Alfonso Dueñas Gonzalez, Medical Oncologist and Senior Researcher at the National Cancer Institute of Mexico, shared a post on X:
“Conclusion of My Review on This Preprint.
The study reports on a protocol that uses sub-therapeutic doses while suffering from high attrition and unverified endpoints. While it presents an interesting anecdotal signal, it fails to meet the rigor required for clinical evidence. Rigorous Phase I/II Trials Using the Higher Pharmacologically Supported Doses are Required Before Any Clinical Utility Can Be Claimed.
1. Critical Attrition and Selection Bias
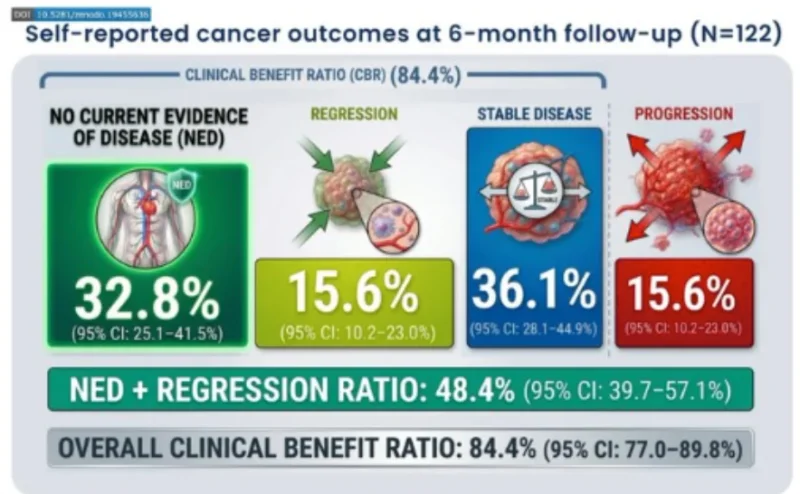
The study’s validity is severely undermined by the catastrophic dropout rate illustrated in Figure 1: The Data Gap: Of the 197 patients enrolled, only 122 (61.9%) completed the 6-month follow-up. Impact: The 75 “lost” patients (38.1%) represent a significant “black box.” In oncology, dropouts often correlate with disease progression or death. Calculating a Clinical Benefit Ratio (CBR) based only on those who survived and chose to respond creates a profound positive responder bias, rendering the 84.4% success rate statistically unreliable.
2. Inadequate Dosing and Pharmacological Misalignment
The most significant scientific flaw is the utilization of sub-therapeutic doses that do not align with established in vitro, in vivo, or pharmacokinetic (PK) data for ivermectin and mebendazole in oncology. Ivermectin: Sub-optimal Concentration The preprint uses a fixed dose of 25 mg. However, evidence in PMC5835698 suggests this is likely insufficient for systemic anti-cancer activity: PK Safety Benchmarks: PK studies in healthy humans have demonstrated that ivermectin is safe at doses up to 2 mg/kg (approximately 140 mg for a 70 kg adult). The preprint’s 25 mg dose is roughly 5–6 times lower than what has been proven safe and potentially necessary for reaching therapeutic tissue concentrations. In Vitro/In Vivo Evidence: Preclinical models indicate that ivermectin’s anti-tumor effects (e.g., inhibition of Wnt/β-catenin and PAK1) are concentration-dependent. The low doses used in this cohort likely fail to achieve the micromolar plasma concentrations required to trigger the 14 anti-cancer mechanisms cited by the authors. This is further evidenced by the study’s own findings on Page 12, which show no dose-response association for efficacy (𝑝=0.91). Mebendazole: Discrepancy with Phase I/II Trials The dose of 250 mg mebendazole is many-fold lower than the Maximum Tolerated Dose (MTD) established in brain cancer trials, where doses up to 200 mg/kg/day or 4,800 mg/day (PMC7333848) have been utilized to overcome poor bioavailability and reach therapeutic levels in solid tumors.
3. Reliability of Outcomes (Unverified Data)
The primary endpoints (regression and “No Evidence of Disease”) were captured via voluntary digital surveys rather than clinical adjudication. Lack of Confirmation: No radiographic evidence (CT/MRI) or medical records were reviewed to verify patient claims. Confounding Therapy: A significant portion of the “benefit” cohort was concurrently receiving chemotherapy (27.9%), radiation (21.3%), or surgery (19.7%). Without a control group, the “signal” cannot be distinguished from the effects of standard-of-care treatments.
4. Ethical Oversight and Conflict of Interest
IRB Irregularity: The authors claim an IRB exemption by labeling a prospective study on vulnerable cancer patients as “quality improvement.” This is a major ethical red flag; prospective interventional data collection requires formal ethical oversight. Commercial Bias: All authors are affiliated with The Wellness Company, which sells the compounded protocol. This direct financial interest, combined with the low-quality data, necessitates extreme skepticism.”
Other articles featuring Alfonso Dueñas Gonzalez on OncoDaily.


