For years, the idea has been tempting. If CDK4/6 inhibitors work so well in metastatic estrogen receptor-positive, HER2-negative breast cancer, and now have established roles in the adjuvant setting, why not move them earlier and use them before surgery to spare some patients chemotherapy? The problem is that neoadjuvant CDK4/6 inhibitor studies have repeatedly run into the same obstacle: biologic activity is clear, but conventional endpoints have been less convincing. Pathologic complete response rates remain low, broad replacement of chemotherapy has not happened, and the field has been left with a more precise question. The issue may not be whether neoadjuvant CDK4/6 inhibition works at all, but rather which tumors truly benefit from it.
The randomized phase II PREDIX LumB trial now provides one of the most clinically relevant answers so far. The study, published in Nature Communications, compared weekly paclitaxel with palbociclib plus endocrine therapy in patients with ER-positive, HER2-negative breast cancer larger than 20 mm and or node-positive disease, while also testing whether treatment sequence mattered.
The headline efficacy result was not practice-changing on its own: there was no statistically significant difference in objective radiologic response at 12 weeks between chemotherapy-first and palbociclib-plus-endocrine-first treatment, and long-term outcomes were similar. But the translational finding may prove more important than the primary endpoint.
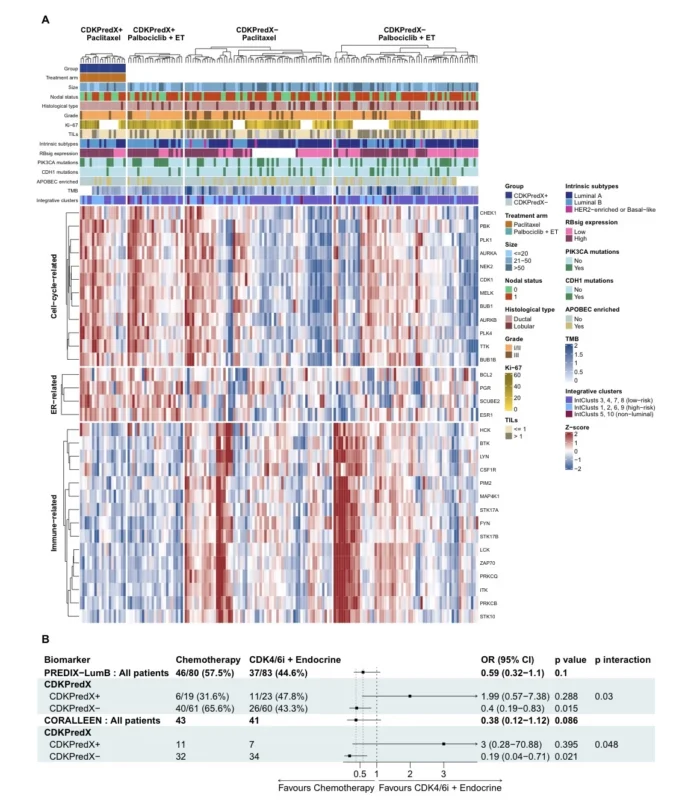
The investigators developed a 31-gene predictive signature, CDKPredX, that identified tumors resistant to chemotherapy yet responsive to palbociclib plus endocrine therapy, and they independently validated it in the CORALLEEN trial (Matikas et al., 2026).
That makes PREDIX LumB more than another “negative” CDK4/6 neoadjuvant trial. It may be one of the first studies to show how neoadjuvant palbociclib could become clinically useful if it is guided by biology rather than offered broadly.
© The Author(s) 2026
Why PREDIX LumB Mattered
Neoadjuvant therapy in ER-positive, HER2-negative breast cancer has always lived in a different world from triple-negative or HER2-positive disease. Pathologic complete response is uncommon, and treatment goals are often more nuanced: tumor shrinkage to facilitate breast-conserving surgery, control of nodal disease, and selection of patients for postoperative escalation or de-escalation. In this subtype, endocrine responsiveness, proliferation, and immune context all influence treatment behavior in ways that standard clinicopathologic variables do not fully capture.
Several earlier neoadjuvant studies suggested that CDK4/6 inhibitors produce strong suppression of proliferation, including complete cell cycle arrest in some tumors, but they did not clearly outperform chemotherapy on the endpoints clinicians rely on most (Ma et al., 2017; Prat et al., 2020; Hurvitz et al., 2020). What has been missing is a validated predictor that separates tumors that truly need chemotherapy from those that may instead remain highly sensitive to CDK4/6 blockade plus endocrine therapy.
PREDIX LumB was built around that gap.
How The Trial Was Designed
PREDIX LumB was an academic, randomized, open-label phase II study conducted in Sweden. Patients with ER-positive, HER2-negative breast cancer were eligible if tumors were larger than 2 cm and or node-positive. Patients were randomized 1:1 to one of two sequences. In arm A, they received weekly paclitaxel for 12 weeks followed by 12 weeks of palbociclib plus endocrine therapy. In arm B, they received the same treatments in the reverse order.
The primary endpoint was objective radiologic response at 12 weeks, which in practical terms compared the early activity of paclitaxel versus palbociclib plus endocrine therapy. Secondary endpoints included objective response at 24 weeks, pathologic complete response, event-free survival, relapse-free survival, overall survival, safety, health-related quality of life, and a large translational program using tissue, imaging, genomic, and transcriptomic data (Matikas et al., 2026).
A total of 179 patients comprised the intention-to-treat population. About two thirds had pathologically confirmed nodal involvement before treatment, more than 80% had progesterone receptor-positive tumors, and baseline characteristics were largely balanced, although median Ki67 was slightly higher in arm A.
The Primary Endpoint Did Not Cross The Line
The main efficacy result was close, but not statistically significant. Objective radiologic response at 12 weeks was 59% in the paclitaxel-first group and 45% in the palbociclib-plus-endocrine-first group, with a p value of 0.058. That means the study did not meet its primary endpoint.
At 24 weeks, after both groups had received both treatments in opposite sequence, objective response rates were 78% in arm A and 71% in arm B, again without a significant difference. Pathologic complete response was rare, as expected in this subtype, occurring in eight patients overall: three in arm A and five in arm B. Event-free survival, relapse-free survival, and overall survival were also similar between the two strategies. Five-year event-free survival was 80.6% in arm A and 79.7% in arm B, and five-year overall survival was 95.7% versus 93.2%, respectively (Matikas et al., 2026).
So, at the level of broad clinical efficacy, PREDIX LumB does not support replacing chemotherapy with palbociclib plus endocrine therapy for all comers in high-risk ER-positive, HER2-negative disease. It also does not suggest that the sequence of chemotherapy and CDK4/6 blockade meaningfully changes long-term outcome.
© The Author(s) 2026
But The Study Did Not End There
If the paper had stopped at the efficacy endpoints, it would have been easy to dismiss. That would be a mistake.
The real value of PREDIX LumB lies in its biomarker work. The investigators performed whole-exome sequencing and RNA sequencing on baseline fresh frozen tumor tissue, then explored which biologic patterns were associated with response to each treatment.
The signal was coherent. Tumors that responded to paclitaxel showed more immune-related activity and lower estrogen receptor-related signaling. In contrast, tumors that responded to palbociclib plus endocrine therapy were characterized by proliferation and cell-cycle related pathways. That is an important distinction because it argues against a simplistic assumption that more proliferative ER-positive tumors are always best served by chemotherapy. In some tumors, high proliferation may still coexist with preserved endocrine and CDK4/6 sensitivity.
Building on these transcriptomic findings, the investigators created a 31-gene predictive signature called CDKPredX. The signature integrated genes related to proliferation, ER signaling, and immune activity. The biologic profile it captured was specific: tumors with high proliferation, high ER pathway activity, and low immune gene expression. These tumors defined the CDKPredX-positive subgroup.
CDKPredX Was The Most Important Result In The Paper
The value of CDKPredX was not merely descriptive. It predicted differential treatment sensitivity.
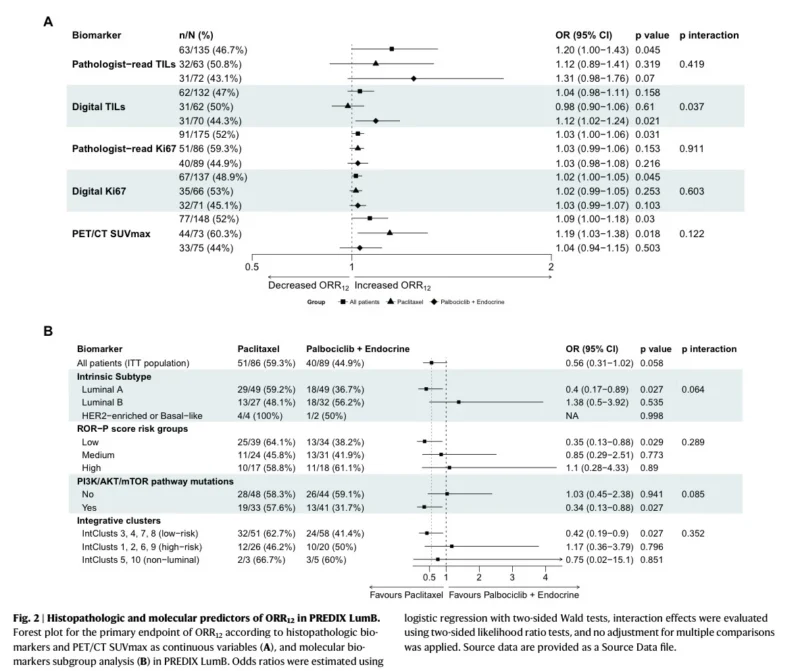
In PREDIX LumB, a statistically significant interaction between CDKPredX subgroup and treatment was observed, with a p interaction of 0.03. CDKPredX-positive tumors were relatively resistant to chemotherapy yet retained sensitivity to palbociclib plus endocrine therapy. Objective response at 12 weeks in CDKPredX-positive disease was 47.8% with palbociclib plus endocrine therapy versus 31.6% with chemotherapy. In contrast, CDKPredX-negative tumors responded better to chemotherapy, with ORR12 of 65.6% versus 43.3% for palbociclib plus endocrine therapy (Matikas et al., 2026).
This finding would already have been notable if it had stayed confined to the discovery cohort. But the investigators then tested the signature in the CORALLEEN trial, another randomized study comparing ribociclib plus endocrine therapy with chemotherapy in luminal B breast cancer. The interaction held up there as well, with a p interaction of 0.048. In that validation set, CDKPredX-positive tumors again favored CDK4/6 inhibitor plus endocrine therapy, while CDKPredX-negative tumors favored chemotherapy.
That is why this study matters. PREDIX LumB did not show that palbociclib should replace chemotherapy broadly. It showed something more useful: a path toward selecting tumors in which chemotherapy may be the weaker biologic choice.
What Else The Biomarker Work Showed
Several traditional and semi-traditional biomarkers were prognostic for response, but not truly predictive of differential treatment effect. Baseline pathologist-read Ki67, digital Ki67, tumor-infiltrating lymphocytes, and PET SUVmax were associated with likelihood of objective response at 12 weeks regardless of treatment. Similarly, intrinsic subtype, ROR-P risk group, PI3K/AKT/mTOR pathway alterations, and integrative clusters did not provide a clean treatment-specific separation.
That is another reason CDKPredX stands out. It did what many individual markers have failed to do in this space: predict relative benefit between chemotherapy and CDK4/6-based endocrine therapy rather than merely stratify prognosis.
Safety And Quality Of Life Tell A Familiar Story
The safety results were consistent with what clinicians would expect.
During paclitaxel, the most common adverse events were peripheral sensory neuropathy, rash, and fatigue. Grade 3 or 4 paclitaxel-related events included peripheral sensory neuropathy and neutropenia. During palbociclib, the most common adverse events were neutropenia and nausea, with grade 3 or 4 toxicity dominated by neutropenia. No major differences in the overall frequency of any-grade or grade 3 or 4 adverse events were seen between the two treatment sequences, and no new safety signals emerged (Matikas et al., 2026).
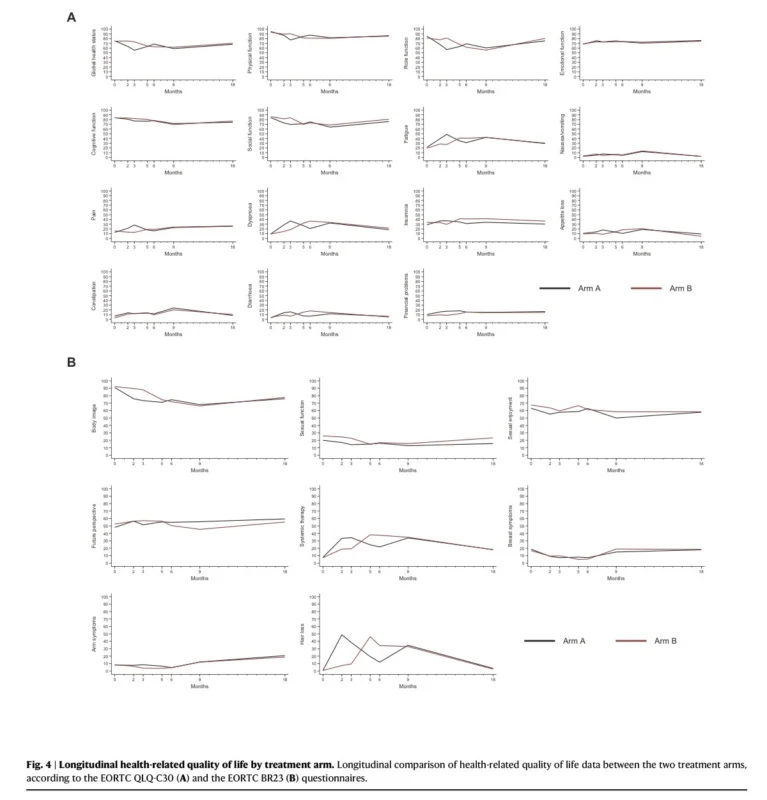
Quality-of-life data, however, help explain why biomarker-guided de-escalation remains attractive. Health-related quality of life was better preserved during the first 12 weeks with palbociclib plus endocrine therapy than with paclitaxel, particularly in domains such as dyspnea, diarrhea, and hair loss. After treatment switch, the burden shifted accordingly. By one year after surgery, most quality-of-life domains had returned to baseline, but during treatment itself, palbociclib was clearly the more tolerable early experience.
That is clinically relevant. A biomarker-defined group that is less sensitive to chemotherapy and still responsive to CDK4/6 blockade would not only receive a biologically rational treatment, but potentially a more tolerable one.
What This Means For Practice
The short answer is that PREDIX LumB does not change current standard practice on its own. Neoadjuvant chemotherapy remains appropriate for many high-risk ER-positive, HER2-negative tumors, and the study did not show superiority of palbociclib plus endocrine therapy in the overall population.
But the longer and more important answer is that this study may change how the field thinks about the problem. The failure of neoadjuvant CDK4/6 trials may not be that the drugs are ineffective before surgery. The problem may be that they have been tested in populations that are too biologically broad.
CDKPredX offers a potential way forward. It identifies a subgroup with high proliferation, intact ER-driven biology, and relatively low immune activation, tumors that appear resistant to chemotherapy but still sensitive to CDK4/6 inhibition plus endocrine therapy. That is exactly the kind of biologic distinction precision oncology is supposed to make.
The authors are right to be cautious. The study was phase II, the sample size was modest, follow-up remains limited for a disease with a long natural history, and CDKPredX still needs development as a practical single-sample predictor and prospective clinical utility testing. But this is one of the strongest biomarker signals yet in the neoadjuvant CDK4/6 field.
© The Author(s) 2026
The Bigger Message
PREDIX LumB also contributes to a broader shift in breast oncology. ER-positive, HER2-negative disease has long been treated with a more generalized framework than its biologic diversity justifies. Yet the transcriptomic and multi-omic work in this trial reinforces what has become increasingly clear: these tumors are not one disease. Some are highly endocrine driven and proliferation rich. Some are more immune active. Some respond well to chemotherapy. Some likely do not.
That heterogeneity matters most when clinicians are trying to choose between therapies that are active but not interchangeable.
Conclusion
PREDIX LumB did not prove that neoadjuvant palbociclib plus endocrine therapy should replace chemotherapy in ER-positive, HER2-negative breast cancer. The primary endpoint was not met, pCR rates remained low, and long-term outcomes were similar between sequences.
But that is not the most important conclusion from the study.
The key result is that a transcriptomic predictor, CDKPredX, identified a subgroup of tumors resistant to chemotherapy yet still responsive to palbociclib plus endocrine therapy, and that finding held up in independent validation. If confirmed prospectively, that would represent one of the clearest biomarker-guided entry points for neoadjuvant CDK4/6 inhibition so far.
In other words, PREDIX LumB may not have changed practice today. But it may have shown exactly how practice could change tomorrow.
Read full article here


