A new genomic analysis provides important insight into why some patients with hormone receptor-positive, HER2-negative breast cancer progress earlier than expected on CDK4/6 inhibitor-based therapy. Tumors arising in patients with germline BRCA2 pathogenic variants appear especially prone to develop acquired RB1 loss-of-function alterations, a recognized mechanism of resistance to CDK4/6 inhibition. This resistance pattern does not appear random. It reflects a combination of baseline genomic vulnerability and ongoing homologous recombination deficiency, raising the possibility that PARP inhibition may deserve earlier consideration in this subgroup (Safonov et al., 2026).

Read About Breast Cancer on OncoDaily
Why This Matters In Breast Cancer
CDK4/6 inhibitors combined with endocrine therapy remain a cornerstone of treatment for metastatic HR-positive, HER2-negative breast cancer. Yet benefit is not uniform across all tumors. Some cancers respond for prolonged periods, while others progress far sooner than expected despite guideline-concordant treatment.
In this setting, germline BRCA2 status may help explain a distinct route of therapeutic resistance. Rather than serving only as a marker of inherited cancer risk and sensitivity to PARP inhibitors, BRCA2 also appears to shape how resistant clones emerge under CDK4/6 inhibitor pressure. That concept adds a new layer to treatment sequencing in metastatic breast cancer and suggests that inherited biology may influence not only which drugs work, but also how tumors evolve when they stop working (Safonov et al., 2026).
A Large Clinicogenomic Dataset
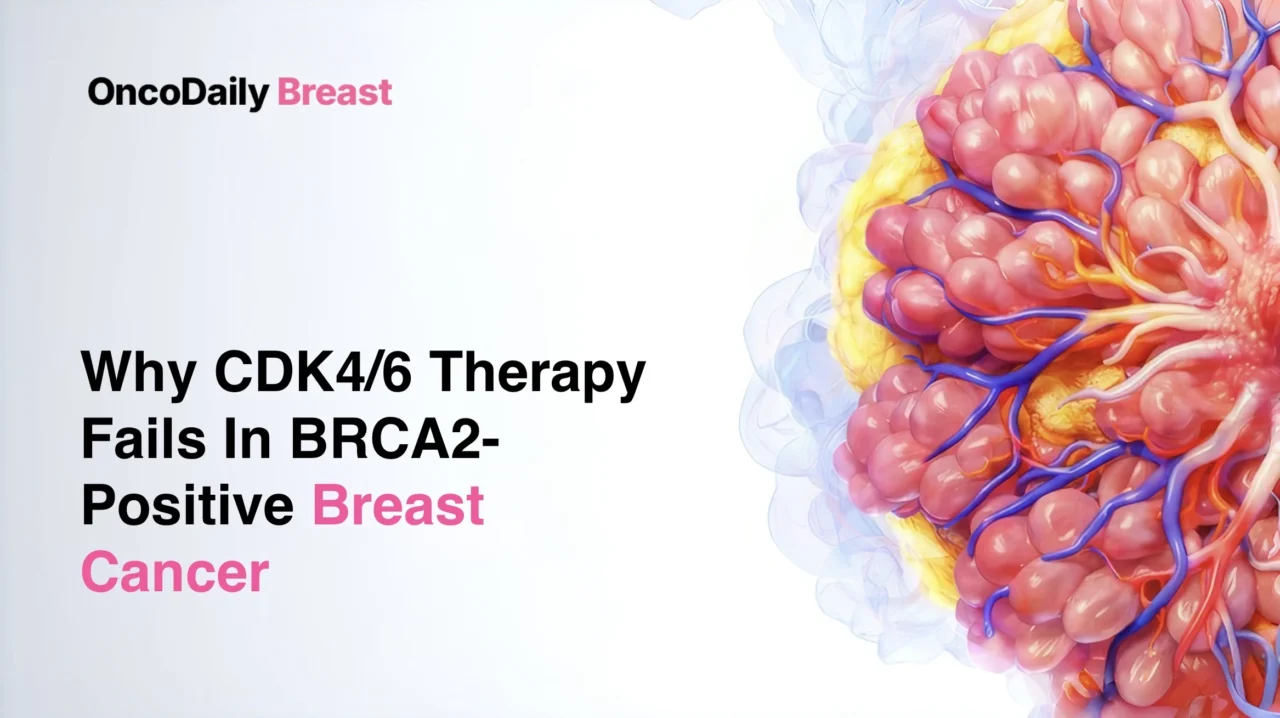
The analysis included 6,927 tumors from 5,881 patients with breast cancer. Tumor tissue and matched blood underwent genomic profiling, allowing assessment of both somatic and germline alterations. Among the germline findings, BRCA2 pathogenic variants were identified in 2.9% of patients, while BRCA1 variants were present in 2.6%, CHEK2 in 1.6%, ATM in 1.1%, and PALB2 in 0.6% (Safonov et al., 2026).
The clinical profile of gBRCA2-associated disease was notable. Most gBRCA2 tumors, 75.2%, were HR-positive and HER2-negative, placing them directly within the population most commonly treated with CDK4/6 inhibitors. In addition, 75.8% of gBRCA2 tumors demonstrated biallelic BRCA2 inactivation, supporting the biological significance of BRCA2 loss in a large proportion of these cancers (Safonov et al., 2026).
RB1 Emerged As A Central Alteration
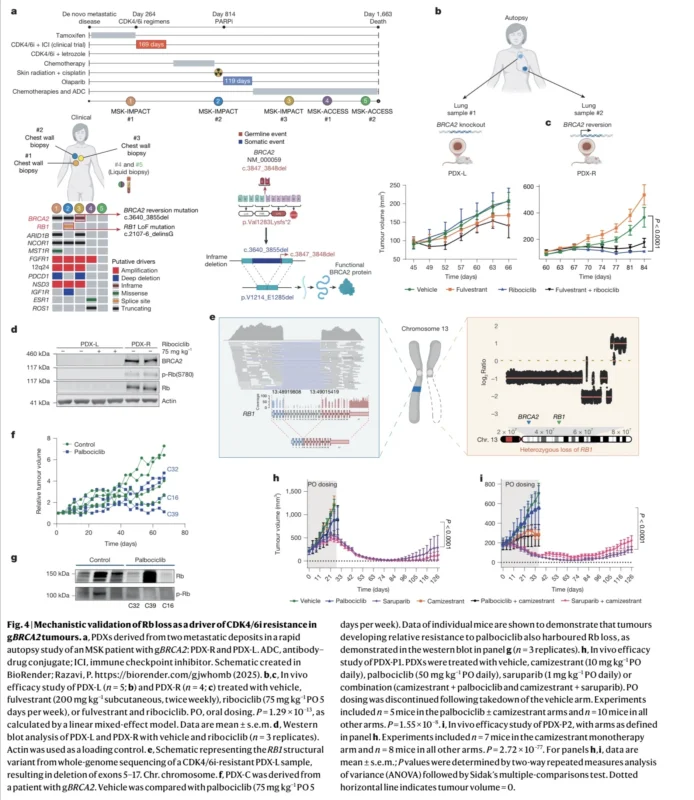
One of the most clinically important genomic signals involved RB1, a key tumor suppressor required for CDK4/6 inhibitor activity. In gBRCA2-driven breast cancers, RB1 somatic variants were significantly enriched. This enrichment became even stronger in the HR-positive/HER2-negative subgroup, where RB1 was the most significantly enriched somatic alteration, with an odds ratio of 5.66. Enrichment of MYC and AURKA amplifications was also observed, both changes previously associated with resistance to CDK4/6 inhibition. By contrast, PIK3CA alterations were more frequent in germline wild-type tumors than in gBRCA2-associated cancers (Safonov et al., 2026).
This pattern immediately suggested that BRCA2-associated HR-positive disease may behave differently under treatment pressure. It also raised a more specific therapeutic concern: if RB1 is already genomically vulnerable in these tumors, then exposure to CDK4/6 inhibition may create especially strong selective pressure for complete RB1 loss.
Shorter Progression-Free Survival On CDK4/6 Inhibitor Therapy
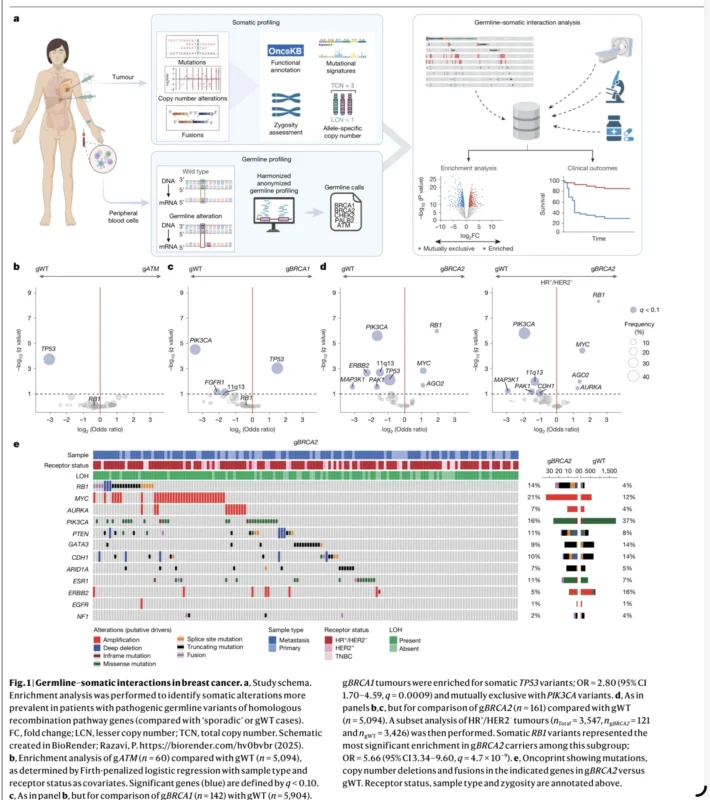
In metastatic HR-positive, HER2-negative breast cancer treated with first-line CDK4/6 inhibitor plus endocrine therapy, germline BRCA2 pathogenic variants were associated with significantly shorter progression-free survival. Median PFS was 9.0 months in gBRCA2 carriers versus 15.6 months in germline wild-type disease, with a multivariable hazard ratio of 2.17. Across treatment lines, the difference remained significant, with a hazard ratio of 1.97 (Safonov et al., 2026).
This pattern was also validated externally in a separate clinicogenomic cohort of 2,158 patients treated with first-line CDK4/6 inhibitor plus endocrine therapy. Median PFS was again shorter in gBRCA2 carriers, 12.0 months compared with 18.3 months, with a hazard ratio of 1.83 (Safonov et al., 2026).
The clinical signal appears specific rather than universal. Germline BRCA2 status did not significantly worsen outcomes across most other systemic treatment classes and did not significantly affect overall survival from the start of first-line CDK4/6 inhibitor plus endocrine therapy. This makes the observation more precise. The issue is not simply aggressive biology in general, but a particular mismatch between this tumor subtype and CDK4/6 inhibitor dependence (Safonov et al., 2026).
PARP Inhibition Performed Better Than The Prior CDK4/6 Regimen
The treatment sequencing analysis was particularly provocative. Among 41 patients with HR-positive/HER2-negative metastatic breast cancer who received a PARP inhibitor after progression on CDK4/6 inhibitor therapy, outcomes with PARP inhibition were generally better than outcomes on the prior CDK4/6 inhibitor regimen, even though PARP inhibitors were introduced later, at a median treatment line of 3.
Progression-free survival on PARP inhibition was significantly improved compared with the earlier CDK4/6 inhibitor course, with a hazard ratio of 0.38. In addition, 73.2% of patients experienced a longer PFS on PARP inhibition than on frontline CDK4/6 inhibitor therapy. Among evaluable cases, the partial or complete response rate reached 84.2% with PARP inhibitors, compared with 39.5% on the preceding CDK4/6 inhibitor plus endocrine regimen (Safonov et al., 2026).
These observations are retrospective, but they strongly support the therapeutic logic behind the biology. If gBRCA2-associated tumors are predisposed to fail CDK4/6 inhibition through a predictable RB1 loss trajectory driven by homologous recombination deficiency, then earlier use of a DNA repair-targeted strategy may offer greater benefit before resistant RB1-deficient clones emerge.
Resistance Through RB1 Loss Is Not Random
Paired pre-treatment and post-treatment samples offered a direct view of how resistance developed. In a cohort of 546 patients with samples collected before and after CDK4/6 inhibitor therapy, acquired RB1 loss-of-function alterations occurred in 28.6% of gBRCA2 tumors compared with 7.1% of gBRCA2 wild-type tumors, corresponding to an odds ratio of 5.17 (Safonov et al., 2026).
This difference reflects a biologically plausible mechanism. BRCA2 and RB1 are located on the same chromosomal arm, 13q. In tumors driven by germline BRCA2 pathogenic variants, loss of heterozygosity affecting BRCA2 often extends to RB1 as well. As a result, many tumors enter therapy already one step closer to complete RB1 inactivation. In an external cohort of primary gBRCA2-associated breast cancers, 82.6% demonstrated concurrent BRCA2 and RB1 loss of heterozygosity, reinforcing how frequently this genomic configuration occurs (Safonov et al., 2026).
That is a key point for breast oncology. Resistance here is not merely acquired in response to treatment. In many cases, the groundwork is already present before therapy begins.
RB1 LOH And Hemizygosity Identify A More Fragile State
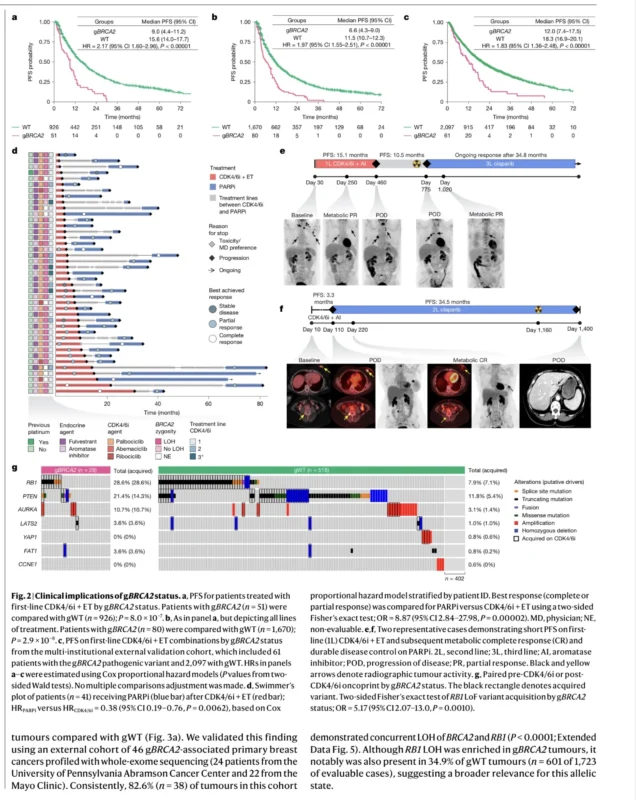
RB1 loss of heterozygosity carried prognostic significance even beyond BRCA2 status alone. In 547 patients with HR-positive/HER2-negative metastatic breast cancer treated with first-line CDK4/6 inhibitors and with evaluable baseline LOH data, RB1 LOH was present in 47.8% and was associated with shorter progression-free survival and worse overall survival. The hazard ratio was 1.53 for PFS and 1.31 for OS (Safonov et al., 2026).
A similar pattern was observed in baseline circulating tumor DNA from PALOMA-3, where RB1 LOH was identified in 7.3% of samples and was associated with shorter progression-free survival and overall survival, with hazard ratios of 2.20 and 3.28, respectively.
The analysis then refined the picture further by focusing on RB1 hemizygosity, meaning true heterozygous loss with only one remaining functional RB1 allele. This allelic configuration emerged as the state most likely to progress into complete RB1 inactivation under treatment pressure. In paired pre- and post-treatment samples, acquired RB1 loss-of-function events occurred almost exclusively in tumors with baseline RB1 heterozygous loss. Total RB1 loss-of-function events were identified in 15.2% of these tumors, and 14.5% represented newly acquired events during therapy.
This concept is especially important because it shifts RB1 from being a static marker to a dynamic evolutionary predictor. Hemizygosity defines a vulnerable intermediate state, one in which only a single additional genomic event is needed for full therapeutic escape.
Homologous Recombination Deficiency Fuels The Second Hit
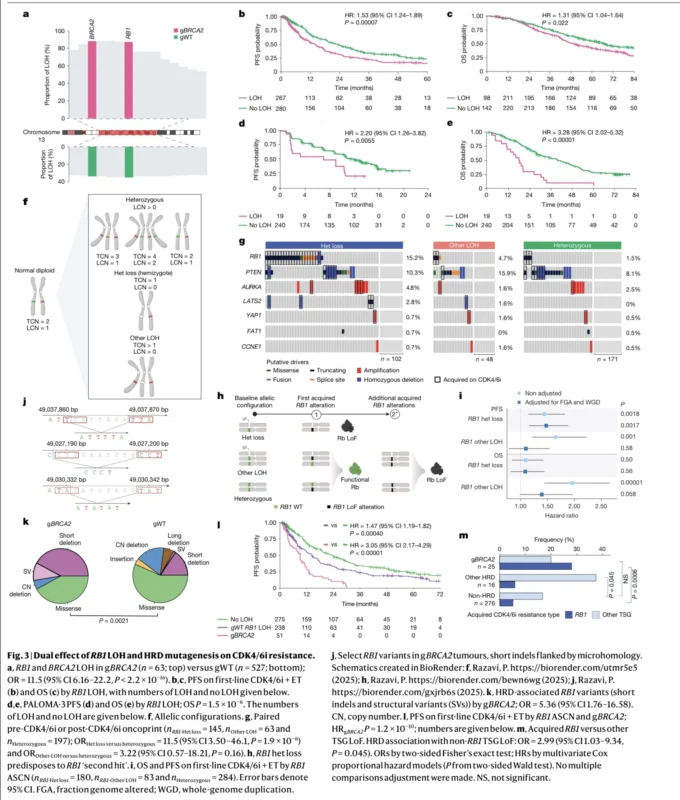
The second force driving this resistance pattern is homologous recombination deficiency itself. In gBRCA2-associated tumors, emerging RB1 variants frequently showed short deletions flanked by microhomology, a pattern consistent with microhomology-mediated end joining, an error-prone DNA repair process that becomes more active in HRD-driven cancers. Enrichment of short indels and structural variants involving RB1 further supports this biology (Safonov et al., 2026).
The clinical consequence was substantial. While RB1 LOH in germline wild-type disease was associated with shorter PFS, with a hazard ratio of 1.47, the effect became much stronger in gBRCA2-associated tumors, where RB1 LOH was associated with a hazard ratio of 3.05 for shorter progression-free survival.
This suggests a two-hit resistance model in which baseline RB1 allelic vulnerability combines with HRD-driven mutational pressure to accelerate emergence of RB1-deficient clones under CDK4/6 inhibitor therapy. In practical terms, the tumor is both structurally primed and biologically equipped to escape.
What This Means
These findings frame gBRCA2-associated HR-positive/HER2-negative breast cancer as a biologically distinct therapeutic subgroup. The challenge is not only inherited susceptibility or PARP sensitivity. It is also a predictable and clinically relevant route of resistance to one of the most commonly used treatment backbones in metastatic breast cancer.
That insight matters because it affects how treatment sequencing may eventually be approached. A tumor with germline BRCA2 alteration, RB1 loss of heterozygosity, and persistent homologous recombination deficiency may be less likely to achieve durable benefit from frontline CDK4/6 inhibition and more likely to evolve through RB1 loss. In that context, earlier use of PARP inhibition becomes a rational strategy worthy of prospective testing (Safonov et al., 2026).
The broader message is equally important. In breast cancer, inherited and somatic genomics do not operate in separate worlds. They intersect. Germline background can shape tumor structure, influence selective pressure, and help determine the exact mechanism by which resistance emerges under therapy. That is a powerful example of how precision oncology is moving beyond static biomarkers toward predicting tumor evolution itself.
Full article is available in Nature