Roquefort Therapeutics plc (LSE: ROQ) has announced the acquisition of the exclusive worldwide licence to AO-252, a clinical-stage, orally administered, brain-penetrant small molecule inhibitor targeting the TACC3 protein, in a transaction valued at £31.875 million, settled entirely in shares.
The deal, sourced from Coiled Therapeutics Inc. a spin-out of A2A Pharmaceuticals is structured as a reverse takeover and will be accompanied by an £8.5 million conditional placing at 10 pence per share. Subject to completion, the company will rebrand as Coiled Therapeutics plc and move its listing from the LSE Main Market to AIM, repositioning itself as a dedicated clinical-stage oncology developer.
The transaction represents one of the more decisive pipeline transformation events in UK biotech in recent months: a pre-clinical company acquiring direct ownership of an asset already in human trials, with early efficacy signals, a compelling mechanism, and a brain-penetrant profile that sets it apart from most mitotic spindle-targeting agents in oncology.
What Is TACC3 and Why Does It Matter in Cancer?
TACC3 (Transforming Acidic Coiled-Coil Containing Protein 3) is a centrosomal scaffolding protein overexpressed in multiple aggressive tumor types, including ovarian, triple-negative breast, endometrial, gastric, prostate, and colorectal cancers, as well as brain metastases.
In healthy tissue, TACC3 plays a limited role, it is not required for the survival of normal cells, a biological asymmetry that makes it an attractive therapeutic target with a potentially favorable therapeutic window.
Within tumor cells, TACC3 serves several oncogenic functions:
- it is integral to mitotic spindle assembly, involved in DNA damage repair and replication
- participates in transcription regulation
- contributes to immune evasion mechanisms.
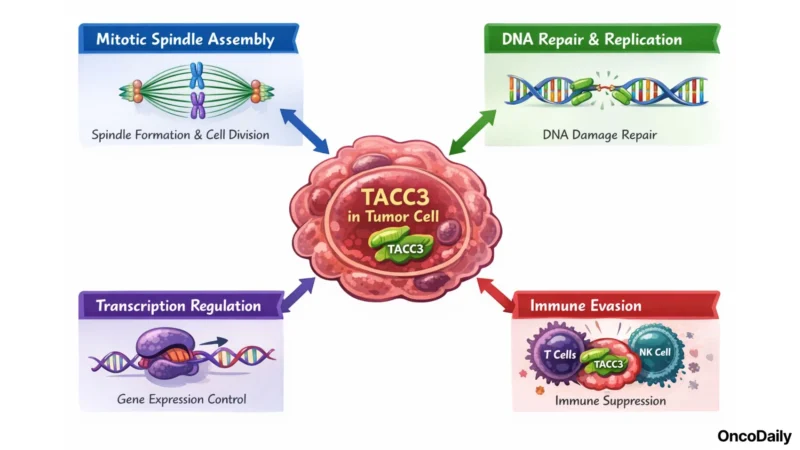
Overexpression of TACC3 has been linked to genomic instability, accelerated tumor proliferation, and resistance to conventional cytotoxic agents.
AO-252 is designed to disrupt TACC3 protein-protein interactions, preventing the protein from fulfilling its role in mitosis and DNA maintenance. Because TACC3 inhibition selectively destabilizes rapidly dividing cancer cells without the broad cytotoxicity of traditional chemotherapy, AO-252 is positioned as a precision oncology alternative, potentially less toxic and more mechanistically targeted than standard-of-care treatment.
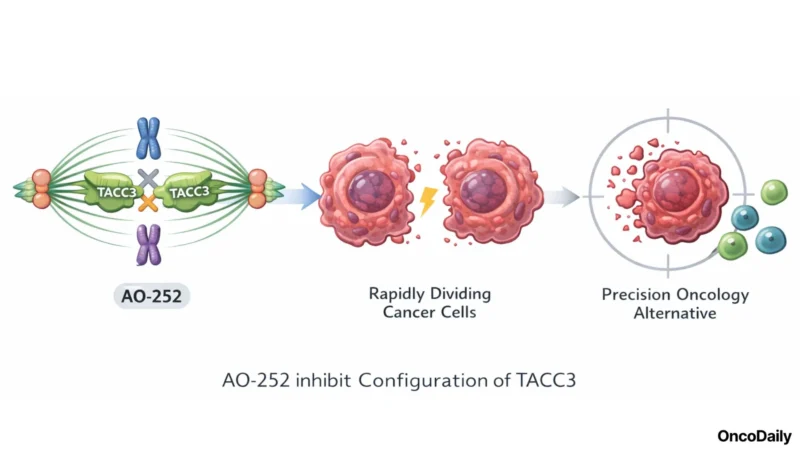
A particularly notable differentiator is AO-252’s blood-brain barrier penetrance. The ability to cross the BBB enables activity against both primary CNS tumors and brain metastases, a clinical domain where effective small-molecule options remain scarce and where many targeted therapies fail.
Phase I Data: Early Signals of Efficacy with a Benign Safety Profile
AO-252 is currently enrolling patients in a U.S. Phase I trial (NCT06136884) for advanced solid tumors, conducted by Coiled Therapeutics prior to the Roquefort acquisition. Early readouts have reported tumor reductions of up to 29% at relatively low dose levels, alongside a consistently described benign safety profile.
The initial trial design has since been expanded, with an amendment filed to broaden enrollment across all solid tumors. Enrollment of ovarian and prostate cancer patients is underway, with the first prostate cancer patient enrolled in November 2025. Dose expansion studies are planned for 2026, and the company has stated its intention to enroll a sufficient patient cohort in 2026 to support planning for Phase 3 registrational trials.
In preclinical models, AO-252 has demonstrated complete tumor regression as a monotherapy across ovarian, triple-negative breast, endometrial, gastric, and prostate cancer models, as well as robust activity in in-vivo brain metastasis models. The company estimates that, based on the biomarker population, AO-252 could be relevant for up to 350,000 patients across multiple indications in the U.S. and European Union.
Competitive Landscape: TACC3 Inhibition in Context
AO-252 occupies a relatively uncrowded competitive position. While the mitotic spindle has long been a validated anti-cancer target, taxanes and vinca alkaloids remain cornerstones of chemotherapy regimens globally, existing agents lack the target specificity of TACC3 inhibition and carry substantial toxicity burdens. There are no currently approved agents that directly target TACC3.
Within the broader landscape of precision oncology targeting mitotic regulators, AO-252 competes conceptually with PLK1 inhibitors (volasertib), Aurora kinase inhibitors, and PARP inhibitors in select indications, but its TACC3-specific mechanism and brain-penetrant profile offer differentiation.
The expansion of the trial into TP53-mutated cancers (ovarian, endometrial, triple-negative breast) is clinically logical: TP53-mutant tumors, which account for the majority of high-grade gynecological and triple-negative breast cancers, are particularly dependent on intact mitotic machinery, potentially amplifying the therapeutic effect of TACC3 disruption.
Implications for the UK Biotech Sector
The Roquefort-to-Coiled Therapeutics transformation is a meaningful signal within the UK oncology biotech ecosystem. AIM, as a market, has historically attracted earlier-stage companies, and the deliberate relocation from the Main Market represents a recognition that the growth-oriented structure of AIM, with its lighter regulatory burden and stronger retail investor participation in biotech, may better suit a company at this stage of clinical development.
The deal also reinforces a broader trend: UK biotechs increasingly accessing U.S.-originated clinical-stage assets as a mechanism for rapid pipeline advancement rather than pursuing de novo drug discovery. With traditional pre-clinical timelines stretching five to seven years before first-in-human studies, reverse takeovers of this type can compress time-to-catalyst substantially.
Forward-Looking Analysis
The next 12 months are laden with inflection points for this asset. The transition to AIM and rebrand to Coiled Therapeutics plc will reset the company’s investor base and visibility. Dose expansion data from the Phase I trial, expected to accumulate through 2026, will be the primary clinical narrative driver. If the benign safety profile holds at higher doses and tumor reduction signals deepen, particularly in prostate and ovarian cancer, the company will be well-positioned to initiate a Phase 3 registrational strategy.
The parallel STAT-6 programme, being advanced toward an IND submission, adds a second clinical asset to what would otherwise be a single-asset pipeline. Regulatory milestones around STAT-6’s IND clearance will provide additional news flow for investors and partners.
What the oncology community will be watching most closely: whether TACC3 inhibition translates into durable responses in refractory solid tumors, and whether the BBB-penetrant profile generates activity in CNS-involved disease, an area of enormous unmet need and limited therapeutic success.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada


