Neurosymbolic AI — a system that integrates data-driven language models with rule-based reasoning — addresses one of oncology’s most persistent structural failures: low clinical trial enrollment. Only 3%–5% of adult cancer patients enroll in research studies (Loaiza-Bonilla et al., 2026). The causes are well-documented: limited trial awareness, restrictive eligibility criteria, fragmented electronic health records (EHRs), and manual screening processes that are time-intensive and error-prone, particularly in community oncology settings (Peppoloni, Leone et al., ASCO abstract, 2026). Neurosymbolic AI offers structured, explainable, and scalable trial matching by combining linguistic comprehension with deterministic clinical logic.
How Does Neurosymbolic AI Process Patient Data?
Neurosymbolic AI deploys a large language model (LLM) to parse EHRs and extract structured clinical entities — tumor histology, prior lines of therapy, biomarker status, and performance status — mapping them to standardized ontologies including SNOMED CT, ICD, and oncology-specific schemas. This step resolves narrative complexity and ambiguity embedded in clinical documentation.
A knowledge graph then encodes trial eligibility criteria as explicit rules. Deterministic evaluation covers:
– Inclusion and exclusion criteria
– Temporal constraints (e.g., prior therapy washout windows)
– Logical dependencies and negation handling
– Numeric thresholds derived directly from trial protocols on ClinicalTrials.gov
This two-layer architecture separates semantic interpretation from logical adjudication, preventing the conflation of language understanding with clinical decision-making (Peppoloni, Leone et al., ASCO abstract, 2026).
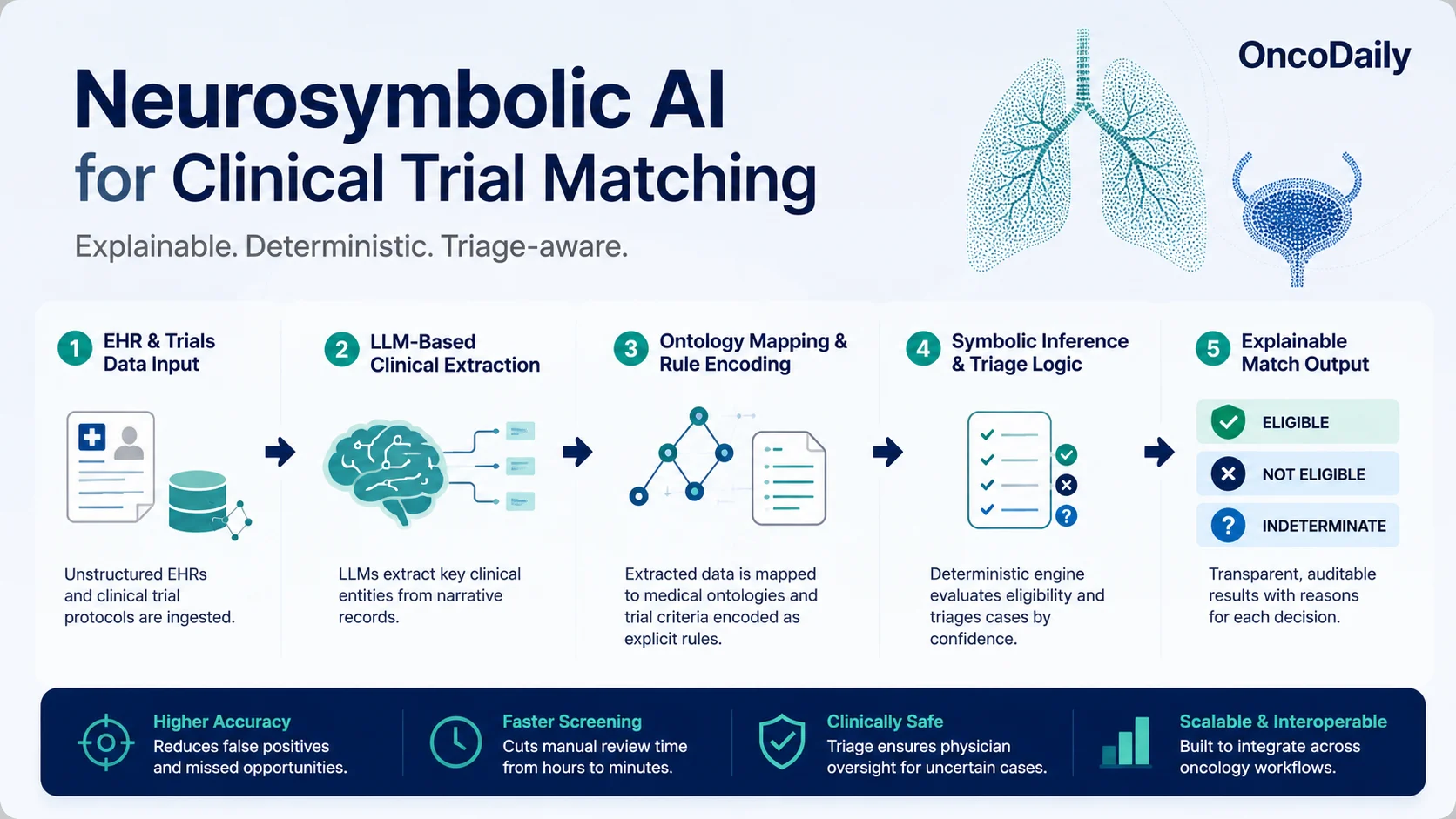
Why Is Deterministic Reasoning Essential in Trial Matching?
Pure LLMs produce probabilistic outputs. In clinical eligibility screening, probabilistic outputs generate inconsistencies and hallucinated interpretations — particularly in temporal reasoning (e.g., therapy sequencing, washout periods) and exception handling. These limitations make LLMs unreliable for eligibility determination without an additional structured validation layer (Prenosil et al., 2025).
Neurosymbolic systems address this through deterministic reasoning that produces transparent, auditable eligibility decisions. Borderline cases — those involving conflicting clinical signals, incomplete data, or ambiguous thresholds — are explicitly flagged for clinician review rather than resolved probabilistically. Real-world evaluations demonstrate improved matching accuracy and reduced screening time compared with manual workflows (Peppoloni, Leone et al., ASCO abstract, 2026).
What Role Does Clinical Triage Play in Patient Safety?
The system incorporates a two-tier triage framework:
– Automated processing: High-confidence matches with clearly satisfied eligibility criteria are resolved without clinician input
– Physician escalation: Cases with incomplete documentation, borderline thresholds, or conflicting signals are routed for review
This design preserves clinical oversight where it matters most. Preliminary evaluations show that hybrid human-AI workflows reduce screening burden while maintaining safety standards across multiple cancer types (Loaiza-Bonilla et al., 2026). The framework is particularly relevant in settings where the volume of active trials exceeds the capacity for manual screening.

Does Neurosymbolic AI Scale Across Oncology Workflows?
These systems are built for interoperability with existing hospital information systems and EHR platforms. Deployment extends across diverse oncology settings and tumor types. Trial databases and eligibility rules update continuously to reflect evolving clinical research landscapes (Loaiza-Bonilla et al., 2026).
Patient-level implementations demonstrate reductions in manual workload and more systematic identification of eligible patients — with direct implications for equity in trial access, particularly in resource-limited settings where manual screening is least feasible. Remaining challenges include data quality variability, incomplete clinical documentation, and the operational burden of continuous rule curation.
Prospective clinical usability and outcome validation represent the next phase before routine implementation.
Giuseppe Leone and Vittoria Peppoloni from AI-ON-Lab, coordinated by Dr. Arsela Prelaj, secured an ASCO poster presentation for their neurosymbolic clinical trial matching project, highlighting its potential to boost enrollment through precise, explainable automation.
Editorial by Giuseppe Leone, MD, Istituto Nazionale Dei Tumori Milano, Università degli Studi di Milano, Italy
FAQ
What is neurosymbolic AI?
Neurosymbolic AI combines neural networks — which learn from data — with symbolic reasoning systems — which apply explicit logical rules. In clinical trial matching, this means a language model interprets free-text EHR data while a rule-based engine applies deterministic eligibility logic.
How does it differ from a standard LLM approach?
Standard LLMs generate probabilistic outputs. Neurosymbolic systems add a deterministic reasoning layer that produces auditable, rule-compliant eligibility decisions rather than probability scores.
Which eligibility criteria does the system evaluate?
The system evaluates inclusion and exclusion criteria, prior therapy timelines, biomarker thresholds, performance status, and logical dependencies between clinical parameters — sourced directly from trial protocols on ClinicalTrials.gov.
What happens when the system is uncertain?
Cases involving incomplete data, conflicting signals, or borderline thresholds are automatically escalated for physician review. The system does not resolve ambiguous cases autonomously.
What are the current limitations?
Data quality variability, gaps in clinical documentation, and the need for continuous protocol-level rule curation remain active challenges. Prospective validation in clinical workflows is ongoing.
Which cancer types are covered?
Current evaluations span multiple cancer types. The system architecture is tumor-agnostic; eligibility rule sets are trial-specific and updated as new protocols are registered.
Does this technology address disparities in trial access?
Systematic patient identification through automated matching reduces the reliance on physician recall and patient proximity to academic centers — two factors that disproportionately limit trial access in community and low-resource settings.


