At the European Lung Cancer Congress 2026, results from Stage 1 of the phase III PRESERVE-003 trial highlighted a promising new therapeutic approach for patients with metastatic squamous non-small cell lung cancer (sqNSCLC) who have progressed after PD-(L)1 inhibitors.
Despite advances in immunotherapy, outcomes remain poor for patients who relapse after checkpoint blockade and platinum-based chemotherapy. In this setting, treatment options are limited, and standard therapies such as docetaxel offer modest survival benefit. PRESERVE-003 introduces gotistobart, a tumor microenvironment-selective CTLA-4 antibody designed to deplete regulatory T cells (Tregs), offering a differentiated mechanism aimed at overcoming immunotherapy resistance.

Study Design and Population
PRESERVE-003 is a randomized, open-label, phase III trial evaluating gotistobart versus docetaxel in patients with metastatic NSCLC who had previously progressed on both PD-(L)1 inhibitors and platinum-based chemotherapy.
Stage 1 included a total of 217 patients, of whom 87 patients with squamous histology were randomized in a 1:1 ratio to receive:
- Gotistobart (6 mg/kg with two loading doses of 10 mg/kg every 3 weeks)
- Docetaxel (75 mg/m² every 3 weeks)
The primary endpoint was overall survival (OS), with secondary endpoints including progression-free survival (PFS), objective response rate (ORR), duration of response (DoR), and safety.
At the time of analysis, the median follow-up was 14.5 months, allowing for meaningful assessment of both short-term and emerging long-term outcomes.
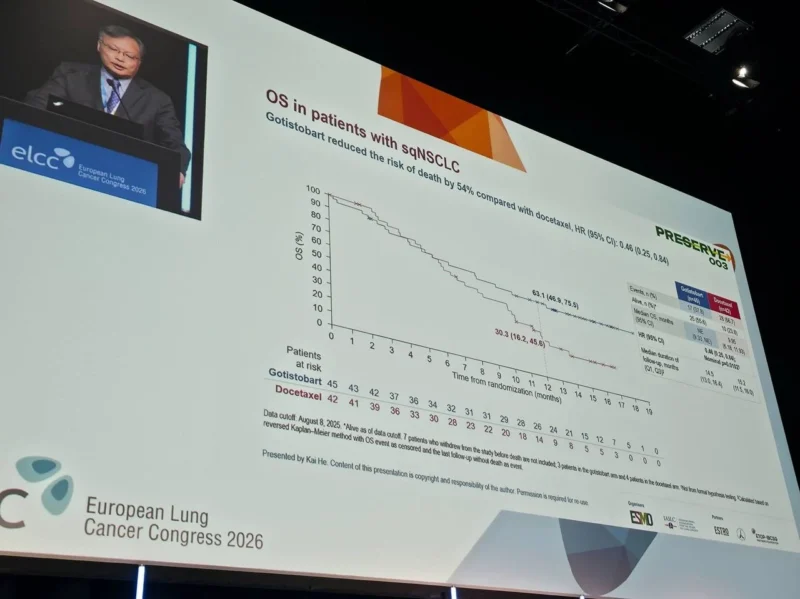
Overall Survival: A Clinically Meaningful Advantage
The most compelling finding from PRESERVE-003 Stage 1 was the significant improvement in overall survival observed with gotistobart.
Median OS was not reached in the gotistobart arm compared to 10.0 months with docetaxel. The hazard ratio for death was 0.46 (95% CI, 0.25–0.84), translating into a 54% reduction in the risk of death.
At 12 months, survival rates further emphasized this benefit:
- 63.1% with gotistobart
- 30.3% with docetaxel
These results are particularly notable in a heavily pretreated population, where survival gains are historically difficult to achieve.

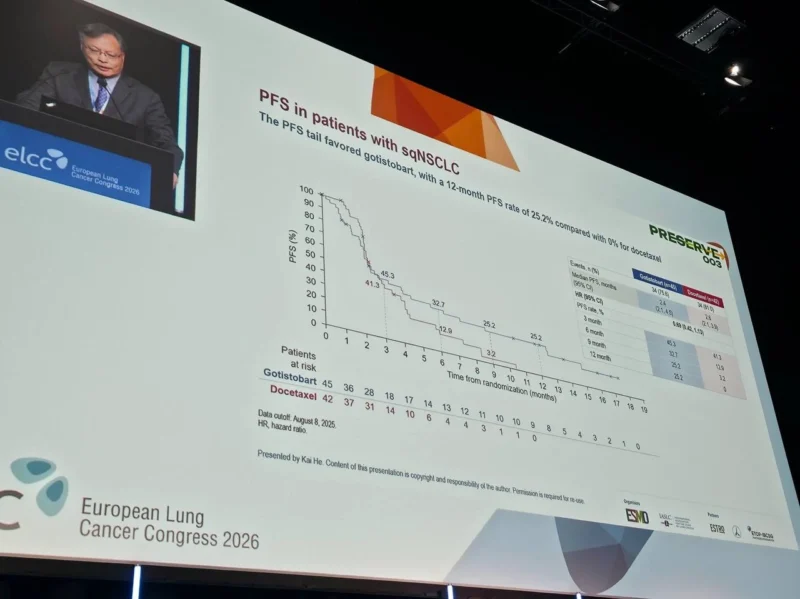
Progression-Free Survival: Beyond Median Values
Median progression-free survival appeared similar between treatment arms:
- 2.4 months with gotistobart
- 2.6 months with docetaxel
However, median values alone did not fully capture the clinical benefit. A closer examination of landmark PFS rates revealed a clear advantage for gotistobart over time.
At 6 months, PFS rates were 32.7% with gotistobart versus 12.9% with docetaxel, and at 12 months, 25.2% of patients treated with gotistobart remained progression-free, compared to 0% in the docetaxel arm.
This pattern suggests that while early progression remains common, a subset of patients derives durable disease control, consistent with immune-mediated mechanisms of action.
Response Rates and Durability
Objective response rates were higher with gotistobart:
- ORR: 20.0% vs 4.8% (gotistobart vs docetaxel)
More importantly, responses were significantly more durable:
- Median duration of response: 11.0 months with gotistobart
- 3.8 months with docetaxel
Durability of response is a key differentiator in immunotherapy, often translating into long-term survival benefit, as observed in this trial.
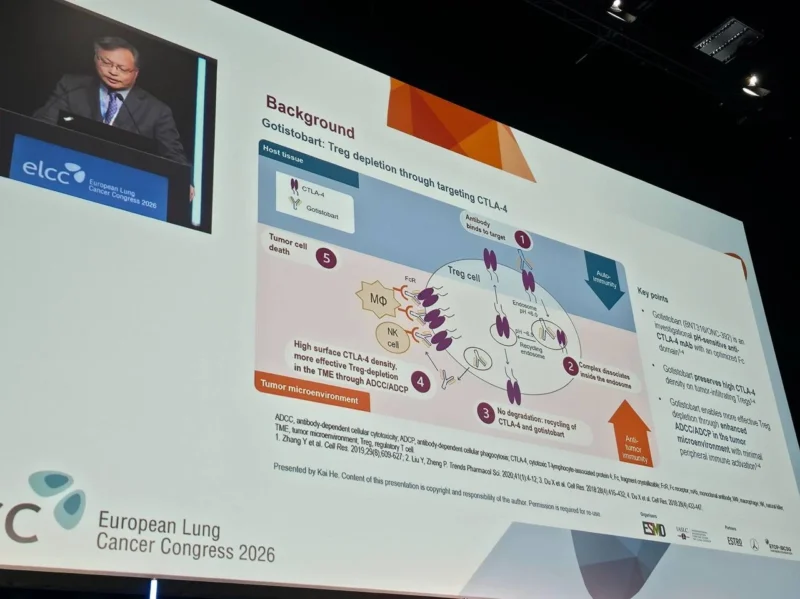
Mechanism of Action: A New Direction in Immunotherapy
Gotistobart represents a next-generation immunotherapy approach. Unlike conventional CTLA-4 inhibitors, which broadly activate the immune system, gotistobart is engineered to selectively deplete regulatory T cells within the tumor microenvironment.
Regulatory T cells play a central role in suppressing anti-tumor immune responses. Their accumulation in tumors is a known mechanism of resistance to PD-(L)1 inhibitors. By targeting these cells, gotistobart aims to restore immune activity where it is most needed, within the tumor itself, while potentially limiting systemic toxicity.
This targeted modulation of the tumor microenvironment may explain the observed durable responses and survival benefit, even in patients previously resistant to immunotherapy.
Clinical Implications
The results from PRESERVE-003 Stage 1 address a critical unmet need in the treatment of metastatic sqNSCLC after progression on PD-(L)1 inhibitors.
Key clinical implications include:
The emergence of a chemotherapy-free treatment option in a setting where standard therapies have limited efficacy, the demonstration of meaningful survival improvement, and the identification of a subgroup of patients achieving long-term disease control.
Importantly, the divergence between median PFS and long-term survival outcomes underscores a well-recognized phenomenon in immunotherapy trials, where traditional endpoints may underestimate the full clinical benefit.
Future Directions
The global Stage 2 portion of the PRESERVE-003 trial is currently ongoing, aiming to confirm these findings in a larger, more diverse patient population.
If these results are validated, gotistobart could represent a practice-changing option in the post–PD-(L)1 setting for sqNSCLC, shifting the treatment paradigm toward targeted modulation of the tumor microenvironment.


