Ahmed Alanazi, Head of Hematology/Oncology Clinical Pharmacy Section at King Fahad Medical City – KFMCK, shared a post on X:
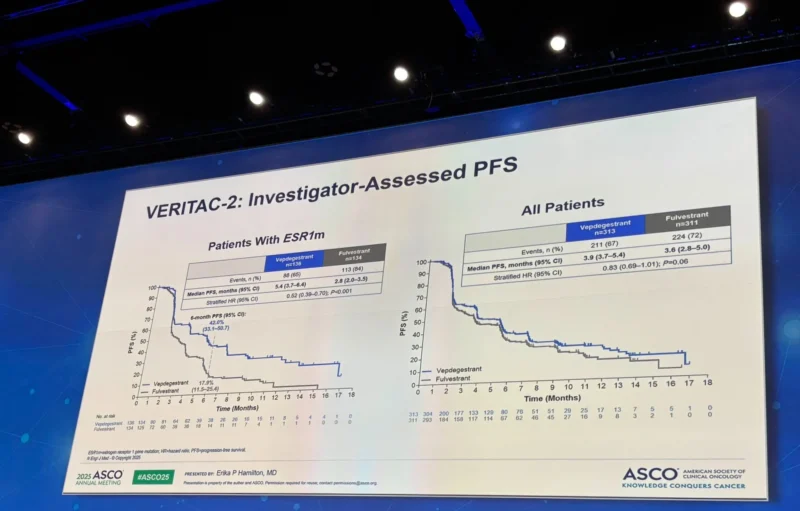
“Vepdegestrant the first PROTAC protein degrader received its FDA
approval based on Veritac2 trial in HR+MBC post ET therapy in ESR1m
3 months vs Fulvesterant, another medication and class to be added to the available option with oral SERDs.
Will this change your practice?”
Gedatolisib in VIKTORIA-1: Positive Topline Data in PIK3CA-Mutant Metastatic Breast Cancer



