Asgard Therapeutics Presents First-in-Class Preclinical Data for AT-108, an In Situ Gene Therapy That Reprograms Tumor Cells Into Dendritic Cells
Asgard Therapeutics has disclosed preclinical data for AT-108, a first-in-class in-vivo gene therapy designed to convert tumor cells directly into antigen-presenting dendritic cells (APCs) within the tumor itself, a conceptually distinct approach to cancer immunotherapy that bypasses the need for ex vivo cell manufacturing or systemic immune checkpoint modulation. The data, presented at a major oncology conference, demonstrate systemic, dose-dependent antitumor activity, supporting continued advancement of AT-108 toward IND-enabling studies in solid tumors.
A Fundamentally Different Approach to Tumor Immunity
Conventional cancer immunotherapy strategies, including checkpoint inhibitors, CAR-T cell therapies, and therapeutic vaccines, seek either to reinvigorate pre-existing immune responses or to deliver engineered immune effectors to the tumor. AT-108 represents a mechanistically orthogonal approach: rather than boosting or supplementing the immune response externally, it aims to convert the tumor itself into a source of immune activation.
AT-108 delivers genetic instructions directly to tumor cells in vivo, driving their transcriptional reprogramming into functional dendritic cells. Dendritic cells are the professional antigen-presenting cells of the immune system and are essential for initiating and amplifying tumor-directed T-cell responses. By transforming tumor cells into APCs, AT-108 is designed to generate a highly localized, tumor-antigen-specific immune response capable of driving systemic antitumor activity, including potential effects on distant, non-injected lesions through an abscopal-like mechanism.
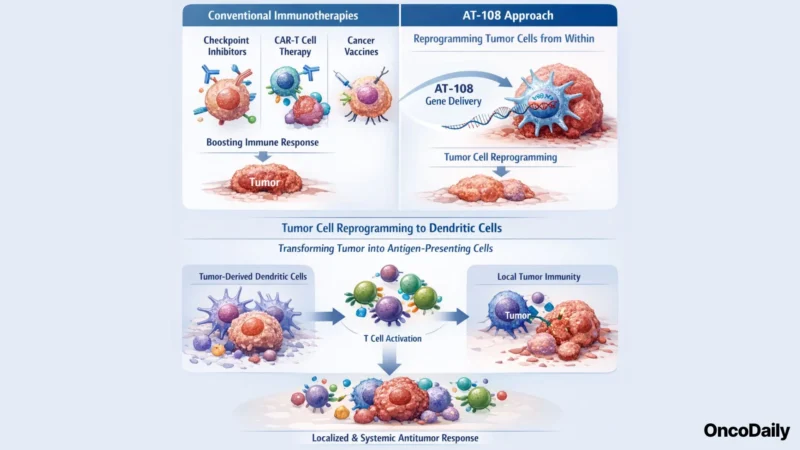
Preclinical Efficacy: Systemic and Dose-Dependent Activity
The preclinical data for AT-108 demonstrate antitumor activity that is both systemic and dose-dependent, two properties essential for clinical translation. Systemic activity suggests that local tumor reprogramming can generate immune effector responses capable of acting beyond the site of injection, a key requirement for the treatment of metastatic solid tumors in which direct intratumorally-accessible lesions may represent only a fraction of total disease burden.
The dose-dependent nature of the response further supports a pharmacologically tractable mechanism and indicates that clinical dose optimization may allow for tailored therapeutic windows in human studies.
IND-Enabling Studies and Development Outlook
Asgard Therapeutics has confirmed that IND-enabling studies are ongoing for AT-108 in solid tumors. The completion of these studies and a subsequent IND filing would represent the first step toward human evaluation of this in situ reprogramming approach. If the preclinical efficacy and safety profile translates to patients, AT-108 could establish a new class of cancer therapy, one that exploits the tumor’s own cellular machinery to initiate immune destruction.
The program is positioned at an early stage of development, and substantial clinical validation will be required before the approach’s full therapeutic potential can be assessed. Nonetheless, the mechanistic novelty and the preliminary evidence of systemic activity make AT-108 one of the more conceptually compelling first-in-class programs to emerge at the intersection of gene therapy and cancer immunology.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada


