Paolo Tarantino, Clinical Research Fellow at Dana-Farber Cancer Institute, shared a post on X:
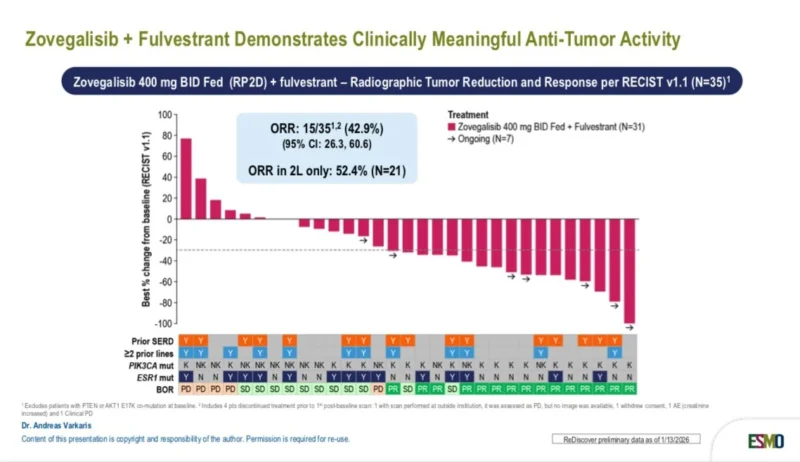
“Updated results from the phase 1 trial of the mutant-specific PI3Ki zovegalisib (RLY2608) + fulvestrant presented at ESMOTAT 2026.
The RP2D (400 mg BID) yielded similar exposure to higher doses, promising activity (ORR 42.9%, mPFS 11.2 months) and favorable safety. Phase 3 ongoing.”
Other articles featuring Paolo Tarantino on OncoDaily.


