For several years, the treatment logic in hormone receptor–positive, ERBB2-negative advanced breast cancer has seemed straightforward: use the most active endocrine-based combination as early as possible. CDK4/6 inhibitors moved rapidly into first-line care because they consistently prolonged progression-free survival when added to endocrine therapy. But one important question remained unresolved. Does giving a CDK4/6 inhibitor in the first-line setting actually improve overall survival compared with reserving it for second-line treatment?
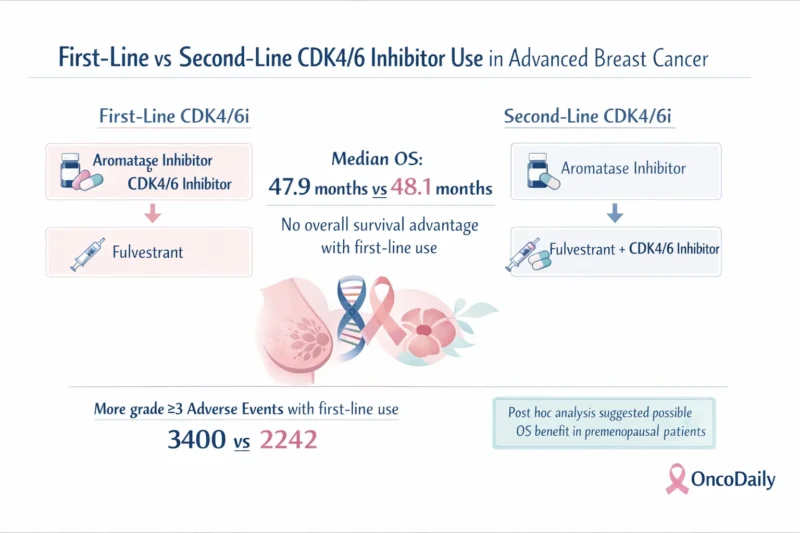
The updated overall survival analysis of the phase 3 SONIA trial now offers one of the clearest answers yet. In this randomized study of 1050 patients, first-line CDK4/6 inhibitor use did not improve overall survival compared with second-line use, with median overall survival of 47.9 months versus 48.1 months, respectively. The first-line strategy also produced substantially more grade 3 or higher adverse events. At the same time, an important post hoc signal emerged: premenopausal patients appeared to derive an overall survival advantage from first-line CDK4/6 inhibitor use, while postmenopausal patients did not (Wortelboer et al., 2026).
This is not a small technical update. It is a study that directly challenges one of the strongest habits in metastatic breast cancer care: assuming that earlier use of CDK4/6 inhibition is always better.
Why the SONIA Trial Mattered From the Beginning
The central question behind SONIA was not whether CDK4/6 inhibitors work. That was already well established. The question was whether they need to be used immediately in all eligible patients.
The trial compared two treatment strategies. In the first, patients received an aromatase inhibitor plus a CDK4/6 inhibitor as first-line treatment, followed by fulvestrant alone in second line. In the second, patients started with an aromatase inhibitor alone and then received fulvestrant plus a CDK4/6 inhibitor in second line. This design allowed investigators to test not only the value of the drug class, but its optimal placement in the treatment sequence.
That distinction is critical in hormone receptor–positive metastatic disease, where patients often receive several lines of endocrine-based treatment over time. The key issue is not simply which first-line regimen delays progression longest. It is which overall strategy gives the best long-term outcome with the least unnecessary toxicity and cost.
Trial Design and Patient Population
SONIA was a multicenter phase 3 randomized clinical trial conducted in the Netherlands. Patients with HR-positive, ERBB2-negative advanced breast cancer who had not received prior treatment for advanced disease were randomized 1:1 to receive a CDK4/6 inhibitor either in first line or in second line.
A total of 1050 patients were enrolled. Among them, 524 were assigned to the first-line CDK4/6 inhibitor strategy and 526 to the second-line strategy. The median age was 64 years, and the data cutoff for this prespecified updated analysis was September 1, 2024. Median follow-up reached 58.5 months, providing a mature dataset for overall survival evaluation (Wortelboer et al., 2026).
The primary endpoint remained progression-free survival after two lines of therapy, or PFS2, while overall survival was a key secondary endpoint planned for analysis once all patients had at least three years of follow-up.
The Main Overall Survival Result
The headline finding was simple and important: first-line CDK4/6 inhibitor use did not improve overall survival.
Median overall survival was 47.9 months in the first-line group and 48.1 months in the second-line group. The hazard ratio was 0.91 with a 95% confidence interval of 0.77 to 1.07, and the difference was not statistically significant (P = .24) (Wortelboer et al., 2026).
This means that, in the overall study population, using a CDK4/6 inhibitor earlier did not translate into longer life compared with using the same class later. That finding is striking because it goes against the assumption that first-line intensification necessarily produces the best long-term outcome.
The Trade-Off: More Toxicity Without Survival Gain
The survival findings would already be important on their own. But they become even more meaningful when paired with the safety results.
First-line CDK4/6 inhibitor use was associated with substantially more grade 3 or higher adverse events, with 3400 events reported in the first-line group compared with 2242 events in the second-line group (Wortelboer et al., 2026).
That imbalance matters. If earlier use had clearly prolonged survival, clinicians might accept more toxicity as the price of better long-term benefit. But when overall survival is essentially the same, the extra toxicity becomes far more clinically relevant. In that context, the study raises a practical question many clinicians and patients will now ask more directly: if survival is not improved, is routine first-line exposure always justified?
Why the Premenopausal Signal Stands Out
The most intriguing finding in the paper came from the post hoc subgroup analysis. Here, the investigators found that premenopausal patients appeared to benefit from first-line CDK4/6 inhibitor use, with a hazard ratio for overall survival of 0.53. By contrast, postmenopausal patients showed no overall survival advantage, with a hazard ratio of 1.00. The interaction between menopausal status and treatment timing was statistically significant (P for interaction = .01) (Wortelboer et al., 2026).
This result should be interpreted cautiously because it was post hoc, not a prespecified primary hypothesis. Still, it is clinically provocative. It suggests that menopausal status may influence not only endocrine sensitivity, but also the value of treatment sequencing.
If confirmed, this could become one of the most practice-changing aspects of SONIA. Rather than using the same sequencing logic for all patients, clinicians may begin to think more selectively, potentially favoring first-line CDK4/6 inhibition more strongly in premenopausal women while feeling more comfortable deferring it in many postmenopausal patients.
Why the Similar Subsequent Therapy Patterns Matter
One possible explanation for the similar overall survival results is that patients in both groups were ultimately able to access effective later therapies. The SONIA analysis supports that interpretation.
Among patients who discontinued second-line treatment, 84.8% in the first-line group and 84.2% in the second-line group went on to receive subsequent anticancer therapy, with similar treatment patterns between groups (Wortelboer et al., 2026).
That finding matters because it reduces the likelihood that the second-line strategy underperformed simply because patients failed to receive later therapy. Instead, it suggests that the sequencing itself may truly be comparable in long-term survival terms, at least in the overall population studied.
What This Means for Clinical Practice
The SONIA trial does not mean CDK4/6 inhibitors are less important. It means their timing may matter differently than many assumed.
For years, the field has often treated first-line CDK4/6 inhibitor use as the obvious default in HR-positive, ERBB2-negative metastatic disease. SONIA introduces more nuance. The updated data suggest that routine first-line use may not improve overall survival for the overall population, while it does increase toxicity. That makes room again for individualized treatment planning, especially in patients with lower-burden, endocrine-sensitive, non-visceral disease where a stepwise approach may still be clinically reasonable.
At the same time, the premenopausal subgroup signal is too important to ignore. Even if exploratory, it points toward a possible group in whom earlier use may matter more.

A Study That Reopens the Sequencing Conversation
What makes SONIA so valuable is that it asks a harder and more clinically relevant question than most trials. It is not simply a comparison of stronger versus weaker treatment. It is a comparison of treatment strategies across time.
That is exactly the kind of question metastatic breast cancer care needs more often. Patients do not experience their treatment as isolated lines of therapy. They experience it as a sequence, with each decision shaping what comes next. SONIA reminds the field that in endocrine-sensitive advanced breast cancer, the sequence itself can be as important as the drugs used.
Bottom Line
The updated overall survival analysis of the phase 3 SONIA trial shows that first-line CDK4/6 inhibitor use did not improve overall survival compared with second-line use in patients with HR-positive, ERBB2-negative advanced breast cancer, with median survival of 47.9 months versus 48.1 months. The first-line strategy also led to more grade 3 or higher toxic effects. However, a post hoc subgroup analysis suggested a possible overall survival advantage for premenopausal patients receiving CDK4/6 inhibitors in first line (Wortelboer et al., 2026).
The broader message is clear: in advanced breast cancer, earlier is not always better, and treatment sequencing deserves the same level of attention as drug selection itself.
Read full abstract here


