Patients with locally advanced resectable colon cancer remain at substantial risk of recurrence despite surgery and standard chemotherapy. While neoadjuvant immunotherapy has shown striking activity in dMMR/MSI-H colorectal cancer, the vast majority of colon cancers are mismatch repair proficient/microsatellite stable (pMMR/MSS), where the benefit of immunotherapy has been far less clear.
The AZUR-4 trial was designed to explore whether adding dostarlimab, a PD-1 inhibitor, to CAPEOX in the neoadjuvant setting could improve early antitumor activity and help identify biomarkers of response in this more common and biologically challenging subgroup.
The study was published as a Current Trial Report (Articles in Press) in Clinical Colorectal Cancer on March 12, 2026.
Title: AZUR-4, a Phase 2, Open Label, Randomized Study of Neoadjuvant Dostarlimab Plus CAPEOX Versus CAPEOX in Previously Untreated T4N0 or Stage III Mismatch Repair Proficient/Microsatellite Stable Resectable Colon Cancer
Authors: Gertjan Rasschaert, Allyson M. Peddle, Sara Verbandt, Sabine Tejpar, Nicholas P. West, Jenny Ross, Julie Switzky, Morad Marco Balas, Ken Edwards, Gabor Kari, Sue Griffin, Daniel N. Cohen, Emily Hammatt, Edmund K. Moon
Background
Colon cancer remains one of the most common cancers worldwide. For patients with high-risk stage II and stage III disease, the current standard approach consists of surgery followed by adjuvant chemotherapy. However, recurrence still occurs in a substantial proportion of patients.
Neoadjuvant treatment has re-emerged as an important strategy in colon cancer. The FOxTROT trial demonstrated that neoadjuvant fluoropyrimidine-oxaliplatin–based chemotherapy could improve 2-year recurrence rates in locally advanced colon cancer, particularly in the pMMR population.
At the same time, immunotherapy has transformed outcomes in dMMR/MSI-H colorectal tumors, but this success has not translated as clearly into pMMR/MSS disease. Because these tumors generally harbor fewer neoantigens, immunotherapy alone may be insufficient in most cases. Still, some reports suggest that selected pMMR/MSS tumors may respond, supporting further investigation of combination strategies.
The rationale behind AZUR-4 is that chemotherapy may enhance tumor antigenicity, while immunotherapy may strengthen adaptive antitumor immune responses before surgery. This combination has already shown benefit in several other solid tumor settings.
Methods
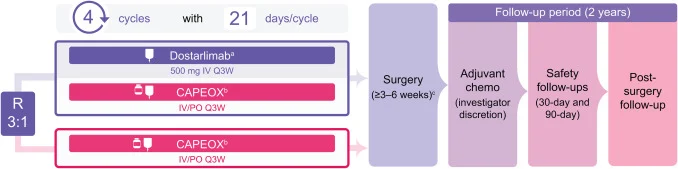
AZUR-4 (NCT06567782) is a multicenter, phase 2, open-label, randomized (3:1) study of 120 patients across multiple countries, sponsored by GlaxoSmithKline. The study started on February 17, 2025, has an estimated enrollment of 120 patients, and was last updated on March 12, 2026. The estimated primary completion date is November 23, 2026, with final study completion expected in October 2028.
The trial enrolls adults aged 18 years or older with previously untreated, resectable pMMR/MSS colon adenocarcinoma clinically staged as T4N0 or stage III. Participants must have adequate organ function and an ECOG performance status of 0 or 1.
Patients are randomized in a 3:1 ratio to receive either dostarlimab plus CAPEOX or CAPEOX alone as neoadjuvant treatment. Both groups receive four 21-day cycles. In the experimental arm, dostarlimab is administered at 500 mg intravenously every three weeks. CAPEOX consists of oxaliplatin 130 mg/m² intravenously on day 1 and capecitabine 1000 mg/m² orally twice daily for 14 days of each cycle.
After completion of neoadjuvant therapy, patients undergo surgery 3 to 6 weeks later. Postoperative adjuvant chemotherapy may be administered at the investigator’s discretion, but no additional dostarlimab is given after surgery.
Endpoints
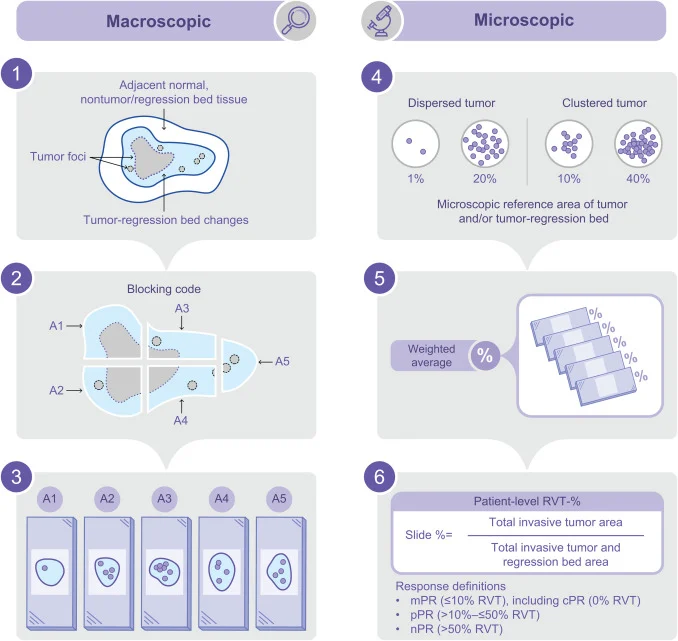
The primary endpoints are major pathological response, determined by local assessment, and safety. Major pathological response is defined as ≤10% residual viable tumor.
Secondary endpoints assess the feasibility of the regimen and pathological response, including the proportion of patients unable to undergo resection because of disease progression or treatment-related toxicity before surgery.
Exploratory endpoints include overall survival, event-free survival, biomarker readouts, and pathological response rate in biomarker subsets. The study incorporates extensive translational research, including genomic, transcriptomic, and proteomic analyses of blood and tumor samples, as well as organoid models and artificial intelligence–based approaches to identify predictive biomarkers.
Statistical Design
The trial uses a Bayesian primary analysis incorporating historical data for the CAPEOX arm, supporting the 3:1 randomization design. The primary estimand is the difference in major pathological response rate between treatment groups, with missing responses considered nonresponders.
Assuming a major pathological response rate of 23% in the CAPEOX arm and a true difference of 27% between arms, a sample size of 120 patients has a 95% probability of declaring success. A secondary frequentist analysis is also planned.
Why This Trial Matters
AZUR-4 addresses a key unmet need in resectable colon cancer. Unlike dMMR/MSI-H tumors, pMMR/MSS tumors typically show limited sensitivity to checkpoint inhibition, making this population particularly challenging.
Importantly, the trial not only evaluates a neoadjuvant chemoimmunotherapy strategy but also integrates a comprehensive biomarker program. It aims to identify which patients may benefit from immunotherapy-based approaches, moving toward more precise treatment selection in pMMR/MSS disease.
Conclusion
The AZUR-4 trial evaluates the effect of adding dostarlimab to CAPEOX in previously untreated T4N0 or stage III pMMR/MSS resectable colon cancer. With its focus on pathological response, surgical feasibility, and deep translational profiling, the study may provide early insight into the role of neoadjuvant chemoimmunotherapy in this setting.
If positive, AZUR-4 could help redefine treatment strategies and support biomarker-driven patient selection in localized pMMR/MSS colon cancer.
The full article is available in Clinical Colorectal Cancer.