Diego Gomez Abreo, Head of the Cancer Institute at Hospital Internacional de Colombia, shared a post on LinkedIn:
“Cancer is evolution under pressure. Melanoma is one of its most aggressive expressions.
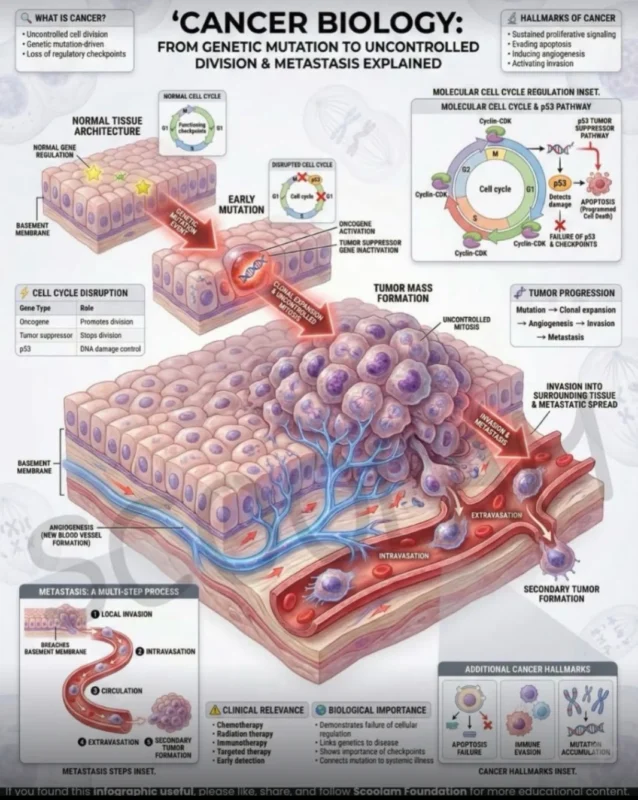
All cancers begin with genetic disruption – activation of oncogenes, inactivation of tumor suppressors, loss of cell-cycle control.
But melanoma adds another layer: extreme genomic instability driven largely by UV-induced DNA damage.
From the general principles of cancer biology:
- Oncogenic activation – In melanoma, often BRAF, NRAS, NF1
- Tumor suppressor loss – CDKN2A, PTEN, TP53 alterations
- Sustained proliferation – MAPK pathway hyperactivation
- Immune evasion – PD-L1 expression, T-cell exhaustion
- Angiogenesis and invasion – Early metastatic competence
Melanoma exemplifies how mutational burden shapes biology.
Its high tumor mutational burden (TMB) makes it immunogenic – which partly explains the transformative impact of immune checkpoint inhibitors.
Yet, the same genomic instability that makes melanoma visible to the immune system also drives heterogeneity and resistance:
- MAPK reactivation after BRAF/MEK inhibition
- Loss of antigen presentation
- Adaptive resistance through microenvironment remodeling
Melanoma is not simply a skin tumor.
It is a dynamic ecosystem – tumor cells, immune cells, stroma, vasculature – constantly evolving.
From early radial growth phase confined above the basement membrane
to vertical growth phase with vascular access
to distant metastasis – including brain tropism –
melanoma reflects the full arc of cancer biology.
The lesson is clear:
Understanding signaling pathways (MAPK, PI3K/AKT), immune regulation, and clonal evolution is not theoretical – it determines whether we choose immunotherapy, targeted therapy, combinations, or neoadjuvant strategies.
In melanoma, biology is destiny – unless we intercept it.”
Other articles on OncoDaily.


