Preetam Jain, Consultant Medical Oncologist and Hemato-Oncologist at Bombay Hospital and Medical Research Centre, shared a post on X:
“Big News in Breast Cancer Treatment
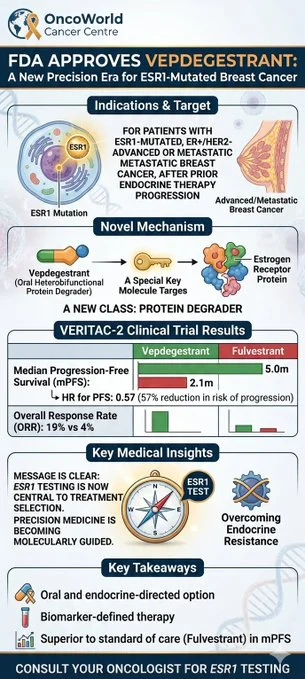
The FDA has approved Vepdegestrant, a novel oral protein degrader for patients with ESR1-mutated ER+/HER2- metastatic breast cancer whose disease has progressed on prior endocrine therapy.
This biomarker-defined approval highlights the critical importance of ESR1 mutation testing to guide treatment decisions. Data from the VERITAC-2 trial shows a significant improvement in median Progression-Free Survival (mPFS) of 5.0 months compared to 2.1 months with fulvestrant (HR: 0.57). An exciting new precision medicine option for our patients.”