For patients with advanced biliary tract cancer, treatment decisions are not guided by efficacy alone. In a disease with limited survival and substantial symptom burden, the amount of time patients spend receiving cancer-related care is also clinically meaningful. This concept, known as time toxicity, reflects the days spent in contact with the healthcare system for treatment, monitoring, procedures, or unplanned care, and may significantly affect quality of life for both patients and caregivers.
A new international retrospective study evaluated time toxicity in patients receiving first-line gemcitabine plus cisplatin (GC) with or without durvalumab (GCD) for advanced biliary tract cancer. The goal was to determine whether adding immunotherapy to standard chemotherapy increases healthcare-related time burden.
The study was published as an Open Access Original Report in JCO Oncology Advances on March 12, 2026.
Title: Time Toxicity in Patients With Advanced Biliary Tract Cancer Receiving Gemcitabine Plus Cisplatin With or Without Durvalumab
Authors: Naohiro Okano, Angelo Pirozzi, Conor O’Donnell, Masato Hayashi, Pedro Luiz Serrano Uson Junior, Celine Hoyek, Oluseyi Abidoye, Cody Eslinger, Binbin Zheng-Lin, Natalie M. Elliott, Elizabeth A. Raupach, Yumei Zhou, Joachim Petit, Daniel Ahn, Mohamad Bassam Sonbol, Christina Wu, Mariko Nishioka, Taro Sato, Maria Fernanda Teixeira, Isabella Sforzin, Hani M. Babiker, Umair Majeed, Nguyen H. Tran, Lorenza Rimassa, Fumio Nagashima, Shuichi Hironaka, Tanios Bekaii-Saab, and Mitesh J. Borad.
Why time toxicity matters in biliary tract cancer
Biliary tract cancer includes intrahepatic and extrahepatic cholangiocarcinoma as well as gallbladder cancer. Most patients present with unresectable or recurrent disease and require palliative systemic therapy. Although gemcitabine plus cisplatin has long been a standard first-line regimen, more recent trials such as TOPAZ-1 established durvalumab plus gemcitabine/cisplatin as a new standard option.
Still, the survival benefit from adding immune checkpoint inhibition remains modest, and this makes the broader treatment burden especially relevant. For patients with advanced cancer, time spent in hospitals, infusion centers, imaging departments, and emergency care can take away from time with family, work, rest, and everyday life. Quantifying this burden may help support more informed and patient-centered treatment discussions.
Study design and methods
This was an international, multicenter, retrospective cohort study conducted across Mayo Clinic sites in the United States, Kyorin University Hospital in Japan, and Hospital Israelita Albert Einstein in Brazil.
The study included adults with histologically confirmed advanced biliary tract cancer who received first-line GC or GCD. Patients were identified between January 2019 and April 2024 at Mayo Clinic sites and between January 2021 and April 2024 at the other participating institutions. Follow-up continued until January 31, 2025.
Time toxicity was defined as the number of days with cancer-related physical contact with the healthcare system from treatment initiation until discontinuation of first-line therapy or last follow-up. This included:
- planned outpatient and inpatient visits for treatment, labs, imaging, symptom control, nutrition, rehabilitation, and related follow-up;
- unplanned outpatient visits for adverse events, symptom management, or biliary drainage issues;
- unplanned hospitalizations.
Multiple services performed on the same day were counted as one visit day. Telephone calls, home care dates, and visits unrelated to biliary tract cancer were excluded.
The primary aim was to quantify time toxicity during first-line systemic therapy and compare it between the GCD and GC groups. The investigators also examined factors associated with higher time toxicity.
Patient population
A total of 193 patients were included:
- 102 received gemcitabine/cisplatin plus durvalumab
- 91 received gemcitabine/cisplatin alone
The median age was 65 years, 50% were male, and 49% had ECOG performance status 0.
The most common primary tumor site was intrahepatic cholangiocarcinoma (58%), and 27% of patients had biliary drainage.
Key findings
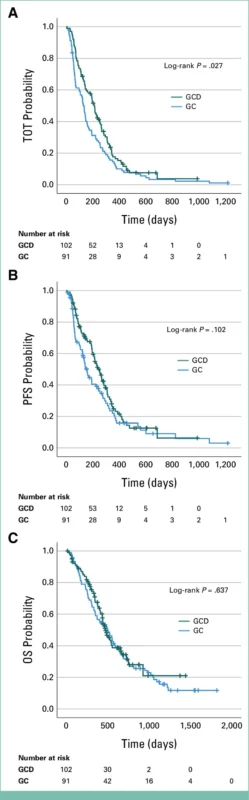
The median time on treatment (TOT) in the overall population was 156 days.
The median proportion of total time toxicity during first-line systemic therapy was 14.4%, meaning that patients spent roughly one-seventh of their treatment time in contact with the healthcare system. Most of this burden came from planned visits, which accounted for 11.9%, while unplanned visits represented a much smaller proportion at 1.8%.
Importantly, although treatment duration was significantly longer in the GCD group than in the GC group, the overall proportion of time toxicity remained similar between the two groups:
- Median TOT: 212 days with GCD vs 134 days with GC
- Median total time toxicity: 27 days with GCD vs 18 days with GC
- Median proportion of total time toxicity during treatment: 13.8% with GCD vs 15.3% with GC
- P = .19 for the difference in proportional time toxicity
This suggests that adding durvalumab prolonged treatment exposure without increasing the proportion of time spent receiving healthcare.
The study also found that total time toxicity strongly correlated with time on treatment and progression-free survival, and moderately correlated with overall survival.
Factors associated with higher time toxicity
On multivariable analysis, two factors were independently associated with higher time toxicity:
- younger age
- poorer ECOG performance status (1–3)
Other variables, including treatment regimen, bilirubin level, biliary drainage, CA19-9 level, and GC schedule, were not independently significant in the final model.
The authors suggested that younger patients may receive more intensive treatment schedules or higher dose intensity, while patients with poorer performance status may require more frequent visits for symptom management and supportive care, although these explanations require further study.
Clinical interpretation
This study introduces an important patient-centered lens for evaluating treatment burden in advanced biliary tract cancer. Even though the addition of durvalumab increases time on treatment, it did not increase the proportion of treatment time spent in healthcare settings. These findings suggest that adding durvalumab prolongs time on treatment without increasing the proportion of healthcare-related time burden compared with GC alone.
At the same time, the study highlights that patients with advanced BTC spend a substantial amount of time receiving care, with a median of nearly one healthcare-contact day per week during first-line therapy. For a population with limited life expectancy, this is highly relevant when discussing treatment goals, quality of life, and expectations with patients and caregivers.
Study limitations
Several limitations should be considered. The study was retrospective and did not capture the quality of patients’ time at home, travel burden, infusion duration, or individual preferences regarding how care is scheduled. It also focused only on first-line systemic therapy, so the full burden of time toxicity across the disease course was not assessed. In addition, comparisons relative to overall survival may have been influenced by shorter follow-up and censoring in the durvalumab group.
Conclusion
This international multicenter study showed that time toxicity is a meaningful and measurable burden in advanced biliary tract cancer, with patients spending a median of 14.4% of their treatment time in contact with the healthcare system. Although durvalumab plus gemcitabine/cisplatin was associated with longer time on treatment, it did not increase the proportion of healthcare-related time burden compared with chemotherapy alone.
These findings suggest that time toxicity can serve as a practical framework for integrating quality-of-life considerations into treatment decision-making in advanced BTC, particularly when survival gains are modest and patient time remains a precious resource.
The full article is available in JCO Oncology Advances.





