Sacituzumab tirumotecan is an emerging antibody–drug conjugate (ADC) that is attracting growing attention in oncology for its potential to expand the therapeutic landscape for patients with solid tumors, particularly lung and gastrointestinal cancers. As precision oncology increasingly relies on targeted delivery of potent cytotoxic payloads, ADCs have become one of the most dynamic areas of anticancer drug development. Sacituzumab tirumotecan represents a next-generation approach in this class, combining targeted antibody specificity with a highly potent topoisomerase I inhibitor payload designed to selectively eliminate tumor cells while limiting systemic toxicity.
The rapid development of sacituzumab tirumotecan reflects the broader shift toward targeted cytotoxic therapy in oncology, where advances in tumor biology, drug engineering, and biomarker-driven treatment strategies have converged to produce highly specialized therapeutics. Early clinical studies suggest that sacituzumab tirumotecan may offer meaningful activity in cancers that remain difficult to treat, particularly after resistance develops to standard chemotherapy or targeted therapies.

Understanding Sacituzumab Tirumotecan
Sacituzumab tirumotecan is an antibody–drug conjugate engineered to deliver a potent cytotoxic payload directly to cancer cells expressing the trophoblast cell-surface antigen 2 (Trop-2). Trop-2 is a transmembrane glycoprotein that is highly expressed in many epithelial malignancies, including breast cancer, lung cancer, gastric cancer, and several gastrointestinal tumors. Elevated Trop-2 expression has been associated with aggressive tumor biology, increased metastatic potential, and poorer clinical outcomes in multiple cancer types (Goldenberg et al., 2015).
Antibody–drug conjugates such as sacituzumab tirumotecan combine three essential components: a monoclonal antibody that targets a specific tumor antigen, a cytotoxic payload capable of killing cancer cells, and a linker that connects the two components and controls the release of the drug within tumor cells. In the case of sacituzumab tirumotecan, the antibody component recognizes Trop-2, enabling selective binding to tumor cells that overexpress this protein.
Once the antibody binds to the Trop-2 receptor on the cancer cell surface, the complex is internalized through receptor-mediated endocytosis. Inside the cell, the linker is cleaved, releasing the payload tirumotecan, a potent inhibitor of topoisomerase I. This enzyme plays a crucial role in DNA replication and transcription. By inhibiting topoisomerase I, tirumotecan induces DNA damage and replication stress, ultimately leading to tumor cell death.
The use of topoisomerase I inhibitors as ADC payloads has become increasingly common because these agents are highly cytotoxic yet can be delivered selectively through antibody targeting. The success of this strategy has been demonstrated in other ADCs such as trastuzumab deruxtecan and sacituzumab govitecan, both of which have transformed treatment paradigms in several tumor types (Bardia et al., 2021; Modi et al., 2020).
Mechanism of Action
The therapeutic effect of sacituzumab tirumotecan relies on multiple biological mechanisms that extend beyond simple drug delivery. First, the targeting of Trop-2 allows selective accumulation of the ADC within tumor tissue. Many epithelial tumors overexpress Trop-2 at levels far exceeding those found in normal tissues, creating an opportunity for targeted therapy.
Second, once internalized into cancer cells, the released tirumotecan payload disrupts DNA replication through inhibition of topoisomerase I. This leads to double-strand DNA breaks during replication, triggering apoptosis or other forms of cell death. The high potency of topoisomerase inhibitors makes them particularly effective when delivered directly to tumor cells.
Third, ADCs such as sacituzumab tirumotecan may produce a “bystander effect.” Because the payload is membrane-permeable, it can diffuse into nearby tumor cells even if those cells express lower levels of Trop-2. This phenomenon may enhance efficacy in heterogeneous tumors where antigen expression varies across the tumor microenvironment.
Finally, the immunogenic cell death triggered by ADC-induced cytotoxicity may also stimulate immune responses within the tumor microenvironment. Although this mechanism remains under investigation, emerging evidence suggests that ADCs may synergize with immune checkpoint inhibitors in certain settings (Lambert & Morris, 2017).
Clinical Development
The development of sacituzumab tirumotecan has progressed rapidly through early clinical trials evaluating its safety, tolerability, and antitumor activity across multiple tumor types. Initial phase I studies focused on dose escalation and the identification of recommended dosing schedules.
Early clinical results demonstrated encouraging antitumor activity in patients with heavily pretreated solid tumors. Responses were observed in several malignancies characterized by high Trop-2 expression, including lung cancer and gastrointestinal cancers. These findings have supported further clinical evaluation in larger trials.
In non–small cell lung cancer (NSCLC), sacituzumab tirumotecan has shown promising activity in patients whose disease progressed after standard therapies. Trop-2 expression is frequently observed in NSCLC, making it an attractive target for ADC-based approaches. Early clinical studies have reported objective responses and disease stabilization in patients with advanced disease who had previously exhausted available treatment options.
Similarly, gastrointestinal cancers represent another area of active investigation. Trop-2 is highly expressed in gastric and colorectal cancers, where treatment resistance remains a major clinical challenge. Preliminary results suggest that sacituzumab tirumotecan may provide meaningful clinical benefit in these tumors, particularly when administered after standard chemotherapy.
The encouraging activity observed in early studies has led to the expansion of clinical trials evaluating sacituzumab tirumotecan in a variety of tumor types. Ongoing studies are exploring the drug both as monotherapy and in combination with other anticancer treatments.
Clinical Evidence in Breast Cancer: OptiTROP-Breast01 and OptiTROP-Breast02
A critical component of the clinical development of sacituzumab tirumotecan is its evaluation in advanced breast cancer through the OptiTROP clinical trial program. These studies have explored the activity of this Trop-2–targeted antibody–drug conjugate in different breast cancer subtypes, particularly those with limited treatment options after progression on standard therapies.
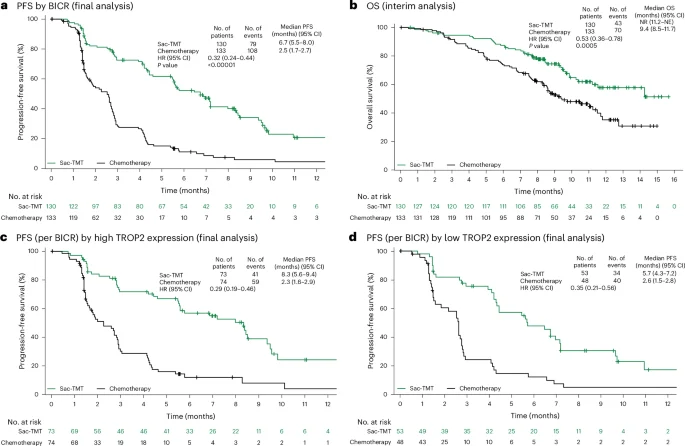
The phase III OptiTROP-Breast01 trial evaluated sacituzumab tirumotecan in patients with previously treated metastatic triple-negative breast cancer (TNBC). TNBC represents one of the most aggressive forms of breast cancer and is characterized by rapid disease progression and a lack of hormone receptor or HER2 targets for therapy (Bianchini et al., 2016). Patients enrolled in OptiTROP-Breast01 had received prior systemic treatments and were randomized to receive either sacituzumab tirumotecan or physician’s choice chemotherapy.
Results from the trial demonstrated that sacituzumab tirumotecan significantly improved clinical outcomes compared with conventional chemotherapy. Patients treated with the antibody–drug conjugate experienced longer progression-free survival, improved overall survival, and higher objective response rates. These findings highlight the therapeutic value of Trop-2 targeting in TNBC and provide important evidence supporting the role of sacituzumab tirumotecan in treatment-resistant disease.
In parallel, the phase III OptiTROP-Breast02 trial expanded the investigation of sacituzumab tirumotecan into hormone receptor–positive, HER2-negative metastatic breast cancer, the most common subtype of breast cancer. Patients with HR+/HER2- disease frequently develop resistance after multiple lines of endocrine therapy and chemotherapy, creating an ongoing need for new therapeutic strategies. OptiTROP-Breast02 was designed to evaluate whether sacituzumab tirumotecan could improve outcomes compared with physician’s choice chemotherapy in this patient population.
The results of OptiTROP-Breast02 demonstrated that sacituzumab tirumotecan significantly improved progression-free survival and objective response rates, reinforcing the potential of Trop-2–targeted ADC therapy beyond TNBC. These findings suggest that sacituzumab tirumotecan may have broad applicability across multiple breast cancer subtypes, particularly in patients with heavily pretreated metastatic disease.
Across both trials, the safety profile of sacituzumab tirumotecan was consistent with other antibody–drug conjugates carrying topoisomerase I inhibitor payloads. The most frequently reported adverse events included hematologic toxicities such as neutropenia and anemia, as well as gastrointestinal symptoms including nausea and diarrhea. These adverse effects were generally manageable with dose adjustments and supportive care.
Together, the OptiTROP-Breast01 and OptiTROP-Breast02 trials represent key milestones in the clinical development of sacituzumab tirumotecan. By demonstrating activity in both TNBC and HR+/HER2- metastatic breast cancer, these studies highlight the broader therapeutic potential of Trop-2–directed ADC strategies and reinforce the growing importance of antibody–drug conjugates in modern breast cancer treatment.
Safety and Adverse Effects
The safety profile of sacituzumab tirumotecan appears broadly consistent with other ADCs containing topoisomerase I inhibitor payloads. Common adverse events reported in early trials include hematologic toxicities such as neutropenia and anemia, as well as gastrointestinal symptoms including nausea, diarrhea, and fatigue.
Neutropenia is among the most frequently observed dose-limiting toxicities associated with topoisomerase I inhibitors. Careful monitoring of blood counts and appropriate dose adjustments are therefore essential during treatment. Supportive measures such as growth factor support may be required in selected patients.
Gastrointestinal side effects are also relatively common but are generally manageable with supportive care and dose modifications. Importantly, the targeted delivery of tirumotecan through the antibody component may reduce systemic exposure compared with traditional chemotherapy, potentially improving the therapeutic index of the drug.
In early studies, most adverse events were manageable and consistent with expectations for ADC therapies. Continued monitoring in larger trials will provide additional insights into the long-term safety profile of sacituzumab tirumotecan.
Potential Role in Future Treatment Strategies
The development of sacituzumab tirumotecan reflects the rapid evolution of antibody–drug conjugates as a cornerstone of modern oncology. ADCs offer a unique therapeutic strategy that bridges targeted therapy and cytotoxic chemotherapy, enabling potent drugs to be delivered selectively to tumor cells.
In the future, sacituzumab tirumotecan may play an important role in several treatment settings. First, it may provide an option for patients with advanced cancers who have progressed after standard therapies. This population represents a significant unmet medical need, particularly in tumors such as lung cancer and gastric cancer.
Second, sacituzumab tirumotecan may eventually be evaluated in earlier lines of therapy. As seen with other ADCs, promising activity in late-stage disease often leads to trials exploring earlier treatment settings where the tumor burden may be lower and therapeutic responses more durable.
Third, combination strategies represent a particularly exciting area of research. ADCs may synergize with immune checkpoint inhibitors, targeted therapies, or other ADCs to enhance antitumor activity. By combining mechanisms of action, these strategies may overcome resistance pathways that limit the effectiveness of single-agent therapy.
Finally, the development of predictive biomarkers may further refine patient selection for sacituzumab tirumotecan therapy. While Trop-2 expression is a key target, additional molecular features may influence response to treatment. Future research will likely explore these factors in greater detail.
The Expanding Landscape of Antibody–Drug Conjugates
Sacituzumab tirumotecan is part of a rapidly expanding class of antibody–drug conjugates that are reshaping the oncology treatment landscape. Over the past decade, advances in linker technology, payload design, and antibody engineering have significantly improved the effectiveness of ADC therapies.
Several ADCs have already demonstrated transformative clinical benefits in cancers such as breast cancer, lung cancer, and hematologic malignancies. The continued development of new ADCs targeting different tumor antigens is expected to further expand treatment options for patients with cancer.
As the field evolves, sacituzumab tirumotecan represents a promising addition to the ADC arsenal. Its ability to deliver a potent cytotoxic payload directly to Trop-2–expressing tumor cells highlights the potential of precision cytotoxic therapy to overcome resistance and improve outcomes in difficult-to-treat cancers.
Conclusion
Sacituzumab tirumotecan represents a promising next-generation antibody–drug conjugate designed to target Trop-2–expressing tumors and deliver a potent topoisomerase I inhibitor directly to cancer cells. By combining precise antibody targeting with a highly cytotoxic payload, sacituzumab tirumotecan exemplifies the growing role of ADC technologies in modern oncology.
Early clinical development demonstrated encouraging antitumor activity across multiple solid tumors, supporting continued investigation of this agent in cancers with high Trop-2 expression. Importantly, the phase III OptiTROP-Breast01 trial has provided pivotal clinical evidence in metastatic triple-negative breast cancer, showing that sacituzumab tirumotecan can improve outcomes compared with conventional chemotherapy in a difficult-to-treat population.
These findings reinforce the therapeutic potential of Trop-2–targeted ADCs and highlight the expanding role of precision cytotoxic therapy in oncology. As additional studies mature and new combination strategies are explored, sacituzumab tirumotecan may become an important component of treatment strategies across multiple tumor types.
More broadly, the development of sacituzumab tirumotecan reflects a larger transformation in cancer drug design. Rather than relying solely on traditional chemotherapy, modern oncology increasingly seeks to deliver potent cytotoxic agents with greater precision. In this evolving landscape, antibody–drug conjugates such as sacituzumab tirumotecan represent a powerful bridge between targeted therapy and chemotherapy, offering new opportunities to overcome resistance and improve outcomes for patients with advanced cancers.
Written by Armen Gevorgyan, MD


