Aakash Desai, Assistant Professor and Associate Director of Phase 1 and Precision Oncology Program at the UAB O’Neal Comprehensive Cancer Center, shared a post on LinkedIn:
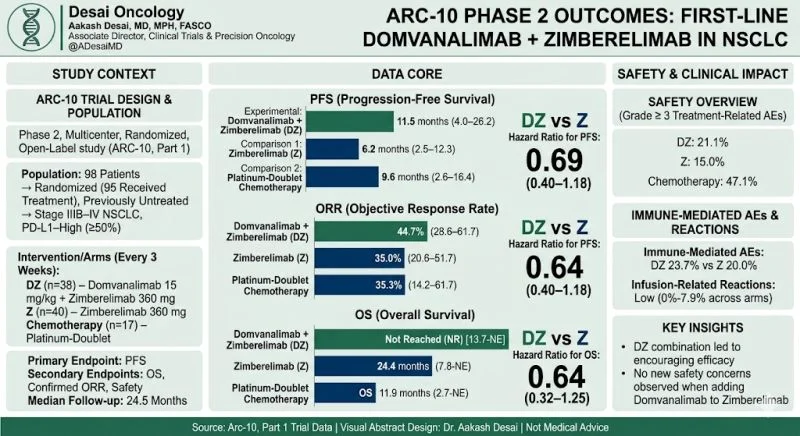
“While PD-1 inhibition is a standard of care in advanced NSCLC, many patients with high PD-L1 expression still experience disease progression. The therapeutic potential of combining Fc-silent anti-TIGIT with anti-PD-1 blockade in the first-line setting required further clinical investigation.
Key findings:
- Primary endpoint: Median PFS was 11.5 (4.0–26.2) months for DZ vs 6.2 (2.5–12.3) for Z, not significant
- Key efficacy: ORR (95% CI) was 44.7% (28.6–61.7) for DZ and 35.0% (20.6–51.7) for Z
- Safety signal: Grade ≥ 3 treatment-related AEs were 21.1% for DZ and 15.0% for Z
How this fits:
Adding Fc-silent anti-TIGIT (domvanalimab) to anti–PD-1 (zimberelimab) led to encouraging efficacy but no statistically significant improvement here, another TIGIT molecule that hits the dust, interesting to see whether we see any additional benefits we can obtain from ongoing PD-1/TIGIT bispecifics being developed.”
Other articles featuring Aakash Desai.


