Innovative immunotherapy approaches continue to reshape the oncology landscape, particularly in areas where conventional therapies have limited effectiveness. One such emerging strategy involves invariant natural killer T (iNKT) cell therapies, a unique form of immune cell therapy designed to harness both innate and adaptive immune responses. On March 10, 2026, MiNK Therapeutics announced a strategic collaboration with C‑Further to develop a PRAME-targeted iNKT cell therapy for pediatric cancers.
The collaboration represents one of the first programs selected by the C-Further consortium since its launch and reflects a growing global effort to accelerate the development of novel, well-tolerated immunotherapies for children with cancer. The initiative is supported by an international network of organizations including Cancer Research Horizons, LifeArc, and Great Ormond Street Hospital Charity.
This partnership highlights a broader shift in pediatric oncology toward targeted cellular immunotherapies designed specifically for childhood cancers rather than relying on treatments developed primarily for adults.
Addressing an Urgent Need in Pediatric Oncology
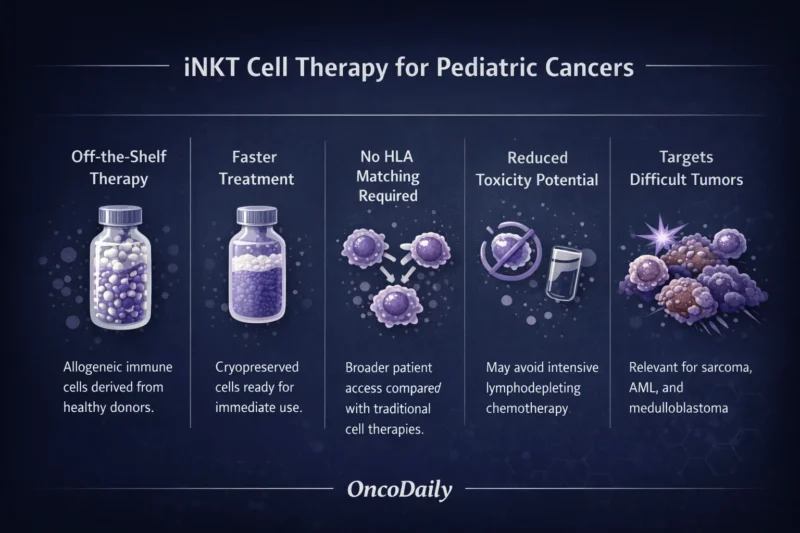
Despite significant progress in childhood cancer survival over the past several decades, many pediatric malignancies remain extremely difficult to treat once they relapse or become resistant to therapy. Tumors such as sarcomas, acute myeloid leukemia (AML), and medulloblastoma often lack well-defined therapeutic targets, limiting treatment options for patients who fail first-line therapies.
The collaboration between MiNK Therapeutics and C-Further aims to address this unmet need by developing a PRAME-targeted iNKT cell therapy that could provide a new immunotherapy option for children with aggressive cancers.
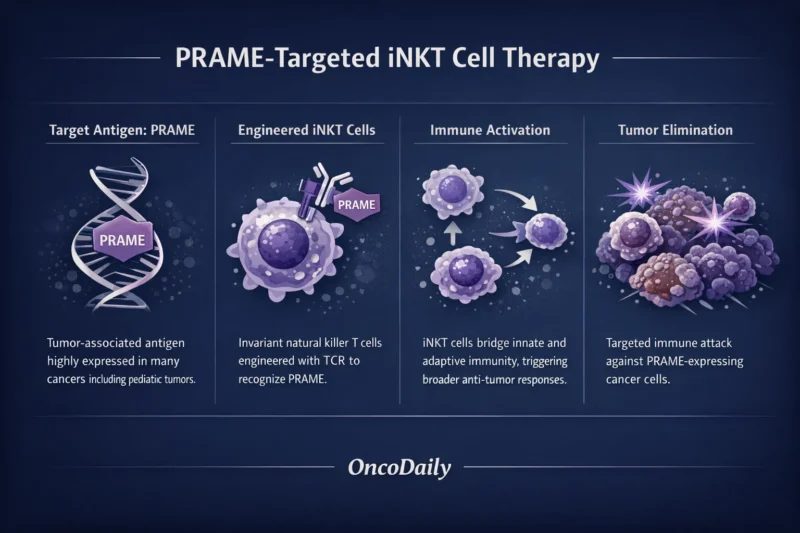
The target antigen in this program, PRAME (Preferentially Expressed Antigen in Melanoma), is a tumor-associated protein expressed across a broad range of malignancies while remaining largely absent in normal tissues. Because of this selective expression pattern, PRAME has emerged as a promising therapeutic target in oncology, particularly in tumors where other actionable molecular markers are scarce.
Importantly, PRAME expression has been documented in several pediatric cancers, including:
- Pediatric sarcomas
- Acute myeloid leukemia
- Medulloblastoma
These malignancies often carry poor outcomes after relapse, highlighting the need for innovative treatment strategies capable of producing durable responses.
The Science Behind iNKT Cell Therapy
The therapeutic platform underlying this collaboration is based on invariant natural killer T cells, a specialized immune cell population with unique biological properties.
Unlike conventional T cells or natural killer cells alone, iNKT cells act as a bridge between innate and adaptive immunity. These cells can rapidly recognize tumor cells and trigger a broader immune response by activating other immune components within the tumor microenvironment.
This dual function gives iNKT cells several potential advantages in cancer therapy:
First, they are capable of direct cytotoxic activity against tumor cells. Second, they can stimulate other immune cells—including dendritic cells, natural killer cells, and conventional T cells—thereby amplifying the anti-tumor immune response.
MiNK Therapeutics has developed a proprietary platform designed to exploit this biology by engineering iNKT cells to recognize specific tumor antigens through T-cell receptor (TCR) modification.
In the new collaboration, these engineered cells will specifically target the PRAME antigen, enabling precise recognition of cancer cells expressing this marker.
Engineering PRAME-Targeted iNKT Cells
The new therapeutic program will combine two important components of modern immunotherapy.
The first is TCR engineering, which allows immune cells to recognize specific tumor antigens with high precision. The second is the iNKT cell platform, which provides a powerful immune effector capable of orchestrating complex anti-tumor responses.
Together, these technologies are expected to produce a therapy that not only targets PRAME-expressing tumor cells but also enhances immune activation within the tumor microenvironment.
Researchers hope that this strategy will produce a more durable and coordinated immune attack against cancer, improving outcomes for patients with tumors that are otherwise difficult to treat.
Advantages of an Off-the-Shelf Cell Therapy
One of the most important features of the MiNK iNKT platform is that it is designed as an allogeneic, off-the-shelf therapy.
Traditional cell therapies, such as CAR-T treatments, often require collecting immune cells from each individual patient, modifying them in specialized laboratories, and then reinfusing them into the patient. This process can take weeks, which may be too long for patients with rapidly progressing cancers.
In contrast, MiNK’s iNKT cells are derived from healthy donor cells, which can be manufactured in advance, cryopreserved, and stored until needed.
This approach offers several potential advantages.
Because the therapy does not require patient-specific manufacturing, treatment could be delivered much faster. Additionally, the therapy does not require strict HLA matching between donor and patient, which simplifies clinical use.
Another potential advantage is that the therapy may be administered without the need for toxic lymphodepleting chemotherapy, which is often required before CAR-T cell therapy. Avoiding this step could improve tolerability, particularly for pediatric patients who are more vulnerable to treatment-related toxicity.
These characteristics could ultimately enable broader access and faster treatment delivery, both of which are crucial in pediatric oncology.
Read About iNKT Cell Therapy on OncoDaily
Financial and Strategic Structure of the Collaboration
The collaboration is structured as a non-dilutive funding agreement, meaning the program will receive financial support without requiring MiNK to issue new company shares.
Under the agreement, the PRAME-TCR-iNKT program will receive approximately $1.1 million in funding to support IND-enabling development, the stage of research required before a therapy can enter human clinical trials.
The funding will support key milestones including:
- Preclinical candidate selection
- Translational research studies
- Safety and efficacy testing in laboratory models
Payments will be tied to specific scientific milestones as the program progresses toward clinical development.
In addition to this development funding, the agreement includes double-digit participation in downstream commercial revenues, reflecting the value of MiNK’s proprietary iNKT platform.
Importantly, the collaboration is non-exclusive, allowing MiNK Therapeutics to continue developing additional therapies based on its iNKT technology across multiple cancer indications.
Academic and Translational Research Collaboration
A central component of the program involves collaboration with researchers at the University of Southampton.
Investigators led by Ali Roghanian and Salah Mansour will conduct independent preclinical studies to evaluate the therapeutic candidates.
These studies will examine several key parameters, including:
- Anti-tumor activity
- Persistence of engineered cells
- Safety profiles in relevant tumor models
Researchers will use patient-derived tumor systems to simulate real-world disease biology and identify the most promising therapeutic candidate.
The goal of this research phase is to nominate a single lead candidate for advancement into first-in-human clinical trials in children.
The C-Further Consortium Model
The collaboration is part of a broader initiative led by the C-Further consortium to accelerate pediatric cancer drug development.
Historically, many therapies used in pediatric oncology have been adapted from adult cancer treatments, sometimes leading to toxicities that can affect long-term development and quality of life.
C-Further aims to shift this paradigm by supporting therapies specifically designed for children.
The consortium brings together drug discovery experts, academic researchers, clinicians, and industry partners to create a collaborative ecosystem capable of moving promising therapies from the laboratory to the clinic more efficiently.
By combining industry-grade drug development infrastructure with academic innovation, the consortium hopes to accelerate the translation of cutting-edge research into real therapies for children with cancer.
Expanding the iNKT Platform
The PRAME-targeted program is part of MiNK Therapeutics’ broader strategy to apply its iNKT platform across multiple tumor antigens and disease indications.
The company’s lead candidate, agenT-797, is currently being evaluated in clinical development for several conditions including:
- Graft-versus-host disease (GvHD)
- Solid tumors
- Severe pulmonary inflammation
The addition of PRAME-targeted TCR engineering represents the next step in expanding the platform into precision cell therapies for oncology.
By targeting well-validated tumor antigens such as PRAME, MiNK hopes to develop therapies capable of producing durable immune responses while maintaining favorable safety profiles.
Looking Ahead: Toward First-in-Human Studies
The immediate goal of the collaboration is to advance the program through preclinical development and identify the strongest candidate for clinical testing.
If successful, the therapy could move into first-in-human trials in pediatric patients, marking an important milestone in the development of next-generation cell therapies for childhood cancers.
For children facing aggressive malignancies with limited treatment options, such innovations could offer new hope.
More broadly, the program reflects a growing recognition that pediatric oncology requires dedicated drug development strategies, rather than simply adapting therapies designed for adult cancers.
Through collaborations like this one, researchers and industry partners are working to build a new generation of treatments tailored specifically to the needs of young patients.
The Future of Pediatric Immunotherapy
The partnership between MiNK Therapeutics and C-Further represents a convergence of several major trends in oncology.
These include the rapid rise of cell therapy technologies, the growing interest in tumor-specific antigens such as PRAME, and the increasing focus on developing therapies specifically for pediatric cancers.
While significant research remains ahead, the collaboration demonstrates how innovative platforms and global partnerships can accelerate progress in areas of high unmet medical need.
If successful, PRAME-targeted iNKT therapy could become an important addition to the expanding arsenal of immunotherapies designed to transform the treatment landscape for children with cancer.
Written by Armen Gevorgyan, MD


