Matthew Kurian, Assistant Professor of Medicine at the University of Kentucky College of Medicine and Physician at St. Elizabeth Healthcare, shared a post on LinkedIn:
“Vepdegestrant is FDA approved… what’s next?
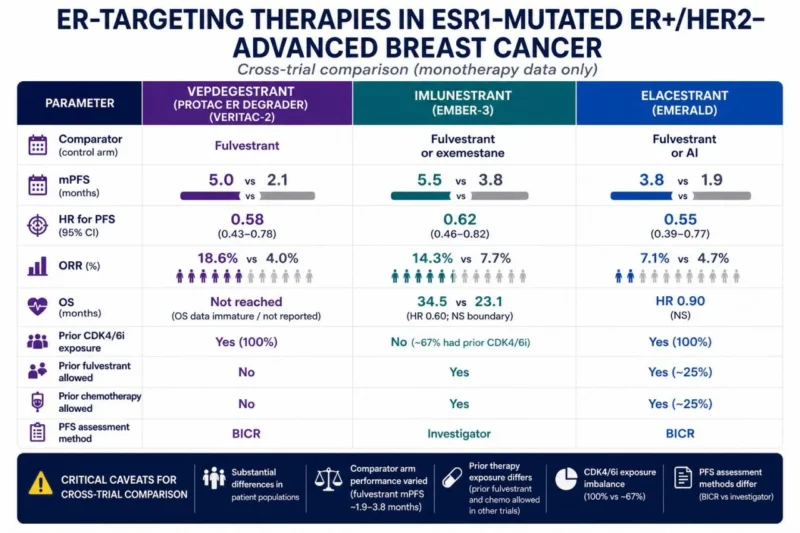
We’ve entered the next phase of ER targeting.
Vepdegestrant (PROTAC ER degrader) is active in ESR1-mutated ER+/HER2 – mBC, but combinations will define its role.
What’s coming next:
- CDK4-selective combos
- TACTIVE-K – + PF-07220060
- Goal: efficacy with ↓ neutropenia
- Early CBR ~52%
- Phase 3 with atirmociclib ongoing
- CDK4/6 (post-CDK space)
- TACTIVE-U – + abema / ribo
- Prior CDK4/6 allowed
- Early signal: CBR 63% (heavily pretreated)
- PI3K / mTOR combos
- Synergy with alpelisib, inavolisib, everolimus
- Potential challenger to inavolisib + palbo + fulvestrant
- Earlier disease
- I-SPY2 – moving PROTACs upfront
Key question:
Will these combinations outperform:
- Fulvestrant + CDK4/6
- Inavolisib + palbociclib + fulvestrant
Especially post-CDK4/6?
Check out my comparison of agents below!”
Other articles featuring Matthew Kurian on OncoDaily.