HER2CLIMB-02 trial was presented during the ESMO Breast Cancer Annual Congress 2026 by Giuseppe Curigliano and evaluated the addition of tucatinib to trastuzumab emtansine (T-DM1) in patients with previously treated HER2-positive locally advanced or metastatic breast cancer. The updated analysis showed that tucatinib plus T-DM1 continued to provide a progression-free survival benefit, including in patients with baseline brain metastases, although the final overall survival analysis did not show a statistically significant difference between treatment arms.
Background
HER2-positive metastatic breast cancer remains a biologically aggressive disease, and treatment decisions become more complex after prior exposure to trastuzumab and taxane-based therapy. Brain metastases are also common in this population and represent an important clinical challenge, making intracranial disease control and durable systemic benefit key treatment goals.
Tucatinib is an oral HER2-directed tyrosine kinase inhibitor with known activity in HER2-positive metastatic breast cancer, including in patients with brain metastases. In the primary analysis of HER2CLIMB-02, adding tucatinib to T-DM1 significantly improved progression-free survival compared with T-DM1 plus placebo, with a manageable safety profile.
The updated analysis presented at ESMO Breast Cancer Annual Congress 2026 focused on longer-term efficacy outcomes, including final overall survival and updated investigator-assessed progression-free survival.
Study Design and Methods
HER2CLIMB-02 was a randomized trial enrolling patients aged 18 years or older with centrally confirmed HER2-positive locally advanced or metastatic breast cancer. Eligible patients had previously received trastuzumab and a taxane, and patients with brain metastases were permitted to participate.
A total of 463 patients were randomly assigned in a 1:1 ratio to receive T-DM1 with either tucatinib or placebo. T-DM1 was given at 3.6 mg/kg intravenously every 21 days. Tucatinib was administered orally at 300 mg twice daily, while the control arm received placebo twice daily.
This updated analysis evaluated overall survival as a secondary endpoint and updated investigator-assessed progression-free survival as an exploratory endpoint. Outcomes were assessed in the total study population and among patients with baseline brain metastases. Subsequent anticancer therapies were also analyzed.
Results from HER2CLIMB-02 Trial
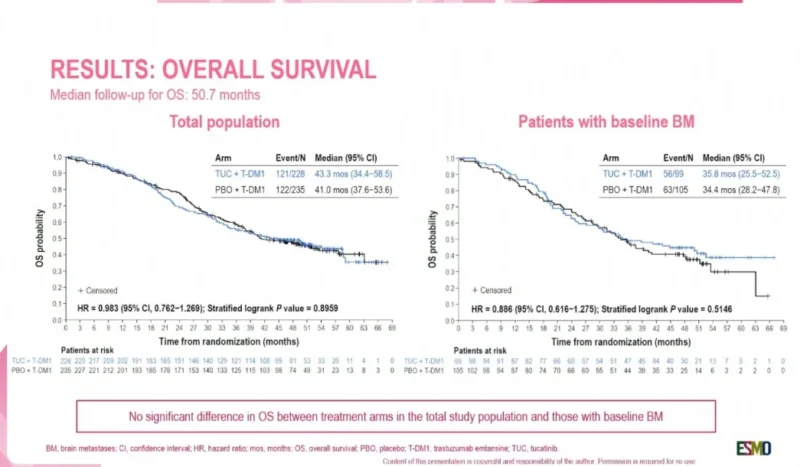
At the data cutoff of October 10, 2025, the median follow-up for overall survival was 50.7 months. In the total study population, final overall survival did not significantly differ between treatment arms. Median overall survival was 43.3 months with tucatinib plus T-DM1 compared with 41.0 months in the control arm, with a hazard ratio of 0.983 and a stratified log-rank P value of 0.8959.
Among patients with baseline brain metastases, median overall survival was 35.8 months with tucatinib plus T-DM1 and 34.4 months in the control arm. This subgroup also showed no statistically significant overall survival difference, with a hazard ratio of 0.886 and a stratified log-rank P value of 0.5146.
Despite the absence of an overall survival advantage, the updated progression-free survival analysis confirmed the benefit of adding tucatinib to T-DM1. Median progression-free survival was 9.5 months in the tucatinib arm compared with 7.4 months in the control arm, corresponding to a hazard ratio of 0.739.
In patients with baseline brain metastases, median progression-free survival was also longer with tucatinib plus T-DM1, at 7.8 months compared with 5.7 months in the control arm.
Subsequent anticancer therapy use was similar between the two arms, with 83.7% of patients in the tucatinib arm and 87.8% in the control arm receiving further systemic therapy. T-DXd was reported as the preferred first subsequent anti-HER2 therapy. The updated safety analysis remained consistent with the primary analysis, with no new safety signals identified.
Key Findings
The final overall survival analysis of HER2CLIMB-02 did not show a statistically significant difference between tucatinib plus T-DM1 and control therapy after 50.7 months of follow-up.
However, the progression-free survival benefit observed in the primary analysis was maintained with longer follow-up. This benefit was seen both in the total population and in patients with baseline brain metastases, supporting the activity of tucatinib plus T-DM1 in a clinically important subgroup.
The safety profile remained consistent with prior findings, and no new safety concerns were reported in the updated analysis.
Conclusion
The HER2CLIMB-02 updated efficacy analysis showed that adding tucatinib to T-DM1 continued to improve progression-free survival in previously treated HER2-positive locally advanced or metastatic breast cancer, including among patients with baseline brain metastases. However, this benefit did not translate into a statistically significant improvement in overall survival in the final analysis.


