 mg IV every 3 weeks plus anlotinib 12 mg orally on days 1–14 of a 21-day cycle, or
- Pembrolizumab 200 mg IV every 3 weeks plus placebo
Randomization was stratified by:
- Histology
- PD-L1 TPS (1–49% vs ≥50%)
- Presence of brain metastases
The primary endpoint was progression-free survival (PFS) by blinded independent review committee (IRC) per RECIST 1.1 in the intention-to-treat population. Overall survival was the key secondary endpoint, but at the time of this analysis remained immature.
Patient Population
The baseline characteristics reflect a population typical of advanced NSCLC in East Asia, though with some notable features:
- Median age: 65 years
- Male: 85%
- PD-L1 TPS ≥50%: 45%
- Squamous histology: 60%
- Brain metastases: 7%
- Stage IV disease: ~75%
A relatively high proportion of patients had squamous histology, and the study population was entirely Chinese, which is important when interpreting generalizability across broader global populations.
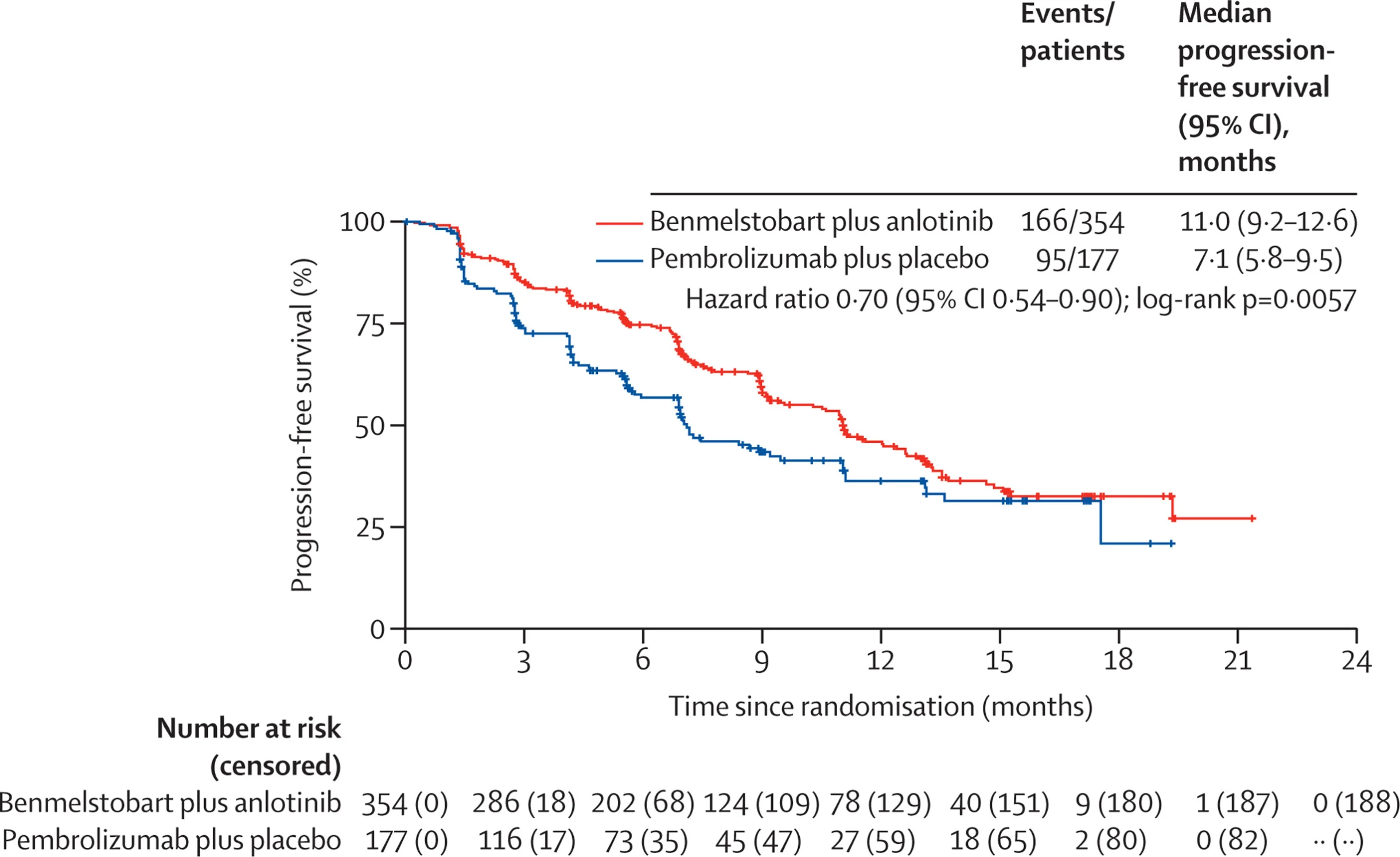
Primary Endpoint: Progression-Free Survival
CAMPASS met its prespecified primary endpoint.
After a median follow-up of:
- 11.4 months in the benmelstobart + anlotinib group
- 10.6 months in the pembrolizumab group
the median PFS by IRC was: - 11.0 months with benmelstobart + anlotinib
- 7.1 months with pembrolizumab + placebo
This translated to:
- HR 0.70 (95% CI 0.54–0.90)
- p = 0.0057
Thus, the combination reduced the risk of progression or death by approximately 30% relative to pembrolizumab monotherapy.
The PFS curves separated early and remained apart for approximately one year, suggesting not only an initial disease-control advantage but also sustained benefit over time.
Landmark PFS rates further supported the benefit:
- 6-month PFS: 75% vs 57%
- 12-month PFS: 46% vs 36%
Although the observed hazard ratio was slightly less favorable than the value assumed in the statistical powering model, the improvement remained both statistically robust and clinically meaningful.
Response Outcomes
The combination of benmelstobart plus anlotinib led to higher tumor response rates compared with pembrolizumab alone.
Based on independent review:
Objective response rate (ORR)
- 57% with the combination
- 40% with pembrolizumab
(p = 0.0001)
Disease control rate (DCR)
- 86% vs 79%
- (p = 0.047)
Complete responses (CRs)
- Rare in both groups (1 patient in each arm)
Most responses were partial rather than complete, but the higher ORR suggests that adding anti-angiogenic therapy may increase the likelihood of tumor shrinkage.
In terms of durability:
- Median duration of response was 12.3 months with the combination
- It was not yet reached with pembrolizumab, although follow-up is still limited
Overall Survival: Not Yet Mature
At the time of this progression-free survival analysis, overall survival data were still immature.
Deaths had occurred in:
- 27% of patients in the benmelstobart + anlotinib group
- 28% of patients in the pembrolizumab group
The estimated 12-month overall survival rates were:
- 74% with benmelstobart + anlotinib
- 71% with pembrolizumab
These early data are not sufficient to define whether the PFS advantage will translate into a meaningful overall survival benefit. This is a critical issue, especially given prior experience with other IO + anti-angiogenic strategies where improved PFS did not always lead to improved OS.
For this reason, the clinical significance of CAMPASS remains promising but not yet definitive.
Subgroup Findings
Prespecified subgroup analyses suggested that the PFS benefit was not equally distributed across all biologic subgroups.
Signals appeared more favorable in:
- PD-L1 TPS ≥50%
- Squamous histology
- Patients with liver metastases
In contrast, benefit was less clear in:
- PD-L1 TPS 1–49%
- Non-squamous histology
- Patients with bone metastases
The absence of a clear benefit in the PD-L1 1–49% subgroup is especially important. In current clinical practice, many such patients are treated with immunochemotherapy rather than immunotherapy alone, which means the control arm in CAMPASS may not fully reflect the most relevant modern comparator for this subgroup.
Therefore, while the trial supports the biologic activity of benmelstobart + anlotinib, its most compelling role may ultimately be in:
- patients with PD-L1–high disease
- patients not suitable for chemotherapy
- or selected biologic subgroups in whom VEGF modulation meaningfully enhances immune responsiveness
Safety and Tolerability
As expected, the addition of anti-angiogenic therapy was associated with a higher incidence of treatment-related toxicity. Nearly all patients receiving benmelstobart plus anlotinib experienced at least one adverse event (98%), compared with 88% in the pembrolizumab arm. Importantly, the rate of grade ≥3 treatment-related adverse events was substantially higher with the combination (59% vs 29%).
The most prominent high-grade toxicity was hypertension, occurring in 26% of patients in the combination arm compared with 3% in the pembrolizumab group. Other adverse events more frequently observed with benmelstobart plus anlotinib were consistent with the known profile of anti-angiogenic therapies and included palmar–plantar erythrodysesthesia, proteinuria, hemoptysis, liver enzyme elevations, and metabolic disturbances.
Despite the higher overall toxicity burden, the incidence of serious treatment-related adverse events was relatively comparable between groups (25% vs 21%). Treatment-related deaths were uncommon, occurring in 1% of patients receiving the combination and 2% in the pembrolizumab arm.
Overall, the safety profile of the combination was consistent with prior experience using checkpoint inhibitors and VEGF-targeted therapies, with no unexpected safety signals identified. Investigators noted that most adverse events were manageable through standard strategies, including dose interruptions, dose modifications, and supportive care. Additionally, the oral administration of anlotinib may offer practical flexibility in toxicity management compared with fixed-dose biologic anti-angiogenic agents.
Clinical Implications
At present, the data support benmelstobart + anlotinib as a potential first-line treatment option for patients with:
- driver-negative
- PD-L1–positive
- advanced NSCLC
The signal appears especially relevant in:
- PD-L1 TPS ≥50%
- chemotherapy-ineligible or chemotherapy-averse patients
- possibly selected squamous NSCLC populations
However, several important caveats remain:
- Overall survival is immature
- The comparator arm may be less clinically relevant for PD-L1 1–49% disease
- Toxicity is clearly greater than with pembrolizumab monotherapy
- The all-Chinese population limits immediate global generalization
So while CAMPASS is clearly positive for PFS, it is not yet fully practice-defining on a global scale.
Key Takeaway Messages
- The phase 3 CAMPASS trial showed that benmelstobart plus anlotinib significantly improved progression-free survival compared with pembrolizumab alone in first-line PD-L1–positive, driver-negative advanced NSCLC.
- Median PFS improved from 7.1 months to 11.0 months with a hazard ratio of 0.70.
- The combination also produced a higher objective response rate and higher disease control rate.
- The benefit appeared more pronounced in PD-L1 TPS ≥50%, squamous histology, and possibly liver-metastatic disease.
- Toxicity was substantially higher, particularly hypertension and anti-angiogenic class-associated events, but remained broadly manageable.
- Overall survival remains immature, and this will ultimately determine whether the regimen becomes broadly practice-changing.
- CAMPASS strengthens the concept that immune checkpoint inhibition plus angiogenesis modulation is a valid and clinically active first-line strategy in NSCLC.
Read Full Article Here