Parag Roy, Medical Oncologist at Tata Main Hospital shared a post on LinkedIn:
“Breakwater Trial – Redefining First-Line Therapy in BRAF V600E–Mutated mCRC
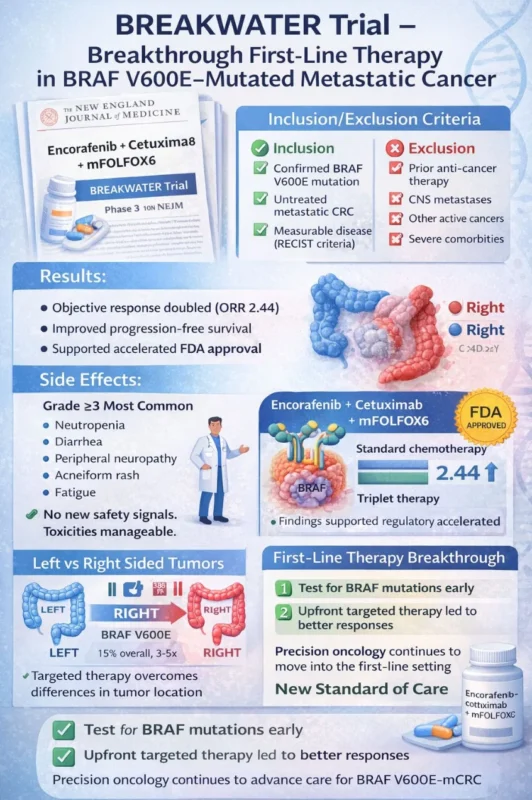
The phase 3 Breakwater study, published in the New England Journal of Medicine, evaluated encorafenib + cetuximab + mFOLFOX6 in previously untreated BRAF V600E–mutated metastatic colorectal cancer (mCRC) – a subgroup historically associated with aggressive biology and poor prognosis.
Key Inclusion Criteria
- Confirmed BRAF V600E mutation
- Previously untreated metastatic colorectal cancer
Key Exclusion Criteria
- Prior systemic therapy for metastatic disease
- Uncontrolled CNS metastases
Results
- Objective Response Rate (ORR) significantly improved
- Odds ratio for response: 2.44 (P < 0.001)
- Higher depth and durability of response
- Progression-free survival benefit observed
This confirms that upfront dual pathway targeting (BRAF + EGFR) combined with chemotherapy improves tumor response compared with standard chemotherapy ± bevacizumab.
Side Effects (Grade ≥3 Most Common)
- Neutropenia
- Diarrhea
- Peripheral neuropathy
- Acneiform rash (EGFR-related)
- Fatigue
Left vs Right-Sided Tumors
BRAF V600E mutations are more frequently associated with right-sided colon cancers, which typically carry worse outcomes and reduced responsiveness to conventional EGFR inhibition alone.
The trial reinforces that:
Right-sided BRAF-mutant tumors benefit from combined BRAF + EGFR blockade
Tumor sidedness remains prognostic, but molecular targeting can overcome some biologic disadvantages—
Conclusion
First-line encorafenib + cetuximab + mFOLFOX6 significantly improves response rates in BRAF V600E–mutated mCRC and represents a new evidence-based therapeutic strategy for this high-risk population.
Take-Home Message
- Test early for BRAF mutations
- Treat biologically, not just anatomically
- Upfront targeted combination therapy improves depth of response
- Precision oncology continues to move into the first-line setting.”
Other articles featuring Parag Roy on OncoDaily.


