International Urology Cancer Summit shared a post on LinkedIn:
“IUCS Journal Club – Highlights from ASCO GU 2026: Targeting HIF-2α in RCC.
This Journal Club session reviews the data from the LITESPARK clinical program, evaluating the role of belzutifan (a selective HIF-2α inhibitor) across different stages of clear cell Renal Cell Carcinoma (ccRCC).
1. LITESPARK-011: Belzutifan + Lenvatinib in the Post-IO Setting.
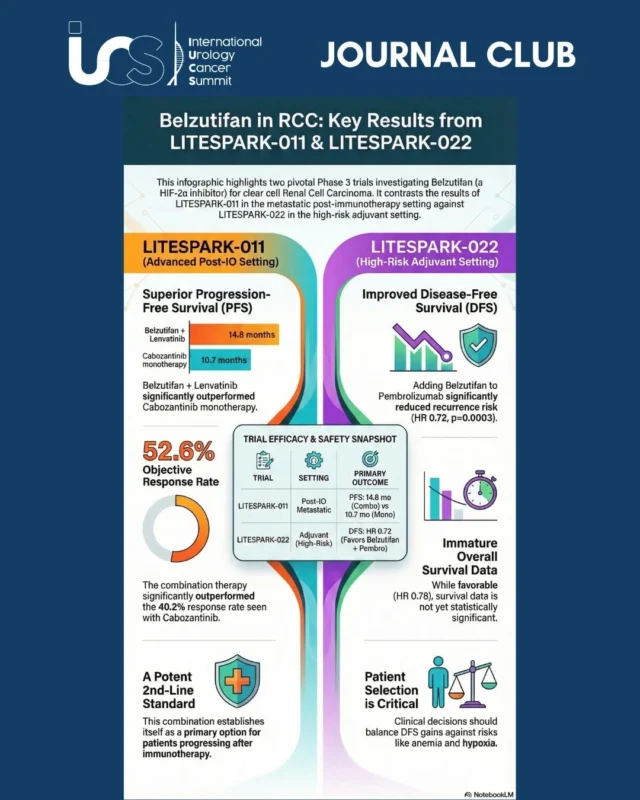
The Phase 3 LITESPARK-011 trial evaluated the combination of belzutifan and lenvatinib versus cabozantinib monotherapy in patients with advanced ccRCC who progressed on or after anti-PD-(L)1 therapy.
Clinical Findings (Data Cutoff April 9, 2025):
- PFS: Significant improvement! 14.8 vs. 10.7 months (HR 0.70; p=0.00007).
- ORR: Higher response rates with the combination (52.6% vs. 40.2%).
- OS: Numerical trend favoring the combo (34.9 vs. 27.6 months), though not yet statistically significant (p=0.06).
- Safety: Manageable profile, with Grade ≥3 TRAEs at 64.1% vs 82.7%.
IUCS Perspective: This combination establishes itself as a potent 2nd-line option for patients progressing after immunotherapy.
2. LITESPARK-022: Adjuvant Belzutifan + Pembrolizumab
The trial investigated whether adding belzutifan to pembrolizumab improves outcomes for patients with resected ccRCC at high risk of recurrence.
Clinical Findings (Data Cutoff August 23, 2025):
- Study design: 1,841 participants randomized to receive Belzutifan + Pembrolizumab vs. Placebo + Pembro (High-risk resected ccRCC)
- DFS: At 28.4 months follow-up, 432 DFS events were observed, favouring the combination of Belzutifan + Pembrolizum over Pembrolizumab (HR 0.72, 95% CI 0.59-0.87), p=0.0003
- Survival Data: OS data remain immature (only 29% information fraction for OS), interim but not significantly improved: OS HR 0.78 (95% CI, 0.51-1.19) p=0.1220.
IUCS Perspective: We are eagerly awaiting mature OS data to see if Belzutifan’s therapeutic window effectively extends to the adjuvant setting.
Let’s discuss! Join the conversation at IUCS 2026 with Stefanie Zschäbitz, Sumanta Pal, MD, FASCO, Vadim Koshkin, Giuseppe Banna, Ravindran Kanesvaran.”
Other articles about ASCO on OncoDaily.


