The TAPUR study was among the first prospective pan-cancer trials to evaluate dual immune checkpoint blockade with nivolumab plus ipilimumab in heavily pretreated patients with advanced high tumor mutation burden (HTMB) solid tumors.
Tumor mutation burden (TMB) has emerged as one of the most clinically relevant biomarkers in immuno-oncology. Tumors with high mutational loads generate larger numbers of neoantigens, potentially enhancing immune recognition and increasing sensitivity to immune checkpoint inhibition.
Although pembrolizumab had already demonstrated tumor-agnostic efficacy in TMB-high malignancies, the role of combined PD-1 and CTLA-4 inhibition across diverse tumor types remained less clearly defined. Investigators hypothesized that simultaneous blockade of these immune checkpoints could induce deeper and more durable immune activation, particularly in tumors enriched for neoantigens and adaptive immune responsiveness.
Beyond evaluating clinical activity across multiple malignancies, TAPUR also explored the broader biologic implications of HTMB as a tumor-agnostic biomarker and examined whether additional genomic features could further shape responsiveness to dual checkpoint inhibition.
Study Design and Treatment Strategy
TAPUR is a pragmatic phase II basket study designed to evaluate targeted and immune therapies outside their conventional indications in genomically selected cancers.
This analysis enrolled patients with advanced solid tumors harboring HTMB who had exhausted standard therapeutic options. Three distinct cohorts were evaluated:
- Breast cancer
- Colorectal cancer
- Histology-pooled advanced solid tumors
Patients received induction nivolumab plus ipilimumab followed by maintenance nivolumab until disease progression or unacceptable toxicity.
Importantly, the study population largely consisted of microsatellite stable tumors, allowing investigators to examine whether HTMB alone — independent of MSI-H biology — could identify tumors sensitive to dual immune checkpoint blockade.
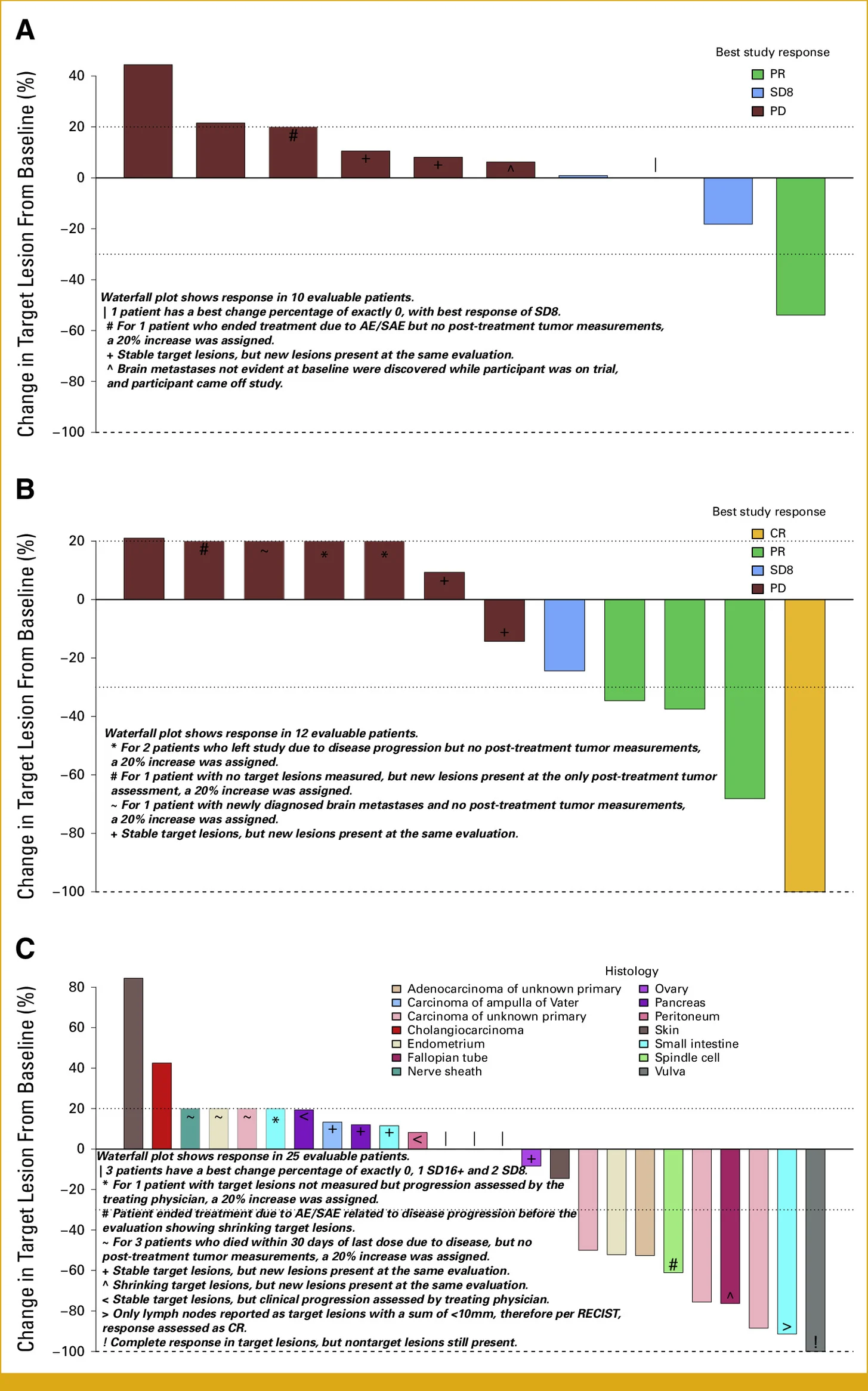
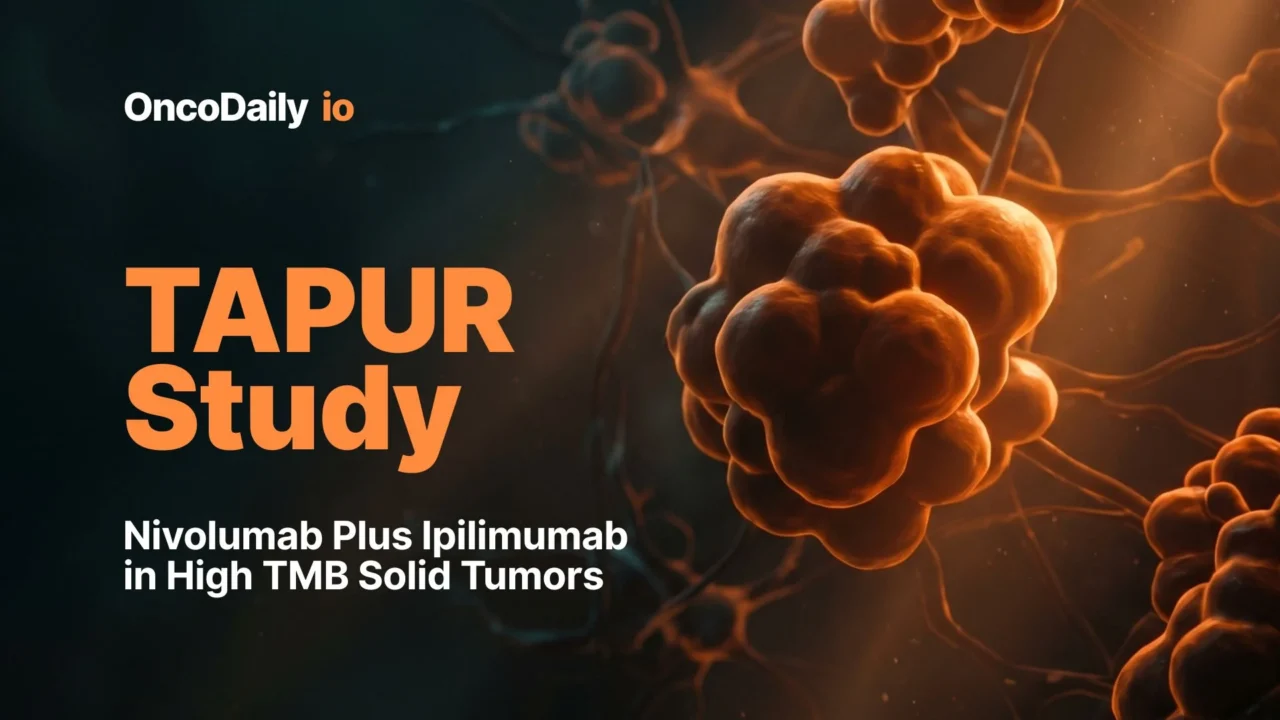
Clinical Activity Across Tumor Types
The study demonstrated that dual checkpoint blockade could produce durable antitumor activity across several HTMB malignancies, although responses varied substantially according to tumor histology.
The breast cancer cohort showed particularly encouraging activity. Durable responses were observed even in tumors traditionally considered immunologically “cold,” including microsatellite stable and PD-L1–negative disease. Several patients experienced prolonged disease control, including one patient who maintained a complete response for nearly six years, emphasizing the potential long-term immune memory generated by combined PD-1 and CTLA-4 inhibition.
The histology-pooled cohort further reinforced the tumor-agnostic potential of this strategy. Responses were documented across multiple malignancies, including endometrial cancer, vulvar cancer, small intestine cancer, and cancers of unknown primary origin. Particularly notable was the observation that several responders harbored coexisting genomic alterations involving POLE/POLD1 and homologous recombination repair pathways, suggesting that genomic complexity beyond HTMB alone may further shape immunotherapy sensitivity.
In contrast, the colorectal cancer cohort demonstrated limited clinical activity despite HTMB positivity. This finding highlighted a growing recognition within immuno-oncology that HTMB alone may not universally predict benefit across all tumor types and that tumor microenvironment composition, immune exclusion, and lineage-specific biology likely influence checkpoint inhibitor responsiveness.
Key Clinical Results
- In the breast cancer cohort, both the disease control rate and objective response rate reached 33%, including one complete response and multiple durable partial responses.
- The histology-pooled cohort demonstrated a disease control rate of 32% and an objective response rate of 28%, with responses observed across several distinct tumor types.
- In contrast, the colorectal cancer cohort showed limited activity, with both disease control and objective response rates of only 10%, leading to early cohort closure for futility.
- Immune-related toxicity remained substantial but manageable, with approximately 37% of patients developing grade 3–4 treatment-related adverse events, consistent with established nivolumab/ipilimumab safety profiles.
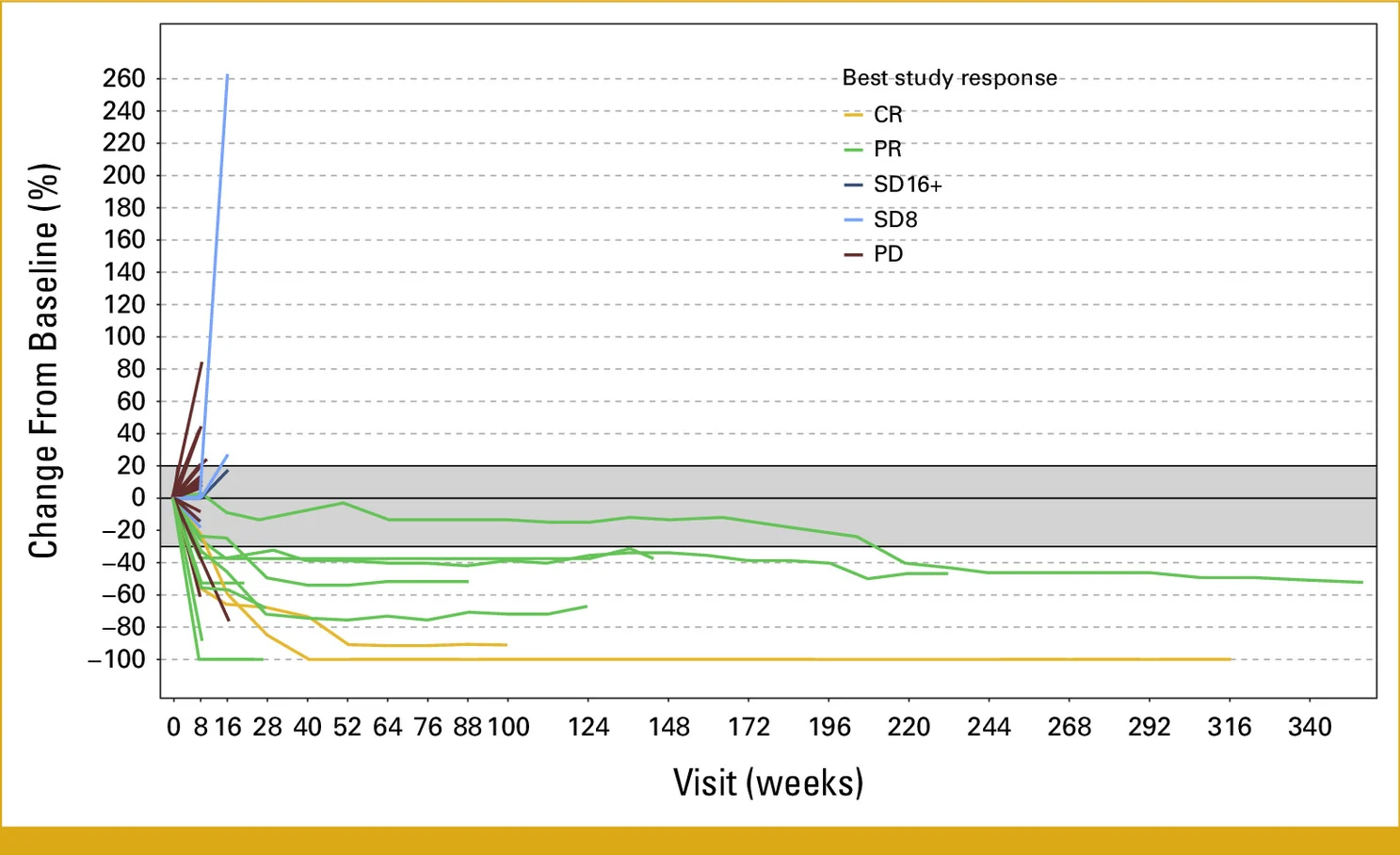
Biologic and Translational Insights
One of the most important aspects of the TAPUR analysis was its demonstration that meaningful responses occurred even in tumors lacking traditional immunotherapy biomarkers such as PD-L1 expression or MSI-H status.
This observation strengthens the concept that HTMB may function as an independent predictor of immune responsiveness in selected cancers. However, the study also illustrated the limitations of relying solely on a single biomarker. Clinical benefit appeared enriched in tumors with additional genomic features involving DNA damage repair, polymerase proofreading deficiencies, and adaptive immune activation pathways.
The data therefore support an evolving model in which future immunotherapy selection will likely require integrated molecular profiling incorporating:
- Tumor mutation burden
- DNA repair alterations
- Immune microenvironment signatures
- T-cell inflamed phenotypes
- Additional genomic and transcriptomic biomarkers
rather than HTMB alone.
Safety Profile
The toxicity profile observed in TAPUR remained consistent with prior dual checkpoint blockade studies.
Immune-related adverse events included:
- Colitis
- Autoimmune hepatitis
- Pneumonitis
- Diarrhea
- Hepatic enzyme elevations
Although grade 3–4 toxicities were relatively common, no treatment-related deaths occurred, and toxicities were generally manageable with standard immunosuppressive approaches and careful monitoring.
Clinical Significance
The TAPUR study provides important evidence supporting the tumor-agnostic potential of dual immune checkpoint blockade in selected HTMB malignancies.
At the same time, the divergent outcomes across tumor types emphasize that HTMB is not biologically equivalent across cancers. The lack of substantial activity in colorectal cancer despite elevated mutational burden highlights the complexity of immune resistance mechanisms and reinforces the need for more refined biomarker-driven patient selection strategies.
Perhaps most importantly, the study demonstrates that durable immunotherapy responses remain achievable even in heavily pretreated advanced cancers traditionally considered poorly immunogenic, expanding the potential role of precision immuno-oncology across a broader spectrum of malignancies.
Read Full Article Here