Tecartus (brexucabtagene autoleucel), a CD19-directed CAR T-cell therapy developed by Kite Pharmaceuticals, has received full approval from the U.S. Food and Drug Administration (FDA) for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL). This decision marks a continued shift toward cellular immunotherapy in hematologic malignancies and emphasizes the lasting clinical value of Tecartus for patients with limited therapeutic alternatives and in earlier lines of therapy.
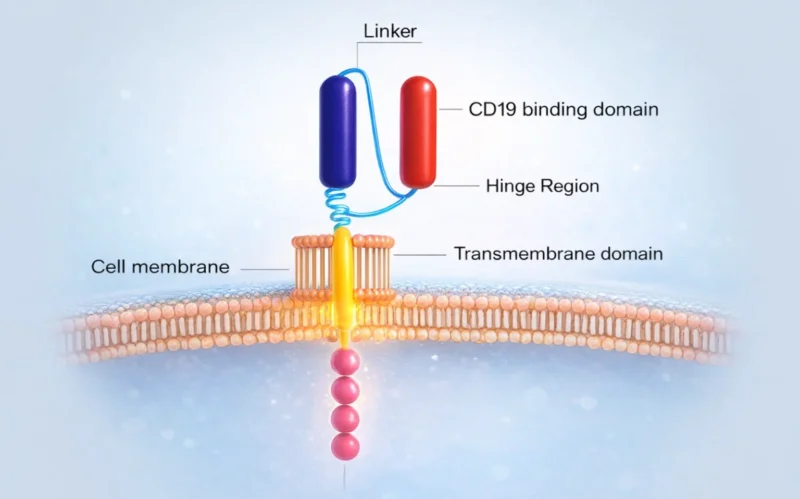
Tecartus: Structure and Mechanism of Action
Tecartus is a CD19-directed genetically modified autologous T cell immunotherapy. indicated for the treatment of:
- Adult patients with relapsed or refractory mantle cell lymphoma (MCL)
- Adult patients with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL)
To manufacture Tecartus, a patient’s own T cells are harvested and genetically modified ex vivo by retroviral transduction to express a chimeric antigen receptor comprising a murine anti-CD19 single-chain variable fragment (scFv) linked to CD28 and CD3-zeta co-stimulatory domains. The anti-CD19 CAR T cells are expanded and infused back into the patient. For MCL, the target dose is 2 × 10⁶ cells/kg, with a maximum dose of 2 × 10⁸ .
Following the engagement with CD19-expressing target cells, the co-stimulatory domains activate downstream signaling cascades that lead to T cell activation, proliferation, acquisition of effector functions, and secretion of inflammatory cytokines and chemokines. These events lead to the elimination of CD19-expressing cells. Following Tecartus infusion, serum cytokine levels peak within 8 days, with the majority returning to baseline by Week 4.
Safety Profile and Adverse Events
As with other CAR T-cell therapies, treatment with Tecartus is associated with specific immune-mediated toxicities:
- Cytokine Release Syndrome (CRS) – occurred in 93% of patients with MCL, including ≥ Grade 3 CRS in 12%
- Neurologic toxicities (including ICANS) – occurred in 80% of patients with MCL, including ≥ Grade 3 in 33%
Most adverse events were manageable with established protocols, including tocilizumab and corticosteroids.
Other commonly observed adverse events include:
- Severe infections – occurred in 63% of patients with MCL, including Grade ≥3 events in 33%, warranting the use of prophylactic antimicrobials in accordance with local guidelines.
- Prolonged cytopenias – among patients with MCL Grade ≥3 cytopenias unresolved by Day 30 occurred in 55% , beyond Day 60 in 11%, therefore, close monitoring of blood counts is recommended following Tecartus infusion.
- B-cell aplasia and hypogammaglobulinemia – reported in 14% of patients with MCL, necessitating monitoring of immunoglobulin levels and management with infection precautions, antibiotic prophylaxis, and immunoglobulin replacement as appropriate.

You can also read: Bispecific Antibodies vs CAR-T in Multiple Myeloma: Mechanisms, Clinical Outcomes, and Key Considerations
Mantle Cell Lymphoma: A Need for Novel Therapies
Mantle cell lymphoma (MCL) is a subtype of non-Hodgkin lymphoma that arises from B lymphocytes in the mantle zone of lymphoid follicles. The characteristic genetic lesion, t(11;14)(q13;q32), leads to cyclin D1 overexpression, driving uncontrolled cell proliferation.
MCL accounts for 3-10% of all non-Hodgkin lymphomas and primarily affects adults around 70 years of age, with a strong male predominance (3:1). The disease commonly presents at an advanced stage and exhibits a propensity for relapse after treatment (with a median OS of 4-6 years).
Despite advances with BTK inhibitors and chemoimmunotherapy, patients with relapsed or refractory disease face poor outcomes (with median OS of 6 – 10 months following BTK inhibitor failure) underscoring the need for transformative approaches such as CAR T-cell therapy.
Early Development and Platform Validation of Tecartus
While not specific to MCL, Tecartus evolved from broader CAR T-cell development programs such as ZUMA-1 in diffuse large B-cell lymphoma, which established the CD19 CAR T-cell platform, characterized its safety profile (including CRS and ICANS), and demonstrated manufacturing feasibility.
In 2020, the FDA granted accelerated approval to Tecartus based on results from ZUMA-2 Cohort 1 (BTKi-exposed patients), marking the first CAR T-cell therapy approved for mantle cell lymphoma.
Between 2021 and 2023, continued follow-up from ZUMA-2 demonstrated durable responses, with sustained remissions extending beyond 2-3 years and a consistent, manageable safety profile, providing the confirmatory evidence required for full approval. Between 2023 and 2024, Cohort 3 evaluated Tecartus in BTKi-naïve patients, showing high response rates and durable remissions, supporting its use in earlier lines of therapy.
Clinical Evidence Supporting Full Approval
Full approval is based on updated data from the ZUMA-2 trial, a multicenter, open-label, single-arm phase II study evaluating a single infusion of Tecartus in adults with relapsed or refractory MCL previously treated with chemoimmunotherapy, anti-CD20 antibodies, and, in Cohort 1, a Bruton tyrosine kinase inhibitor.
In Cohort 1 (BTKi-exposed), 68 patients received TECARTUS, of whom 60 were efficacy-evaluable. Patients had a median age of 65 years, were heavily pretreated (median 3 prior lines), and predominantly had advanced-stage disease (83% stage IV). High-risk features included refractory disease (60%) and prior autologous HSCT (43%). Bridging therapy was administered in 35% of patients.
In Cohort 1, Tecartus demonstrated the following efficacy results:
- ORR of 87%
- CR rate of 62%.
- PR rate of 25%
The MDR was not reached, with ongoing responses observed up to 29.2+ months.
In Cohort 3 (BTKi-naïve), 86 patients were efficacy-evaluable. Patients had a median age of 64 years, with predominantly advanced-stage disease (65% stage IV) and a median of 1 prior line of therapy. The majority had relapsed disease (86%), and bridging therapy was administered in 36% of patients.
- ORR of 91%
- CR rate of 79%
- PR rate of 12%
The MDR was not reached, with ongoing responses observed up to 35.3+ months.
Why This Approval Matters?
Key implications for clinical practice and future treatment strategies include:
- Positions CAR T-cell therapy as a standard treatment option following BTK inhibitor failure
- Shifts treatment expectations toward potential long-term remission or cure
- Supports growing confidence in cellular immunotherapy in B-cell malignancies
The conversion from accelerated to full FDA approval reflects the maturation of efficacy data and the expansion of clinical evidence, including BTKi-naïve populations, confirming the long-term clinical benefit of Tecartus and solidifying its place in the treatment paradigm for R/R MCL.
Detailed information is available here.
Written by Susanna Mikayelyan, MD


