On May 15, 2026, the U.S. Food and Drug Administration (FDA) approved atezolizumab (Tecentriq, Genentech, Inc.) and atezolizumab and hyaluronidase-tqjs (Tecentriq Hybreza, Genentech, Inc.) as adjuvant treatments for adults with muscle-invasive bladder cancer (MIBC) after cystectomy who have circulating tumor DNA molecular residual disease (ctDNA MRD), as determined by an FDA-authorized test.
On the same day, the FDA also approved Signatera CDx (Natera, Inc.) as a companion diagnostic device to identify patients with MIBC after cystectomy who have ctDNA MRD and may be selected for adjuvant treatment with Tecentriq or Tecentriq Hybreza.
Background
Muscle-invasive bladder cancer is a clinically aggressive form of bladder cancer in which the tumor has grown into the muscular layer of the bladder wall. For many patients, treatment may include radical cystectomy with lymph node dissection. However, even after surgery, some patients remain at risk of disease recurrence.
In this approval, the key biomarker is ctDNA molecular residual disease, meaning that circulating tumor DNA remains detectable in the blood after cystectomy. This supports biomarker-based patient selection for adjuvant treatment, and the FDA approval reflects a biomarker-guided approach, selecting patients based on ctDNA MRD positivity after surgery.
Read more about Bladder Cancer on OncoDaily.
How Does Atezolizumab Work?
Atezolizumab is an immune checkpoint inhibitor that targets programmed death-ligand 1 (PD-L1), a protein involved in suppressing anti-tumor immune responses. Some tumor cells can use the PD-L1 pathway to evade immune detection and reduce T-cell activity.
By blocking PD-L1, atezolizumab may help restore T-cell–mediated anti-tumor immunity, allowing the immune system to better recognize and attack cancer cells. In the adjuvant setting after cystectomy, this mechanism is intended to help target residual cancer cells that may remain after surgery, particularly in patients with detectable ctDNA molecular residual disease.
IMvigor011 trial and study design
The efficacy of atezolizumab was evaluated in IMvigor011 (NCT04660344), a multicenter, randomized, double-blind, placebo-controlled trial. The study enrolled 250 patients with muscle-invasive bladder cancer who had undergone radical cystectomy with lymph node dissection. Patients were eligible if molecular residual disease was detected through serial ctDNA MRD blood testing during the 12 months after surgery, beginning at least 6 weeks after cystectomy.
Patients were randomized in a 2:1 ratio to receive either atezolizumab 1680 mg or placebo intravenously every 4 weeks. Treatment continued for up to 12 cycles or 1 year, whichever occurred first, unless disease recurrence or unacceptable toxicity occurred.
The major efficacy outcome measure was investigator-assessed disease-free survival (DFS). Overall survival (OS) was an additional efficacy outcome measure.
Key Findings from ESMO 2025
The IMvigor011 results were presented by Prof. Thomas B. Powles at the ESMO Congress 2025 in Berlin and later published in The New England Journal of Medicine on October 20, 2025, under the title “ctDNA-Guided Adjuvant Atezolizumab in Muscle-Invasive Bladder Cancer”.
In the broader study, 761 patients with high-risk MIBC and no radiographic evidence of disease after radical cystectomy entered ctDNA surveillance. Among them, 250 ctDNA-positive patients were randomized to atezolizumab or placebo.
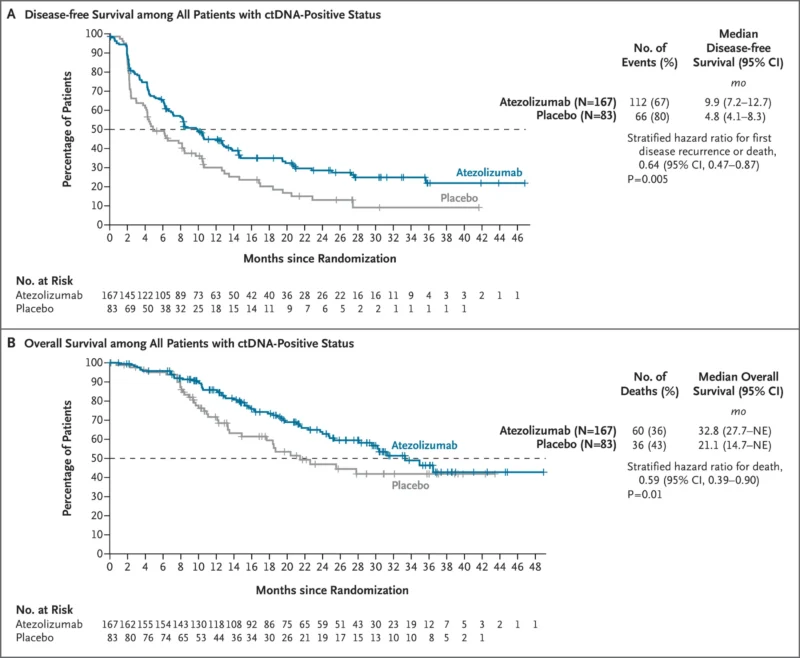
At a median follow-up of 16.1 months, atezolizumab improved DFS compared with placebo, with a median DFS of 9.9 months versus 4.8 months. Overall survival was also improved, with a median OS of 32.8 months versus 21.1 months. Among ctDNA-negative patients, 1-year and 2-year DFS rates were 95.4% and 88.4%, respectively.
Read more about IMvigor011 Trial at ESMO 2025 on OncoDaily.
Efficacy Outcomes
These findings supported the FDA approval of atezolizumab in the adjuvant setting for patients with MIBC after cystectomy who have ctDNA MRD detected by an FDA-authorized test. Atezolizumab showed a statistically significant improvement in disease-free survival compared with placebo, with a median DFS of 9.9 months versus 4.8 months (HR 0.64; p=0.0047). Overall survival was also significantly improved, with a median OS of 32.8 months versus 21.1 months (HR 0.59; p=0.0131).
Safety Profile
The prescribing information for atezolizumab and atezolizumab and hyaluronidase-tqjs includes warnings and precautions for immune-mediated adverse reactions, infusion-related reactions, complications of allogeneic hematopoietic stem cell transplantation, and embryo-fetal toxicity.
Dosing and Administration
For intravenous atezolizumab, the recommended dose is:
- 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks for up to 1 year, unless disease recurrence or unacceptable toxicity occurs.
For atezolizumab and hyaluronidase-tqjs, the recommended dosage is:
- 1875 mg of atezolizumab and 30,000 units of hyaluronidase administered subcutaneously every 3 weeks for up to 1 year, unless disease recurrence or unacceptable toxicity occurs.
Patients with negative Signatera CDx results should continue serial testing until a positive test result or completion of the recommended 12-month testing window.
Clinical Significance of the Approval
This FDA approval introduces a molecular residual disease–guided adjuvant treatment strategy for patients with muscle-invasive bladder cancer after cystectomy. By using ctDNA MRD testing to identify patients at higher risk of recurrence, adjuvant atezolizumab may be directed toward patients with detectable molecular evidence of residual disease.
The IMvigor011 results showed statistically significant improvements in both disease-free survival and overall survival with atezolizumab compared with placebo, supporting its role in this biomarker-selected post-cystectomy population.
The full information about approval is available on the official FDA website.
Read more about What Changed Immunotherapy in Bladder Cancer on OncoDaily.