Recurrent or metastatic cervical cancer remains a major therapeutic challenge, with long-term survival still poor despite advances in systemic treatment. Platinum-based chemotherapy, bevacizumab, and immune checkpoint inhibitors have improved outcomes in the first-line setting, but many patients ultimately experience progression and require additional effective options.
Tisotumab vedotin, a tissue factor–directed antibody–drug conjugate, has already established activity as monotherapy in previously treated recurrent or metastatic cervical cancer. The next important question has been whether tisotumab vedotin can be successfully combined with other active agents to improve durability of response and extend survival in both first-line and later-line settings.
The updated 5-year results from the innovaTV 205/ENGOT-cx8/GOG-3024 study help answer that question by evaluating tisotumab vedotin in combination with carboplatin, pembrolizumab, and, in one arm, bevacizumab.
Study Objective
The aim of this phase 1b/2 study was to assess the long-term efficacy and safety of tisotumab vedotin–based combinations in patients with recurrent or metastatic cervical cancer treated in the first-line and second-line or later settings.
The study evaluated four dose-expansion cohorts:
- first-line tisotumab vedotin plus carboplatin,
- first-line tisotumab vedotin plus pembrolizumab,
- second-/third-line tisotumab vedotin plus pembrolizumab,
- and first-line tisotumab vedotin plus carboplatin and pembrolizumab, with or without bevacizumab.
Study Design and Patient Population
innovaTV 205 was a multicenter, open-label phase 1b/2 trial that enrolled patients with recurrent or stage IVB cervical cancer, including squamous, adenosquamous, and adenocarcinoma histologies.
Patients in the first-line arms had not received prior systemic therapy for recurrent or metastatic disease, whereas patients in the later-line pembrolizumab combination arm had progressed during or after one or two prior systemic regimens.
Importantly, enrollment was not restricted by PD-L1 status or tissue factor expression, although most patients across the expansion cohorts ultimately had PD-L1–positive disease.
At the October 15, 2025 data cutoff, 139 patients had been enrolled across the four dose-expansion arms.
Efficacy Across the Doublet Regimens
First-Line Tisotumab Vedotin Plus Carboplatin
This combination produced a confirmed objective response rate of 54.5%, including complete responses in 15.2% of patients. Median duration of response was 8.6 months, median progression-free survival was 6.9 months, and median overall survival reached 25.5 months.
These findings suggest that combining tisotumab vedotin with platinum chemotherapy has meaningful first-line activity and can deliver survival outcomes extending beyond two years in some patients.
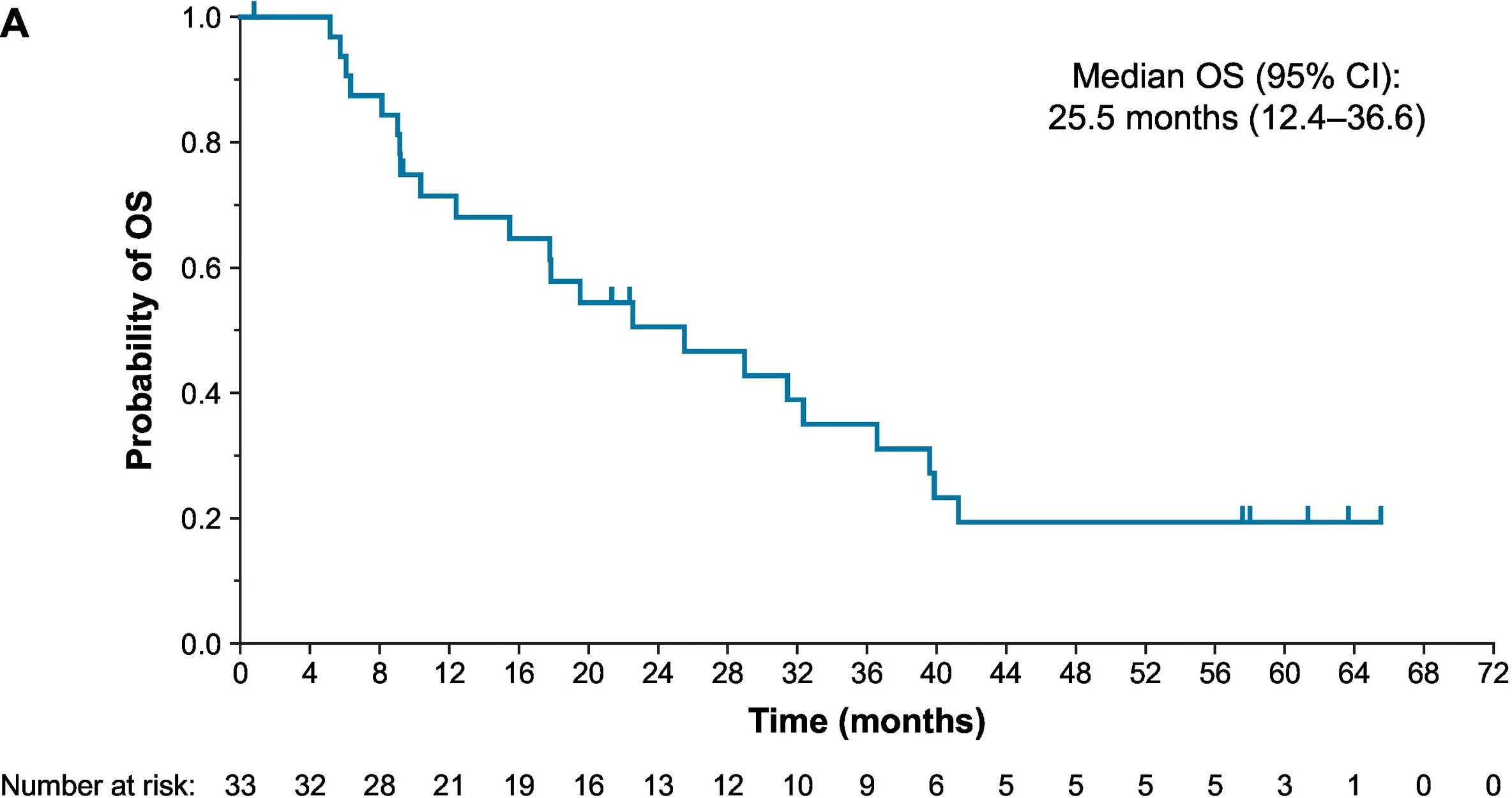
First-Line Tisotumab Vedotin Plus Pembrolizumab
The first-line pembrolizumab doublet achieved a confirmed objective response rate of 40.6%, including a notably high complete response rate of 21.9%. Median duration of response was not reached, indicating durable responses in a subset of patients, while median progression-free survival was 5.3 months and median overall survival was 30.7 months.
This is one of the most interesting signals in the study. Although the objective response rate was numerically lower than with the carboplatin doublet or the triplet/quadruplet regimen, the durability of benefit and overall survival appear particularly encouraging.
Second-/Third-Line Tisotumab Vedotin Plus Pembrolizumab
In previously treated patients, the pembrolizumab doublet produced a confirmed objective response rate of 35.3%, with a median duration of response of 18.2 months, median progression-free survival of 5.6 months, and median overall survival of 15.3 months.
These later-line results remain clinically meaningful. In a disease setting where durable activity is difficult to achieve, the prolonged duration of response stands out and supports the role of tisotumab vedotin as a strong combination partner beyond first-line therapy.
The Most Intensive Regimen: TV + Carboplatin + Pembrolizumab ± Bevacizumab
The most active regimen in the study was the first-line triplet/quadruplet approach combining tisotumab vedotin, carboplatin, pembrolizumab, and optional bevacizumab.
This arm achieved a confirmed objective response rate of 65.8%, including 18.4% complete responses, with a median duration of response of 13.3 months. Median progression-free survival reached 10.6 months, and median overall survival was 28.0 months. At two years, the estimated progression-free survival rate was 29.3%, while the overall survival rate was 62.4%.
These are impressive findings for recurrent or metastatic cervical cancer and suggest that TV-based multidrug regimens may have meaningful front-line potential, particularly for patients able to tolerate a more intensive strategy.
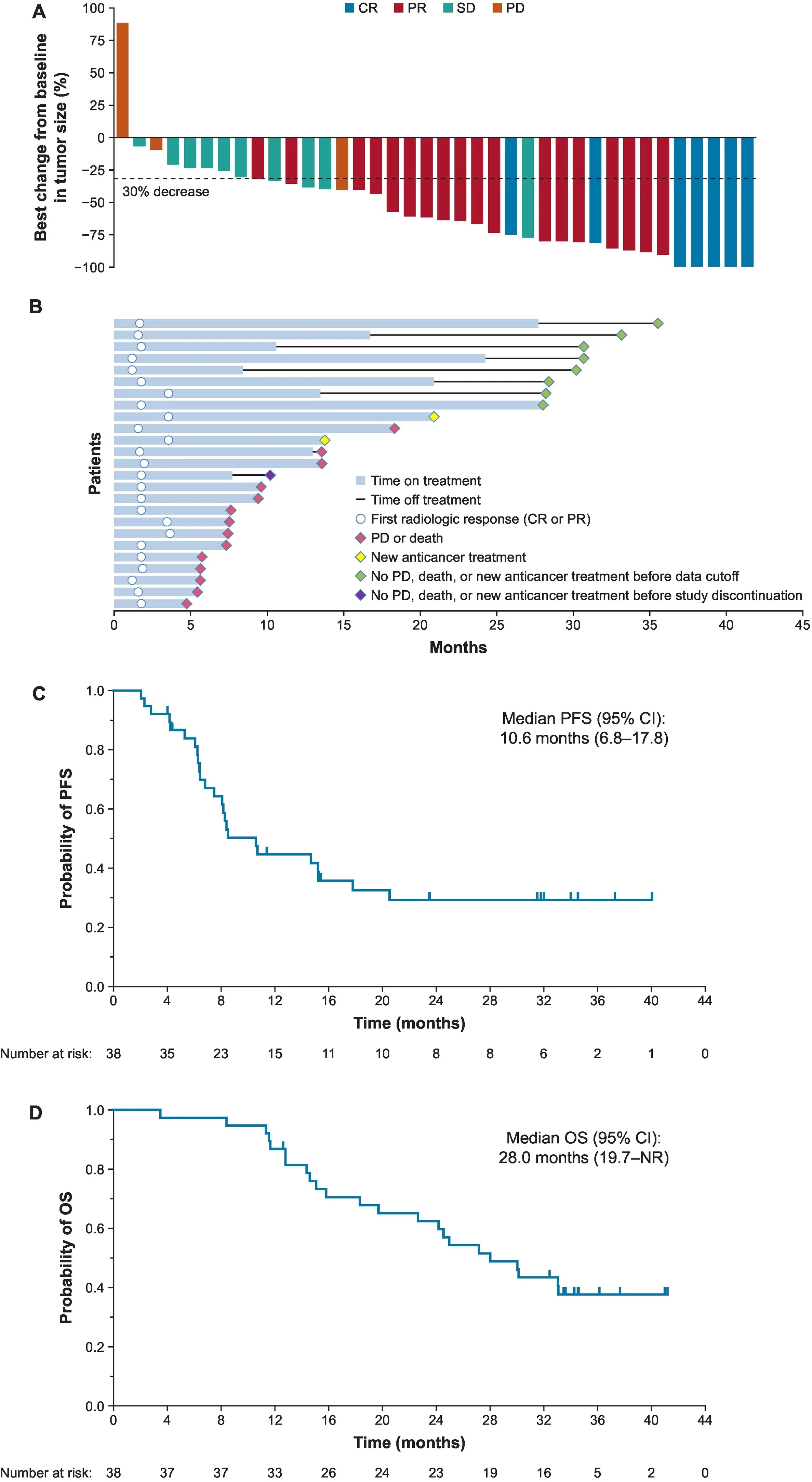
Why These Results Matter
The main significance of this study lies in the durability of benefit. After more than five years of follow-up in the doublet cohorts, responses remained consistent with earlier analyses, and survival outcomes continued to look encouraging.
This matters because recurrent or metastatic cervical cancer remains a disease in which durable long-term control is uncommon. The study suggests that tisotumab vedotin is not only active as a single agent, but also flexible enough to be incorporated into doublet and triplet/quadruplet regimens without unexpected new safety concerns.
The data also reinforce the idea that antibody–drug conjugates are moving beyond salvage therapy and increasingly being explored as earlier-line combination partners.
Safety Profile
Across all treatment arms, adverse events were common, as expected in this patient population and with combination regimens. The most frequently reported toxicities included nausea, diarrhea, anemia, alopecia, and fatigue.
The known adverse events of special interest with tisotumab vedotin were again observed:
ocular toxicity, peripheral neuropathy, and bleeding events.
Ocular events occurred in more than half of patients in every cohort, but most improved or resolved over time with mitigation strategies and ophthalmologic monitoring. Peripheral neuropathy was also common and appeared particularly relevant in the more intensive arm, where it contributed to treatment discontinuation in a meaningful number of patients. Bleeding events were frequent overall, though high-grade bleeding remained less common.
The most intensive first-line regimen had the highest toxicity burden:
grade 3 or higher treatment-related adverse events occurred in 86.8% of patients, and 55.3% discontinued tisotumab vedotin in that arm. Even so, the safety findings were broadly consistent with the known toxicity profiles of the individual agents, and no new safety signals emerged with longer follow-up.
Clinical Interpretation
Several clinically relevant themes emerge from this study.
- First, tisotumab vedotin appears to be a versatile partner in cervical cancer therapy. It showed activity with platinum chemotherapy, with pembrolizumab, and in a more aggressive multi-agent regimen that included both.
- Second, durability appears especially important here. Some response rates were already promising in earlier analyses, but the long follow-up now confirms that a proportion of patients can derive prolonged clinical benefit.
- Third, the triplet/quadruplet regimen seems particularly active in the first-line setting, but at the cost of increased toxicity and discontinuation. This raises an important future question: which patients are most likely to benefit from more intensive TV-based combinations, and which may be better served by a doublet strategy.
Finally, cross-trial comparisons must remain cautious, but the reported overall survival outcomes compare favorably with historical benchmarks from major cervical cancer studies, including chemotherapy-bevacizumab and pembrolizumab-based regimens.
Limitations
This remains a non-randomized expansion analysis, so interpretation must be careful. The cohorts were relatively small, and direct comparisons between arms are not appropriate.
In addition, most enrolled patients had PD-L1–positive disease, which limits conclusions about efficacy in PD-L1–negative populations. The optional use of bevacizumab in the most intensive arm also adds some heterogeneity to interpretation.
Still, the long duration of follow-up adds unusual strength to the findings, especially in a disease where durable outcomes are rarely documented over such an extended time horizon.
Conclusion
The 5-year results from innovaTV 205 show that tisotumab vedotin–based combinations can deliver durable antitumor activity in recurrent or metastatic cervical cancer across both first-line and previously treated settings.
The doublet regimens with carboplatin or pembrolizumab demonstrated sustained efficacy and encouraging long-term survival, while the first-line triplet/quadruplet combination with carboplatin, pembrolizumab, and optional bevacizumab produced the highest response rates and meaningful survival outcomes, albeit with greater toxicity.
Overall, these data support tisotumab vedotin as a promising and adaptable combination partner in cervical cancer and strengthen the rationale for further development of ADC-based combination strategies in earlier lines of treatment.
Cervical Cancer: Symptoms, Causes, Stages, Diagnosis and Treatment
Giuseppe Caruso, Gynecologic oncologist consultant at IEO European Institute of Oncology IRCCS (Milan, Italy):
” Very happy to see ADCs finally moving earlier in cervical cancer — because in real life, using them late is often too late!!!
Recurrent/metastatic cervical cancer is still an unmet need. In daily practice, when we give Tisotumab Vedotin (TV) after immunotherapy, patients are often already compromised — fistulas, nephrostomies, frailty. That’s not when you unlock the full potential of an ADC.
The 5-year innovaTV 205 data clearly show that TV-based combinations (triplet/quadruplet regimens) are active and durable, especially in 1L, with response rates up to ~66% and OS approaching 2+ years.
If we believe in these drugs, we should use them earlier — not as a last resort. This shift toward early lines is not just interesting. It’s necessary.”
You Can Read Full Article Here


