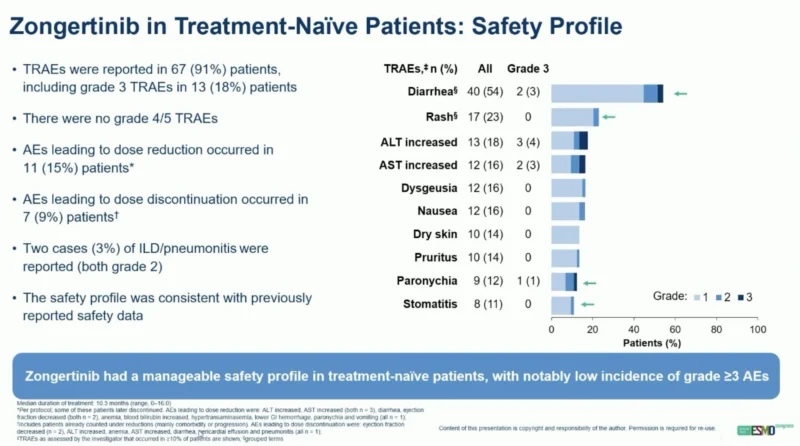
Oncology Brothers shared a post on X:
“Zongertinib now FDA Oncology (accelerated) approved for Her2 (ERBB2) mutated NSCLC in 1L based off Beamion LUNG1 (single arm n=72)
- ORR: 76%
- Responders 64% w/ more than 6 months response and 44% w/ more than 12 months of response
- New SoC.”
Zongertinib Receives FDA Accelerated Approval in HER2-Mutant NSCLC



