Parag Roy, Medical Oncologist at Tata Main Hospital shared a post on LinkedIn:
“New evidence in KIT exon 9–mutant GIST just dropped in JAMA Oncology.
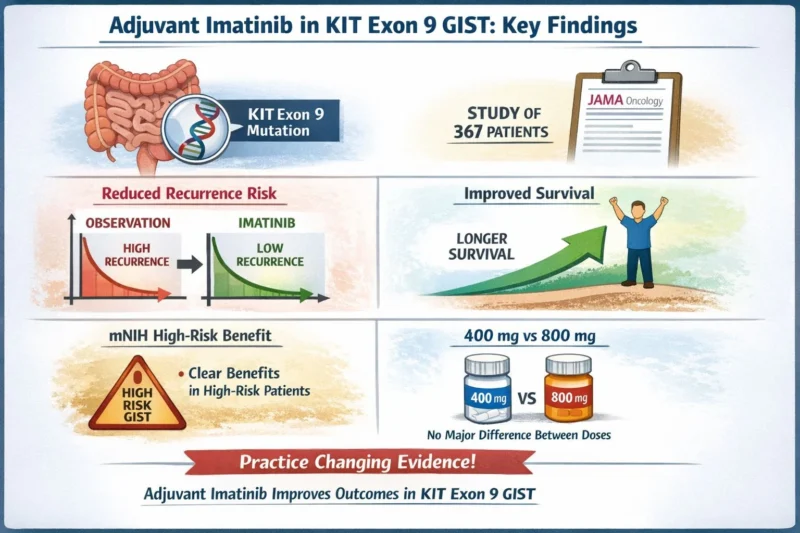
Study: Adjuvant Imatinib or Observation in Patients With Gastrointestinal Stromal Tumors With KIT Exon 9 Mutations
First author: Andrea Napolitano
International multicenter cohort (n=367)
Why this matters
KIT exon 9 GIST has long been considered relatively less sensitive to standard-dose imatinib—especially in advanced disease. But what about the adjuvant setting after curative surgery?
Key Findings
- Adjuvant imatinib significantly reduced early recurrence risk
- Associated with improved overall survival
- Benefit confirmed in mNIH high-risk patients
- No significant difference between 400 mg vs 800 mg daily dosing in high-risk subgroup
Despite its cytostatic nature (with diminishing effect over time), adjuvant imatinib delayed recurrence and translated into an OS benefit.
Take-Home Messages
- Adjuvant imatinib should not be withheld in high-risk KIT exon 9 GIST.
- Survival benefit is observed—even in this historically “dose-challenging” molecular subtype.
- Dose escalation to 800 mg in the adjuvant setting may not provide additional benefit.
- Recurrence remains common (~75% in high-risk), highlighting the need for better strategies and prospective trials.
This study provides the strongest real-world evidence to date supporting adjuvant therapy in KIT exon 9–mutant GIST.
Precision oncology is not just about mutation identification—
It’s about understanding context: stage, biology, and timing.”
Other articles featuring Parag Roy on OncoDaily.


