The phase III evERA Breast Cancer trial continues to strengthen the clinical profile of giredestrant plus everolimus in patients with estrogen receptor-positive, HER2-negative advanced breast cancer previously treated with CDK4/6 inhibitors. New safety and patient-reported outcome (PRO) analyses presented by Miguel Martin at the ESMO Breast Cancer 2026 showed that the combination demonstrated manageable toxicity, preserved quality of life, and low rates of treatment discontinuation despite the known risk of everolimus-associated stomatitis.
The updated presentation focused specifically on safety, oral toxicity prevention, and how patients experienced treatment while receiving giredestrant plus everolimus compared with standard endocrine therapy plus everolimus.
Giredestrant is a next-generation oral selective estrogen receptor degrader (SERD) and full estrogen receptor antagonist. In the primary evERA Breast Cancer analysis previously presented at ESMO 2025, the combination of giredestrant plus everolimus demonstrated a statistically significant and clinically meaningful progression-free survival benefit over standard endocrine therapy plus everolimus in patients whose disease progressed after CDK4/6 inhibitor therapy.
The new analysis provides additional context regarding tolerability, an increasingly important issue in metastatic HR-positive breast cancer where prolonged treatment duration depends heavily on maintaining quality of life and minimizing chronic toxicities.
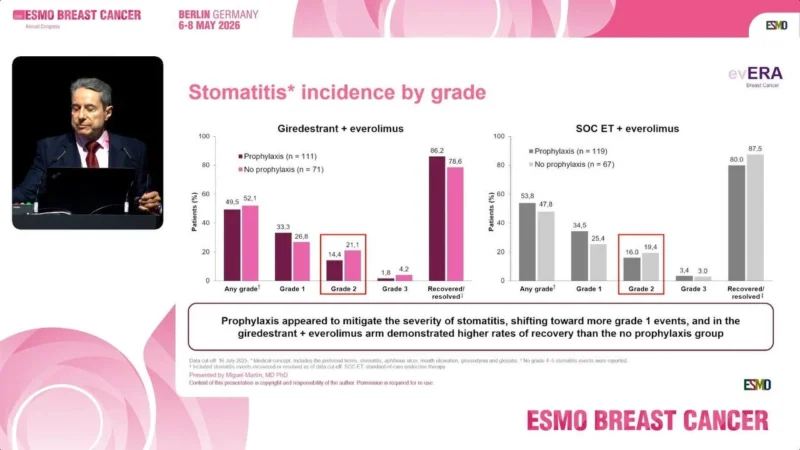
Oral Steroid Mouthwash Reduced Clinically Significant Stomatitis
Stomatitis remains one of the most common adverse events associated with everolimus-containing regimens. In evERA Breast Cancer, dexamethasone mouthwash prophylaxis was strongly recommended and used in 62.5% of patients overall, including 61.0% of patients receiving giredestrant plus everolimus and 64.0% receiving standard endocrine therapy plus everolimus.
The slide data presented at ESMO Breast Cancer 2026 demonstrated that prophylactic steroid mouthwash reduced the incidence of clinically significant stomatitis and delayed symptom onset.
In the giredestrant plus everolimus arm, grade 2 stomatitis occurred in 14.4% of patients receiving prophylaxis compared with 21.1% in those without prophylaxis. Grade 3 stomatitis was also numerically lower with prophylaxis, occurring in 1.8% versus 4.2%, respectively.
Importantly, prophylaxis delayed the median time to stomatitis onset from 13 days to 20 days in the giredestrant-containing arm.
A similar pattern was observed in the standard endocrine therapy plus everolimus group, where prophylaxis delayed median onset from 14.5 days to 25 days. The investigators noted that prophylaxis appeared to shift toxicity toward lower-grade events while improving recovery rates.
Notably, no grade 4 or grade 5 stomatitis events were reported.
Treatment Discontinuations Were Low
Despite the frequency of oral toxicity, treatment modifications and discontinuations remained relatively uncommon across both treatment arms.
In patients receiving giredestrant plus everolimus:
- Dose interruption due to stomatitis occurred in 2.7%
- Everolimus dose reduction occurred in 6.0%
- Everolimus discontinuation occurred in only 1.1%
The standard endocrine therapy plus everolimus arm showed similarly low discontinuation rates.
The presentation specifically highlighted that discontinuations related to stomatitis were low overall, supporting the clinical feasibility of prolonged therapy with proactive supportive care.

Patient-Reported Outcomes Favored Giredestrant Plus Everolimus
Beyond clinician-reported toxicity, the evERA Breast Cancer analysis incorporated extensive patient-reported outcome assessments using EORTC QLQ-C30, PRO-CTCAE, and FACT-GP5 instruments.
The results suggested that the efficacy advantage of giredestrant plus everolimus was not accompanied by worsening patient experience.
Median time to deterioration in role functioning numerically favored the giredestrant arm:
- 7.3 months with giredestrant plus everolimus
- 4.6 months with standard endocrine therapy plus everolimus
- HR 0.73 (95% CI 0.55–0.97)
Physical functioning also numerically favored the investigational regimen:
- 10.4 months versus 7.8 months
- HR 0.80 (95% CI 0.59–1.09)
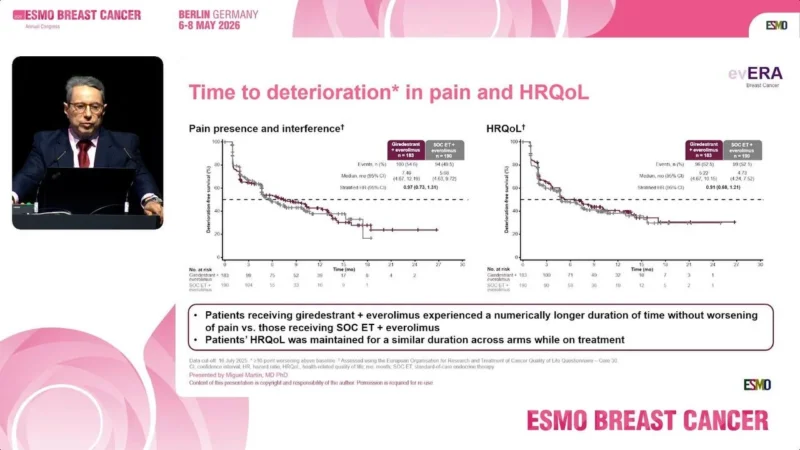
Pain deterioration was delayed numerically with giredestrant plus everolimus:
- 7.5 months versus 5.7 months
Meanwhile, overall health-related quality of life was maintained similarly across both treatment arms.
The investigators emphasized that patients receiving giredestrant plus everolimus experienced a numerically longer duration without worsening pain and no meaningful increase in treatment burden from the patient perspective.
Safety Profile Consistent With Known Everolimus Toxicity
An important observation from the slide conclusions was that no major new toxicity signal appeared attributable to giredestrant itself.
According to the investigators, the safety profile of the combination remained consistent with the known adverse-event profile of everolimus and the underlying disease setting.
The majority of pneumonitis events were described as low grade and reversible, with incidence rates consistent with previous everolimus studies.
This distinction is clinically important because the development of oral SERDs has increasingly focused not only on efficacy but also on tolerability and long-term treatment sustainability.

Why These Findings Matter
The post-CDK4/6 inhibitor setting in HR-positive metastatic breast cancer has become increasingly crowded, with multiple endocrine-targeted strategies competing for sequencing relevance. In this environment, maintaining patient quality of life while preserving efficacy has become a critical differentiator.
The evERA Breast Cancer safety analysis suggests that giredestrant plus everolimus may offer a clinically manageable regimen capable of extending disease control without substantially increasing treatment burden.
The findings also reinforce the importance of prophylactic supportive care strategies, particularly steroid mouthwash prophylaxis when using everolimus-containing regimens.
As oral SERDs continue to move into later-phase development and clinical practice, the balance between efficacy, chronic toxicity, and patient-reported outcomes will likely become increasingly central in treatment selection decisions for metastatic breast cancer.


