Uveal melanoma is the most common primary intraocular malignancy in adults, but it remains a rare cancer overall. Its rarity creates major practical challenges, because optimal care depends on timely diagnosis, specialized pathology, expert imaging, access to ocular oncology services, and coordinated multidisciplinary management. Unlike cutaneous melanoma, uveal melanoma is biologically distinct, follows a different metastatic pattern, and has historically shown limited sensitivity to conventional systemic treatments. For this reason, the guideline emphasizes that all patients should ideally be managed through, or at least discussed with, reference centers that have dedicated expertise in primary and metastatic uveal melanoma.
The document focuses specifically on uveal melanoma, not cutaneous or mucosal melanoma, and provides recommendations for diagnosis, staging, molecular assessment, local treatment, metastatic management, surveillance, and survivorship.
Epidemiology and Risk Factors
Uveal melanoma differs from cutaneous melanoma not only in biology, but also in epidemiology. Its incidence has remained relatively stable over time and, except for iris melanoma, it is not linked to ultraviolet exposure. A geographic gradient exists, with higher incidence in Northern and Western Europe and Oceania, and lower incidence in Asia, Africa, and South America.
The disease occurs predominantly in non-Hispanic white populations, especially in individuals with light-colored irises, and incidence rises with age. Although it can occur at any age, pediatric cases are rare. Most tumors arise from the choroid, while fewer originate in the ciliary body or iris.
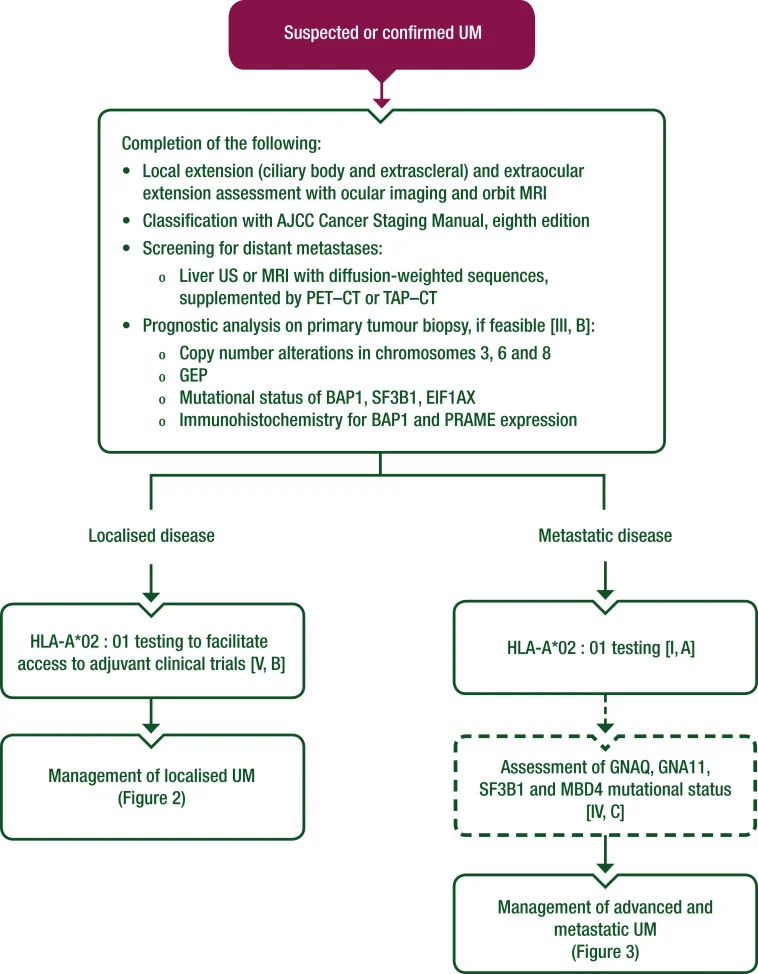
Most cases are sporadic, but a small proportion are familial. Germline predisposition is particularly important in patients with BAP1 tumor predisposition syndrome, which may include uveal melanoma, mesothelioma, cutaneous melanoma, and renal cell carcinoma. In selected patients, such as those with bilateral disease, diagnosis before age 40, or suggestive family histories, genetic counseling and germline testing should be considered.
 Diagnosis and Pathology
Diagnosis and Pathology
The diagnosis of uveal melanoma is usually made clinically, not histologically. In most patients, typical findings on:
- ophthalmoscopy,
- fundus photography,
- and conventional ocular ultrasound
are sufficient to establish the diagnosis. This is an important practical point: biopsy is not mandatory in a typical case.
Clinically, uveal melanoma presents as a pigmented or amelanotic intraocular mass, most often within the choroid. Histologically, tumors may be composed of spindle cells, epithelioid cells, or a mixture of both. Certain pathological features correlate with worse prognosis, including:
- epithelioid morphology,
- a high mitotic count,
- ciliary body involvement,
- extrascleral extension,
- and the presence of extravascular matrix patterns.
When the diagnosis is uncertain, biopsy is recommended. Fine-needle aspiration biopsy may also be discussed in patients managed conservatively when prognostic or molecular information is expected to influence surveillance or trial eligibility. However, biopsy carries real limitations and risks, including hemorrhage, tumor seeding, and the possibility of insufficient tissue sampling. Therefore, the decision must be individualized and made after a clear discussion with the patient.
Molecular Biology and Prognostic Testing
The molecular landscape of uveal melanoma is markedly different from that of cutaneous melanoma. Uveal melanoma typically has a low tumor mutational burden, but its biology is strongly shaped by chromosomal abnormalities and specific driver mutations.
Early oncogenic events most commonly involve mutually exclusive activating mutations in GNAQ or GNA11. Less frequently, early events include CYSLTR2 or PLCB4 mutations. These activate signaling pathways such as MAPK, PI3K, and YAP.
The most clinically relevant prognostic alterations include:
- monosomy 3,
- 8q gain,
- BAP1 inactivation,
- SF3B1 mutations,
- EIF1AX mutations,
- and PRAME expression.
These molecular features help define biologic risk groups.
Low-risk tumors generally retain chromosome 3, lack adverse chromosome 8 abnormalities, often show 6p gain, tend to be GEP class 1, and may harbor EIF1AX mutations.
Intermediate-risk tumors are often associated with SF3B1 mutations, PRAME positivity, and delayed metastatic relapse.
High-risk tumors are strongly associated with:
- monosomy 3 or isodisomy 3,
- 8q polysomy,
- GEP class 2,
- and BAP1 loss or mutation.
The guideline does not endorse one prognostic platform over another, because multiple techniques are currently used, including FISH, copy-number assays, targeted NGS, and gene expression profiling, without definitive comparative superiority data.
An important practical point is that HLA-A*02:01 testing should be carried out in the metastatic setting because it determines eligibility for tebentafusp, now the most important systemic therapy advance in metastatic uveal melanoma.

KIMMTRAK (Tebentafusp) uses in Cancer: Dosages, Indications, Expectations and more
Staging and Risk Assessment
Uveal melanoma should be staged according to the AJCC 8th edition. This staging system remains anatomy-based and incorporates tumor thickness, basal diameter, ciliary body involvement, and extraocular extension. These features correlate strongly with metastatic risk.
However, the guideline makes clear that AJCC staging is no longer sufficient by itself for modern prognostication. Molecular and cytogenetic data substantially refine risk prediction. In practice, staging and risk assessment should combine:
- clinical features,
- ocular imaging,
- and, when available, molecular profiling.
Patients should also undergo evaluation for distant metastases, especially of the liver, using liver ultrasound or preferably liver MRI, often supplemented by thoraco-abdomino-pelvic CT or PET-CT when extrahepatic disease is suspected.
Management of Localized Primary Uveal Melanoma
Most patients present with localized disease. The major goal of treatment is local tumor control while preserving the eye and useful vision whenever possible. The choice of treatment depends on:
- tumor size,
- tumor location,
- extraocular extension,
- visual function,
- local expertise,
- and patient preference.
Enucleation
Enucleation remains necessary for selected patients, particularly those with:
- very large tumors,
- extensive extraocular extension,
- painful blind eyes,
- or tumors replacing a major portion of the globe.
Although eye-preserving strategies are preferred whenever feasible, enucleation is still an appropriate and sometimes necessary oncologic procedure.
Conservative Surgery
Local resection may be considered in selected small iris or ciliary body tumors, but these procedures are technically demanding and carry risks such as hemorrhage, retinal detachment, and ocular hypertension. They are not the standard approach for most patients.
Radiotherapy
Radiotherapy is the cornerstone of globe-preserving treatment.
Plaque brachytherapy
Plaque brachytherapy is widely used for small- to medium-sized tumors and is often more available and less expensive than proton therapy. Long-term survival appears comparable to enucleation in appropriately selected patients, and local control rates are high.
Proton beam radiotherapy
Proton beam radiotherapy is especially useful for larger tumors or tumors close to critical ocular structures. It provides excellent local control and is often preferred when brachytherapy is technically difficult. However, it is more expensive and less widely accessible.
Stereotactic radiotherapy
Stereotactic radiotherapy is an alternative when brachytherapy or proton therapy are unavailable. It is effective but may be associated with more ocular complications and has less robust prospective validation.
Overall, the guideline recommends that if eye-preserving treatment is feasible, radiotherapy should generally be preferred over local resection.
Follow-Up After Local Treatment
Ophthalmologic follow-up remains essential after local therapy, not only to detect recurrence but also to manage radiation-related complications. These complications can include:
- radiation maculopathy,
- optic neuropathy,
- retinopathy,
- neovascular glaucoma,
- cataract,
- and dry eye.
Radiation-induced posterior segment injury is a major cause of visual loss. Intravitreal anti-VEGF therapy is recommended to reduce intraocular neovascular complications and may help preserve anatomy and vision in selected cases.
Patients should generally undergo ophthalmologic examination every 6 months for the first 2–5 years, then yearly thereafter, although closer follow-up may be appropriate if tumor stability is uncertain. Color fundus photography is highly effective for recurrence detection when the lesion is visible; ultrasound is preferred when complete visualization is not possible.
Management of Local Recurrence
There is no single standard approach to local recurrence. Management depends on the size, location, and extent of recurrence, as well as prior therapy. Options include:
- transpupillary thermotherapy for selected small recurrences,
- repeat radiotherapy,
- and enucleation.
Extraocular recurrence requires a broader staging work-up and often more extensive surgery, potentially with adjuvant radiotherapy.
Adjuvant Therapy in Localized Disease
At present, there is no standard adjuvant systemic therapy for resected or irradiated uveal melanoma. This remains an area of major unmet need.
Trials with adjuvant chemotherapy and targeted therapy have not demonstrated clear benefit. One study suggested that nivolumab plus ipilimumab might prolong metastasis-free survival, but toxicity was substantial and data remain limited. Several important trials are ongoing, including:
- ATOM, evaluating adjuvant tebentafusp in high-risk HLA-A*02:01-positive patients,
- and studies of darovasertib in the perioperative setting.
For now, the guideline recommends that patients with high genomic or clinical risk of metastasis should be offered clinical trial participation whenever possible.
Adjuvant external beam radiotherapy may be considered after enucleation in patients with positive margins or significant extraocular extension.
Metastatic Uveal Melanoma
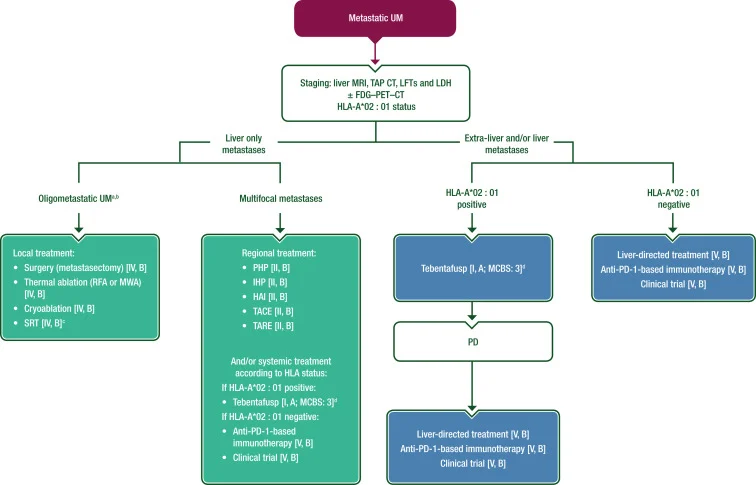
Metastatic uveal melanoma remains one of the most challenging diseases in oncology. The liver is the dominant site of metastasis, reflecting the disease’s strong hepatic tropism. Brain metastases are uncommon.
Management should be discussed in an experienced multidisciplinary tumor board, ideally including medical oncology, liver surgery, interventional radiology, radiation oncology, pathology, and ocular oncology expertise.
Liver-Directed Therapies
Because metastatic uveal melanoma is often liver-dominant, liver-directed treatment plays an unusually important role.
Surgery and Ablation
Selected patients with limited hepatic disease may benefit from resection or thermal ablation. Candidates are usually highly selected and have:
- good performance status,
- limited number of liver lesions,
- technically accessible disease,
- and no diffuse miliary involvement.
These approaches can produce prolonged survival in carefully chosen patients, although recurrence remains common.
Regional Hepatic Therapies
For patients with multifocal liver-dominant metastases, regional approaches can be considered.
Percutaneous hepatic perfusion
Melphalan-based percutaneous hepatic perfusion has emerged as an important option in selected patients with liver-confined or liver-dominant disease. Trials have shown improved hepatic and overall progression-free survival compared with best alternative care, although toxicity can be significant, especially hematologic and occasionally cardiovascular.
Isolated hepatic perfusion
Isolated hepatic perfusion is the surgical counterpart of percutaneous hepatic perfusion. It is not repeatable and has higher morbidity, so it is less commonly favored.
Hepatic arterial infusion, chemoembolization, and radioembolization
Other regional strategies such as hepatic arterial infusion, TACE, and TARE have shown variable activity, mostly in retrospective or small prospective studies. They remain reasonable options in selected patients at experienced centers.
Systemic Therapy in Metastatic Disease
For many years, systemic therapy in metastatic uveal melanoma was disappointing. Cytotoxic chemotherapy produced very low response rates and no meaningful survival benefit. Immune checkpoint inhibition also underperformed compared with cutaneous melanoma, likely because uveal melanoma has:
- low mutational burden,
- weak baseline antigenicity,
- and a highly immunosuppressive microenvironment.
Tebentafusp
The major change in the field came with tebentafusp, a T-cell receptor bispecific molecule targeting gp100 in the context of HLA-A*02:01.
A phase III trial showed that tebentafusp significantly improved overall survival in previously untreated metastatic uveal melanoma, despite limited conventional radiographic response rates and no major progression-free survival gain. This is one of the most important conceptual advances in the disease: survival benefit may occur even when RECIST responses appear modest.
Tebentafusp is now the standard first-line systemic therapy for HLA-A*02:01-positive metastatic uveal melanoma.
Immune Checkpoint Inhibitors
Single-agent PD-1 inhibitors have shown very limited activity. Combination nivolumab–ipilimumab has produced somewhat higher response rates and modest survival signals, but toxicity is considerable. The guideline suggests that combination immunotherapy may be considered mainly in selected fit patients, especially those with predominant extrahepatic disease, but it is not equivalent to the impact seen in cutaneous melanoma.
Targeted Therapy
Targeted therapies remain investigational, but combinations such as darovasertib plus crizotinib are generating interest, with encouraging early response data. These approaches remain best pursued in clinical trials.
Follow-Up and Surveillance
There is still no universally accepted surveillance strategy after treatment of primary uveal melanoma. Importantly, no prospective evidence proves that earlier detection of metastasis improves survival. Nevertheless, surveillance remains standard practice, especially for high-risk patients.
Liver function tests are insufficient as a surveillance tool. Imaging is far more important.
Low-risk disease
In low-risk patients, liver ultrasound every 6–12 months for at least 5 years is reasonable, with MRI reserved for suspicious findings.
High-risk disease
In high-risk patients, liver MRI every 4–6 months for at least 10 years is recommended, because MRI is the most sensitive modality for small hepatic metastases.
The guideline strongly supports risk-adapted surveillance, integrating both clinical and molecular risk factors, and recommends that surveillance planning be managed at expert centers.
Key Clinical Messages for Residents
- Uveal melanoma is biologically distinct from cutaneous melanoma and should not be managed as if the two diseases are interchangeable.
- Diagnosis is usually clinical; biopsy is not mandatory in a classic presentation.
- Prognosis depends not only on tumor size and location, but also on molecular features such as monosomy 3, 8q gain, BAP1 loss, SF3B1, EIF1AX, and PRAME.
- Localized disease is primarily managed with radiotherapy-based globe-preserving approaches when feasible.
- Adjuvant systemic therapy is not yet standard, and clinical trials are strongly encouraged for high-risk patients.
- Metastatic disease is predominantly liver-directed in both biology and treatment strategy.
- Tebentafusp has changed first-line treatment for HLA-A*02:01-positive metastatic disease and is now the systemic standard in that population.
- In HLA-A*02:01-negative disease, liver-directed therapies, checkpoint inhibitors, and clinical trials remain the main options.
- Surveillance should be risk-adapted, with MRI favored in high-risk patients.
- Expert multidisciplinary care is essential at every stage.
You Can Watch More on OncoDaily Youtube TV


