The phase III INAVO120 trial added an important practical message at ESMO Breast Cancer 2026: the efficacy benefit of inavolisib plus palbociclib and fulvestrant was observed in patients with PIK3CA-mutated, hormone receptor-positive, HER2-negative, endocrine-resistant advanced breast cancer, regardless of whether patients developed inavolisib-associated hyperglycemia.
Presented by Prof. Dr. med. Sibylle Loibl during Rapid Oral Session 2, this analysis focused on a clinically relevant question for PI3K pathway inhibition: does the occurrence of hyperglycemia, an on-target toxicity of PI3K inhibitors, affect the interpretation of treatment benefit?
Why This Analysis Matters
PIK3CA mutations are among the most common actionable alterations in HR-positive, HER2-negative breast cancer. Targeting the PI3K pathway has become an important strategy, but toxicity, especially hyperglycemia, remains a central management issue.
INAVO120 had previously shown statistically significant improvements in investigator-assessed progression-free survival and overall survival with inavolisib plus palbociclib plus fulvestrant compared with placebo plus palbociclib plus fulvestrant in this endocrine-resistant population (Loibl et al., 2026).
At the final analysis, hyperglycemia was reported in 63.4% of patients receiving inavolisib compared with 13.5% in the placebo arm. This ESMO Breast Cancer 2026 analysis therefore examined efficacy outcomes among patients in the inavolisib arm who did and did not develop hyperglycemia.

INAVO120 Study Design
INAVO120 enrolled 325 patients with PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer. The study enriched for patients with poor-prognosis endocrine-resistant disease, including those who had progression during or within 12 months of adjuvant endocrine therapy.
Patients were randomized 1:1 to receive either inavolisib 9 mg orally once daily plus palbociclib 125 mg orally on days 1–21 of each 28-day cycle plus fulvestrant 500 mg, or placebo plus the same palbociclib and fulvestrant backbone.
Key eligibility criteria included measurable disease, no prior therapy for advanced breast cancer, fasting glucose below 126 mg/dL, and HbA1c below 6.0. The primary endpoint was investigator-assessed progression-free survival, while secondary endpoints included overall survival, objective response rate, duration of response, and patient-reported outcomes.
For this analysis, hyperglycemia was defined as any reported hyperglycemia adverse event, regardless of grade, duration, or treatment received. The data cutoff was November 15, 2024.
Efficacy Was Improved Regardless of Hyperglycemia Status
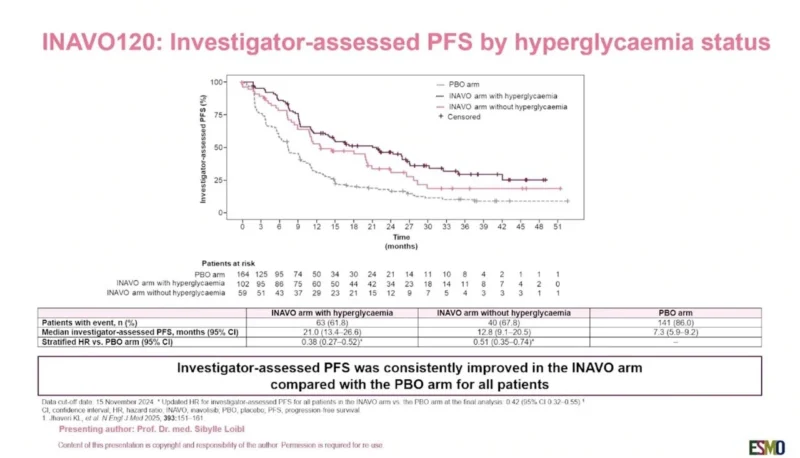
Among the 161 patients in the inavolisib arm, 102 patients developed any-grade hyperglycemia and 59 patients did not. All inavolisib-treated patients, whether they developed hyperglycemia or not, showed improved efficacy compared with the placebo arm.
Median investigator-assessed progression-free survival was 21.0 months in patients receiving inavolisib who developed hyperglycemia and 12.8 months in those who did not develop hyperglycemia, compared with 7.3 months in the placebo arm. The stratified hazard ratio versus placebo was 0.38 for patients with hyperglycemia and 0.51 for those without hyperglycemia.
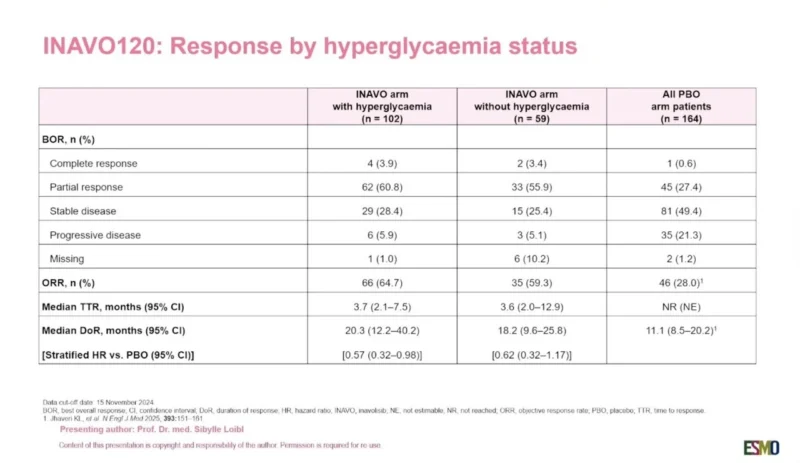
Objective response rates were also higher with inavolisib regardless of hyperglycemia status. ORR was 64.7% in the inavolisib-with-hyperglycemia group and 59.3% in the inavolisib-without-hyperglycemia group, compared with 28.0% in the placebo arm.
Duration of response also favored inavolisib. Median DoR was 20.3 months in patients with hyperglycemia and 18.2 months in patients without hyperglycemia, compared with 11.1 months in the placebo group.

Overall Survival Remained Favorable in Both Inavolisib Groups
Overall survival was also numerically improved in both inavolisib subgroups compared with placebo.
Median overall survival was 34.9 months in patients with hyperglycemia and 32.7 months in patients without hyperglycemia, compared with 27.0 months in the placebo arm. The hazard ratio versus placebo was 0.66 for patients with hyperglycemia and 0.72 for patients without hyperglycemia.
These results suggest that the inavolisib benefit was maintained across hyperglycemia-defined subgroups. However, the comparison between patients with and without hyperglycemia should be interpreted cautiously because hyperglycemia was not a randomized variable and baseline differences existed between the groups.
Does Hyperglycemia Mean Better Efficacy?
Patients who developed hyperglycemia often had numerically better outcomes than those who did not, but this should not be interpreted as proof that hyperglycemia itself causes improved efficacy.
The abstract noted some baseline imbalances between inavolisib-treated patients with and without hyperglycemia, including median weight and body mass index. Patients who developed hyperglycemia also had a longer median treatment duration with inavolisib, 14.6 months versus 11.2 months, which may partly reflect longer exposure to therapy.
The clinically important message is more practical: hyperglycemia did not eliminate the treatment benefit of inavolisib, and glucose management allowed many patients to remain on long-term treatment.
Long-Term Treatment Was Possible With Glucose Management
Despite the high prevalence of hyperglycemia in the inavolisib arm, a meaningful proportion of patients remained on treatment for at least 24 months. In the inavolisib arm, 29 patients with hyperglycemia and 12 patients without hyperglycemia were treated for 24 months or longer, compared with 20 patients in the placebo arm.
This supports the role of proactive metabolic monitoring and glucose management when using PI3K pathway inhibitors. For clinicians, the question is not only whether hyperglycemia occurs, but whether it can be identified early, managed effectively, and prevented from compromising treatment continuity.
Clinical Meaning
This INAVO120 analysis reinforces the efficacy of inavolisib plus palbociclib and fulvestrant in a high-risk, endocrine-resistant, PIK3CA-mutated advanced breast cancer population. The treatment benefit was seen both in patients who developed hyperglycemia and those who did not.
For clinical practice, the data highlight two parallel messages. First, inavolisib provides meaningful disease control compared with placebo when added to palbociclib and fulvestrant. Second, hyperglycemia remains an important, expected toxicity that requires active monitoring and management, but it does not appear to negate the efficacy benefit.
Clinical Takeaway
INAVO120 showed that inavolisib plus palbociclib and fulvestrant improved outcomes compared with placebo plus palbociclib and fulvestrant in patients with PIK3CA-mutated, HR-positive, HER2-negative endocrine-resistant advanced breast cancer, regardless of hyperglycemia status.
The analysis supports the continued importance of PI3K pathway targeting in this setting while emphasizing that glucose management is essential to sustain long-term therapy.


