Final overall survival results from the phase III CAPItello-291 trial presented at ESMO Breast Cancer 2026 add an important layer to the story of capivasertib plus fulvestrant in hormone receptor-positive, HER2-negative advanced breast cancer.
The main message is nuanced. Capivasertib plus fulvestrant did not significantly improve overall survival compared with placebo plus fulvestrant in either the PIK3CA/AKT1/PTEN-altered population or the overall trial population. However, the regimen continued to show clinically meaningful benefit beyond first progression, including prolonged PFS2 and delayed time to first subsequent chemotherapy.
For a post-aromatase inhibitor population in which resistance biology often involves PI3K/AKT pathway activation, these final data reinforce capivasertib plus fulvestrant as a treatment that improves disease control, even if the final OS endpoint was not statistically positive.

Why CAPItello-291 Matters
CAPItello-291 previously established capivasertib plus fulvestrant as an important targeted strategy for patients with HR-positive, HER2-negative advanced breast cancer whose disease relapsed or progressed during or after aromatase inhibitor therapy. The primary analysis showed a statistically significant and clinically meaningful improvement in progression-free survival in both the overall population and the biomarker-altered population.
That biomarker-altered group included tumors with PIK3CA, AKT1, or PTEN alterations, reflecting activation of the PI3K/AKT pathway. This pathway is a key mechanism of endocrine resistance and has become one of the most clinically relevant therapeutic targets in HR-positive metastatic breast cancer.
The final OS analysis now asks a harder question: does delaying progression translate into longer survival?
Study Design
CAPItello-291 was a global, randomized phase III trial evaluating capivasertib plus fulvestrant versus placebo plus fulvestrant in patients with HR-positive, HER2-negative advanced breast cancer.
Patients were randomized 1:1 to receive capivasertib 400 mg twice daily on an intermittent schedule of four days on and three days off, combined with fulvestrant, or placebo plus fulvestrant.
Overall, 708 patients were randomized:
- 355 patients received capivasertib plus fulvestrant.
- 353 patients received placebo plus fulvestrant.
The PIK3CA/AKT1/PTEN-altered population included 155 patients in the capivasertib arm and 134 patients in the placebo arm.
Overall survival in both the altered and overall populations was a key secondary endpoint. PFS2, defined as time from randomization to second progression or death after subsequent treatment, was a secondary endpoint. Time to first subsequent chemotherapy or death was assessed as an exploratory endpoint.
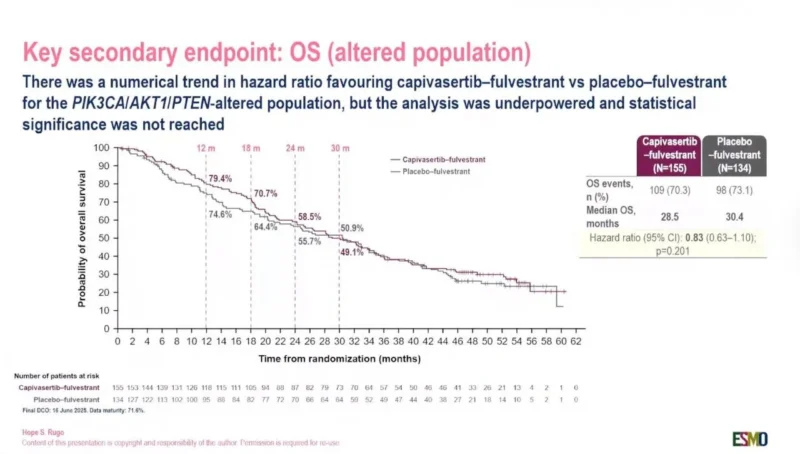
Final OS Results: A Trend, but Not Statistical Significance
In the PIK3CA/AKT1/PTEN-altered population, overall survival numerically favored capivasertib plus fulvestrant, but the difference did not reach statistical significance.
Median OS was 28.5 months with capivasertib plus fulvestrant versus 30.4 months with placebo plus fulvestrant. The hazard ratio was 0.83 with a 95% confidence interval of 0.63–1.10 and a p value of 0.201.
In the overall population, there was no OS difference. Median OS was 29.4 months with capivasertib plus fulvestrant and 28.6 months with placebo plus fulvestrant, with a hazard ratio of 1.00.
This makes the interpretation clear: CAPItello-291 remains a positive trial for progression-free survival, but not for final overall survival.
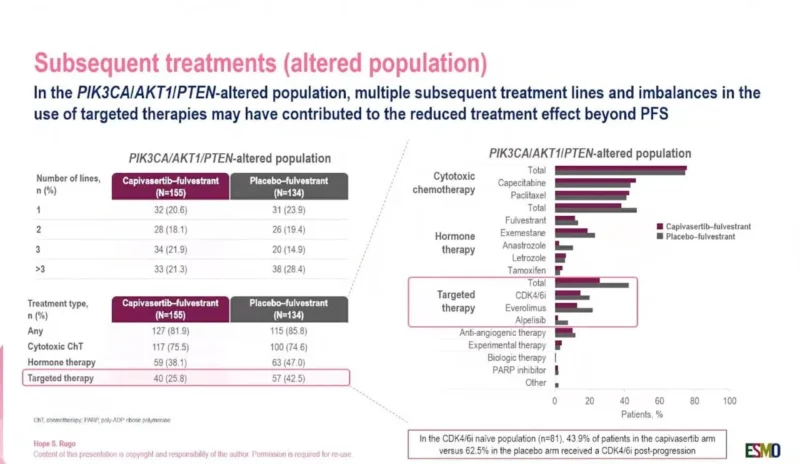
Subsequent Therapy May Have Blunted the OS Signal
The slides presented at ESMO Breast Cancer 2026 provide important context for the survival findings. In the altered population, multiple subsequent treatment lines and imbalances in post-progression therapy may have reduced the ability to detect an OS difference.
Targeted therapy after progression was used more frequently in the placebo–fulvestrant arm than in the capivasertib–fulvestrant arm. In the altered population, subsequent targeted therapy was reported in 25.8% of patients in the capivasertib arm compared with 42.5% in the placebo arm.
The slide also noted that among CDK4/6 inhibitor-naïve patients, 43.9% of those in the capivasertib arm versus 62.5% in the placebo arm received a CDK4/6 inhibitor after progression.
These imbalances matter. In metastatic breast cancer, overall survival is increasingly influenced by therapies received after trial treatment. A strong post-progression treatment effect in the control arm can dilute OS differences, even when the experimental therapy improves earlier disease-control endpoints.
PFS2 Supports Continued Benefit Beyond First Progression
Although OS was not significantly improved, capivasertib plus fulvestrant improved PFS2, suggesting that the benefit of treatment was not limited to first progression.
In the PIK3CA/AKT1/PTEN-altered population, median PFS2 was 15.9 months with capivasertib plus fulvestrant versus 11.1 months with placebo plus fulvestrant, with a hazard ratio of 0.68.
In the overall population, median PFS2 was 15.4 months versus 12.7 months, with a hazard ratio of 0.85.
This endpoint is clinically relevant because it reflects disease control across the treatment sequence rather than only during the study regimen. The PFS2 findings suggest that capivasertib plus fulvestrant provides a sustained delay in disease progression even after subsequent therapy is considered.
Capivasertib Delayed Time to Chemotherapy
The final analysis also showed a meaningful delay in time to first subsequent chemotherapy or death.
In the altered population, median time to first subsequent chemotherapy or death was 11.0 months with capivasertib plus fulvestrant compared with 6.0 months with placebo plus fulvestrant. The hazard ratio was 0.62.
In the overall population, median time to chemotherapy or death was 11.0 months versus 7.0 months, with a hazard ratio of 0.74.
This finding is important in HR-positive metastatic breast cancer, where delaying chemotherapy remains a major treatment goal. For many patients, maintaining an endocrine-based targeted strategy for longer can preserve quality of life and avoid chemotherapy-associated toxicity.
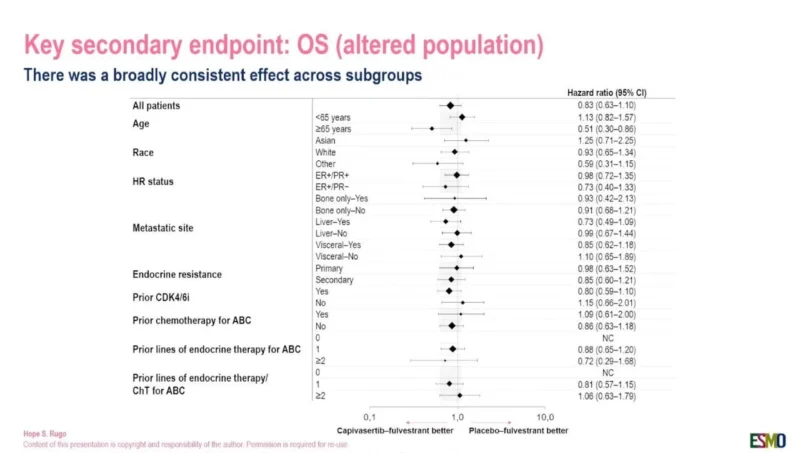
Subgroup Findings Were Broadly Consistent
The subgroup analysis in the altered population showed a broadly consistent OS effect across most clinical subgroups, although confidence intervals were wide and many analyses were underpowered.
The strongest numerical signal appeared in patients aged 65 years or older, with a hazard ratio of 0.51, while patients younger than 65 did not show the same magnitude of benefit. Other subgroups, including liver metastasis status, endocrine resistance category, prior CDK4/6 inhibitor exposure, and prior chemotherapy for advanced breast cancer, showed variable but generally non-definitive patterns.
These data should be interpreted cautiously. The subgroup results are exploratory and do not override the main OS finding.
Safety Remained Consistent With Prior Analysis
With longer follow-up, no new safety signals were observed. This is important because capivasertib targets AKT, and pathway inhibition can be associated with adverse events such as diarrhea, rash, hyperglycemia, and treatment interruptions.
The final analysis supports the previously described safety profile and does not suggest new cumulative toxicity concerns.
Clinical Interpretation
The CAPItello-291 final OS analysis does not change the central evidence base for capivasertib plus fulvestrant. The regimen remains supported by its progression-free survival benefit, particularly in the PIK3CA/AKT1/PTEN-altered population.
However, these final data refine the message. Capivasertib plus fulvestrant should not be described as improving overall survival. Instead, its value lies in improving disease control, delaying second progression, and postponing the need for chemotherapy.
That distinction matters for clinicians and patients. In advanced HR-positive breast cancer, meaningful benefit is not always captured by OS alone, especially when post-progression therapies are active and unevenly distributed between arms.
Clinical Takeaway
Final OS results from CAPItello-291 showed no statistically significant overall survival improvement with capivasertib plus fulvestrant. In the PIK3CA/AKT1/PTEN-altered population, there was a numerical OS trend favoring capivasertib, but the analysis was underpowered and did not meet statistical significance.
The clinically important signal remains in disease control: capivasertib plus fulvestrant improved PFS2 and delayed time to first subsequent chemotherapy in both the altered and overall populations. These findings support the regimen as a targeted endocrine-based option that can extend treatment benefit and delay chemotherapy after progression on aromatase inhibitor-based therapy.


