Neurocrine Biosciences has entered into a definitive agreement to acquire Soleno Therapeutics, marking a strategic expansion into rare disease treatments and strengthening its late-stage pipeline. The deal signals continued consolidation in biotech and highlights growing interest in high-value, niche indications.
Neurocrine Biosciences made the following announcement on LinkedIn:
Today we announced a definitive agreement for Neurocrine to acquire Soleno Therapeutics, a biopharmaceutical company developing and commercializing novel therapeutics for the treatment of rare diseases. Learn more.

Neurocrine Biosciences/LinkedIn
The news drew immediate reactions from industry professionals, with several key perspectives outlined below.
Bill Emker – Senior Director of Recruiting, The Lotus Group LLC
Neurocrine Biosciences has moved aggressively to cement its lead in the rare endocrine space with a 2.9 billion dollar acquisition of Soleno Therapeutics. The deal, priced at 53.00 dollars per share in cash, is a surgical strike to secure Vykat XR, the first and only FDA-approved treatment for hyperphagia in Prader-Willi Syndrome. This isn’t just another bolt-on acquisition; it is a clear strategic pivot toward high-margin, de-risked orphan assets that can leverage Neurocrine’s existing commercial engine.
The logic here is sound. Vykat XR is already on the market and performing, generating 190 million dollars in 2025 revenue with a steep growth trajectory. By folding this into a portfolio that already features Ingrezza and Crenessity, Neurocrine is building a formidable endocrine and rare disease franchise. For the broader market, this deal underscores the premium placed on assets that offer a durable moat and clear pricing power, especially as mid-cap players look to diversify beyond their legacy products.
Melanie Senior – Biopharma Reporter and Science Writer, Nature Portfolio
How valuable is 2026’s M&A fit so far? Buyers have paid over $38 billion in up-front cash (mostly in March) across two dozen deals, according to BioCentury’s database.
The splurge, worth over $47 billion in total, includes hefty payouts for risk-mitigated rare disease drugs. Biogen’s $5.6 billion deal for Apellis (rare kidney)) and Neurocrine’s $2.9 billion Soleno deal (Prader-Willi) both involved marketed products. Merck’s $6.7 billion Terns buy comes with a Phase 1/2 CML program with a “de-risked” MoA.
Look back at our Evaluate Ltd report for a reminder of orphans’ sales trajectory. (As for reasons to curb risk: remember the patent cliff and the world we’re in right now.)
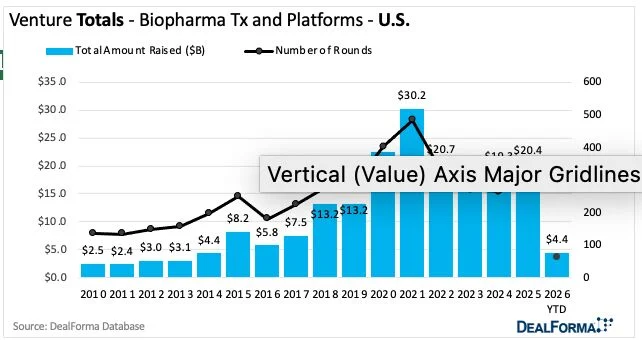
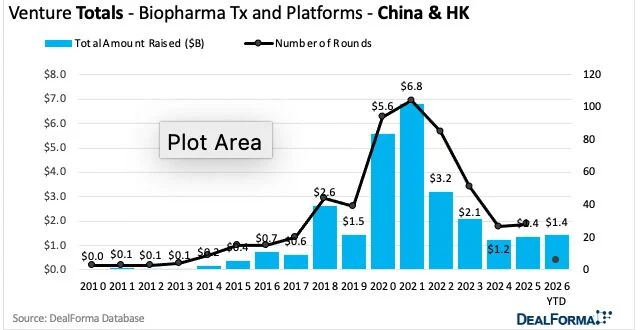
What about VC funding? First quarter totals in the US are trending similarly to last year ($4.4bn). Europe looks slow (0.8bn). But China-based firms raised as much in Jan-Mar 2026 as in the whole of 2025 ($1.4 bn), according to Chris Dokomajilar’s DealForma data covering biopharma therapeutics and platform companies.
I’m asking investors for their “what3words” to describe biotech financing so far in 2026. Mine might be: China. Data. Resilience.
(Should “AI” count as one or two words?)
Melanie Senior/LinkedIn
Melanie Senior/LinkedIn
Jack Shuang Hou – Scientific Director, Jtests
Neurocrine Biosciences to acquire Soleno Therapeutics, Inc. for $2.9B — adds first-and-only Prader-Willi hyperphagia therapy VYKAT XR and expands rare endocrinology leadership
A major rare disease and endocrinology M&A move just landed.
Neurocrine Biosciences announced a $2.9B cash acquisition of Soleno Therapeutics, Inc., bringing in VYKAT XR (diazoxide choline) — the first and only FDA-approved treatment for hyperphagia in Prader-Willi syndrome (PWS).
The deal immediately expands Neurocrine’s commercial portfolio to three first-in-class growth drivers: INGREZZA, CRENESSITY, and now VYKAT XR.
Why this deal stands out
1. Commercial validation is already strong
VYKAT XR generated $190M in 2025 revenue, including $92M in Q4 alone, showing rapid early adoption after its 2025 launch.That significantly de-risks the acquisition and turns this into an immediate revenue expansion move, not a pipeline bet.
2. Strategic fit is unusually clean
As Kyle W. Gano, Ph.D., Chief Executive Officer, Neurocrine Biosciences, highlighted, this directly strengthens Neurocrine’s leadership at the intersection of neuroscience, endocrinology, and rare disease.PWS is a neurodevelopmental disorder with profound metabolic and behavioral burden, making VYKAT XR highly synergistic with Neurocrine’s scientific and commercial infrastructure.
3. Durable exclusivity matters
The IP estate extending into the mid-2040s is one of the most attractive parts of this transaction.For a rare disease therapy with strong first-line positioning, this creates a long-duration cash flow engine.
4. My takeaway
This is a textbook example of strategic commercial M&A done right: approved asset, strong launch curve, durable IP, clear medical need, and immediate portfolio diversification.Credit also to Anish Bhatnagar, M.D., Chairman and Chief Executive Officer, Soleno Therapeutics, Inc. for building a highly differentiated PWS franchise.
The bigger signal: rare endocrine and neurodevelopmental diseases are becoming premium acquisition territory.

Jack Shuang Hou/LinkedIn
Overall, the Neurocrine-Soleno acquisition reflects the broader direction of biotech in 2026: companies are rewarding de-risked innovation, whether through rare disease therapeutics or AI-driven advances, with capital flowing toward assets that offer clear differentiation, commercial traction, and long-term strategic value.
Read more biotech insights on OncoDaily Biotech.


