Stephen V Liu, Director of Thoracic Oncology, Chief of the Division of Hematology and Oncology, and Associate Professor at Georgetown Lombardi Comprehensive Cancer Center, shared a post on shared a post on X:
“Phase Ib TROPION-Lung02 Trial of 1L datopotamab deruxtecan (TROP2 ADC) + pembro +/- platinum in NSCLC. Dato-DXd + pembro: RR 55%, DOR 20m, PFS 11m. Dato + pembro + plat: RR 56%, DOR 14m, PFS 7m with more AEs. Phase III AVANZAR trial underway.
TROPION-Lung02 also featured exploratory analysis of Trop2 expression using normalized membrane ratio (NMR) by quantitative continuous scoring (QCS).
This can assess receptor internalization (not just expression), needed for payload release of this ADC. In biomarker positive subset, RR was higher (for doublet and triplet) with trends for longer PFS and OS. For example, using doublet therapy, PFS for TROP2 NMR+ was 21.3m vs 10.9m for negative.”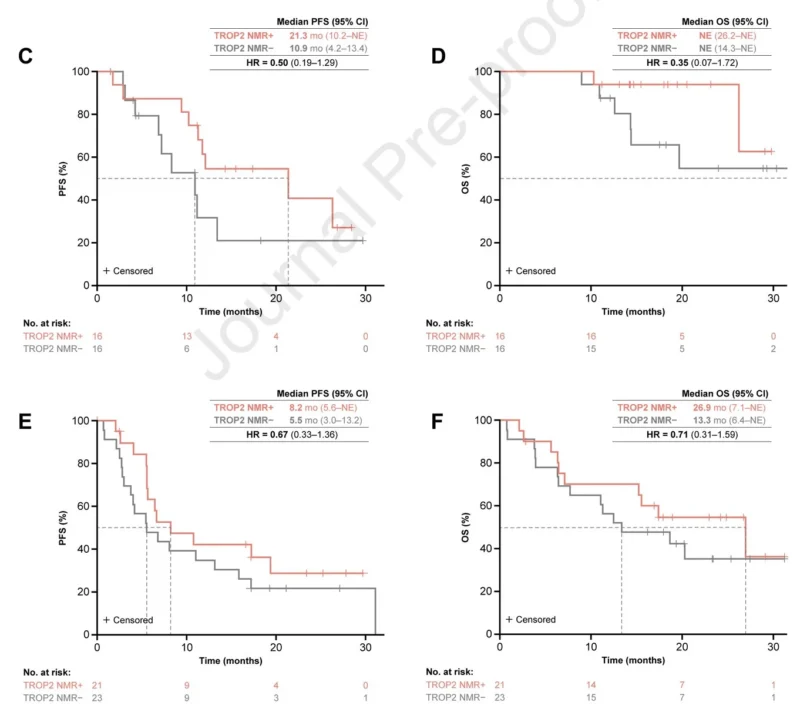
Title: Datopotamab Deruxtecan Plus Pembrolizumab With or Without Platinum-Based Chemotherapy for Advanced or Metastatic NSCLC: The Phase Ib TROPION-Lung02 Trial
Authors: Benjamin Levy, Luis Paz-Ares, Chien-Chung Lin, Scott Herbert, Tsung-Ying Yang, Anthony Tolcher, Yanyan Lou, Yoshitaka Zenke, Diego Cortinovis, Enriqueta Felip, Manuel Domine, Konstantinos Leventakos, Emiliano Calvo, Atsushi Horiike, Edward Pan, Priyanka Basak, Daisy Lin, Wen Gu, Rachel Chiaverelli, Michael J. Chisamore, Yasushi Goto
Read the Full Article.

Other articles featuring Stephen V Liu on OncoDaily.


