Siren Biotechnology reported that the FDA granted Fast Track designation to SRN-101 for the treatment of recurrent high-grade glioma, following the company’s recent IND clearance enabling a first-in-human study in adults.
Background: recurrent high-grade glioma remains a high-unmet-need setting
High-grade gliomas—including glioblastoma—are among the most aggressive primary brain tumors, and recurrence after initial therapy is common; published reviews continue to describe the recurrent setting as lacking a single standard-of-care systemic therapy, with treatment approaches often involving repeat surgery and/or re-irradiation when feasible and variable use of systemic agents. Reported long-term outcomes remain poor, with glioblastoma five-year survival commonly cited in the single-digit range (often ~5–10%) (Vaz-Salgado et al., 2023). Evidence from randomized studies illustrates the challenge of moving the needle on overall survival at recurrence, for example, trials evaluating combinations such as lomustine plus bevacizumab have shown progression-free survival signals without clear overall survival gains in larger confirmatory work (Wick et al., 2017).
What is SRN-101?
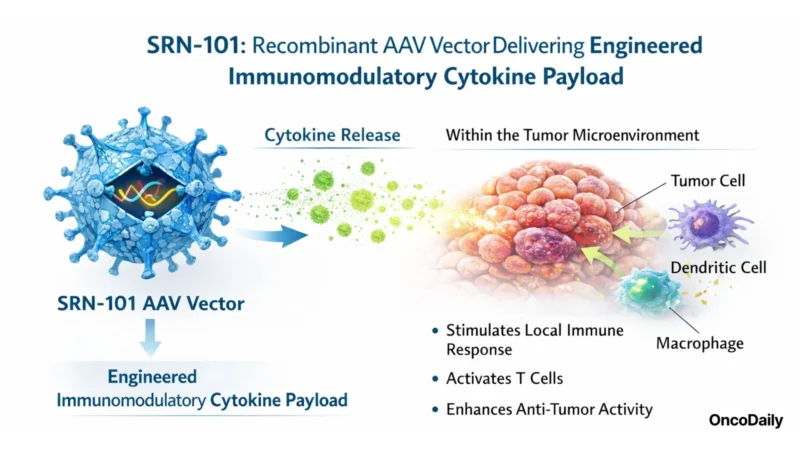
Siren describes SRN-101 as a recombinant adeno-associated virus (AAV) vector delivering an engineered immunomodulatory cytokine payload intended to stimulate a local anti-tumor immune response within the tumor microenvironment. The company positions SRN-101 within a broader “Universal AAV Immuno-Gene Therapy” platform designed around localized and durable intratumoral delivery of immune-modulating payloads in solid tumors, starting in brain cancers.
Regulatory context: Fast Track and what it can change
Fast Track is an FDA expedited program intended to facilitate development and expedite review of drugs that treat serious conditions and address an unmet medical need. FDA materials describe practical features that can matter operationally (e.g., increased interaction with FDA and the possibility of rolling review of a marketing application when appropriate). In SRN-101’s case, the designation arrives immediately after IND clearance, meaning the near-term impact is most likely to be on trial-design feedback cadence and alignment on endpoints and data expectations, rather than any automatic shortening of clinical timelines.
Why is this important?
AAV vectors are established tools in gene therapy more broadly, but their use as a delivery modality for anti-cancer payloads has been less common, in part due to tumor-delivery constraints and the complexity of balancing local expression with safety. If SRN-101 can translate localized cytokine expression into clinically meaningful activity in recurrent high-grade glioma, it would represent a notable entry of AAV-based gene delivery into an area of oncology where systemic therapies frequently struggle and where durable local control is difficult to achieve.
What to watch next
Siren has said it plans to initiate a first-in-human trial in adults with recurrent high-grade glioma; as of the company’s posted materials, SRN-101 was not yet listed on ClinicalTrials.gov, so early protocol specifics (route, dose escalation plan, and immune-monitoring strategy) may become clearer as trial registration and site activation proceed.
Be the first to read the latest FDA approvals in OncoDaily’s FDA Approval articles.


