The tumor microenvironment (TME) is characterized by intense metabolic competition between cancer cells and immune cells. Tumor cells consume large amounts of nutrients required for immune-cell activation, often driving CD8+ T cells into dysfunctional or exhausted states that weaken anti-tumor immunity and reduce responsiveness to immune checkpoint blockade. Dietary interventions have increasingly attracted attention as potential strategies to reshape tumor metabolism, yet prolonged fasting and restrictive diets remain difficult to implement clinically because many patients with cancer are already metabolically vulnerable or malnourished.
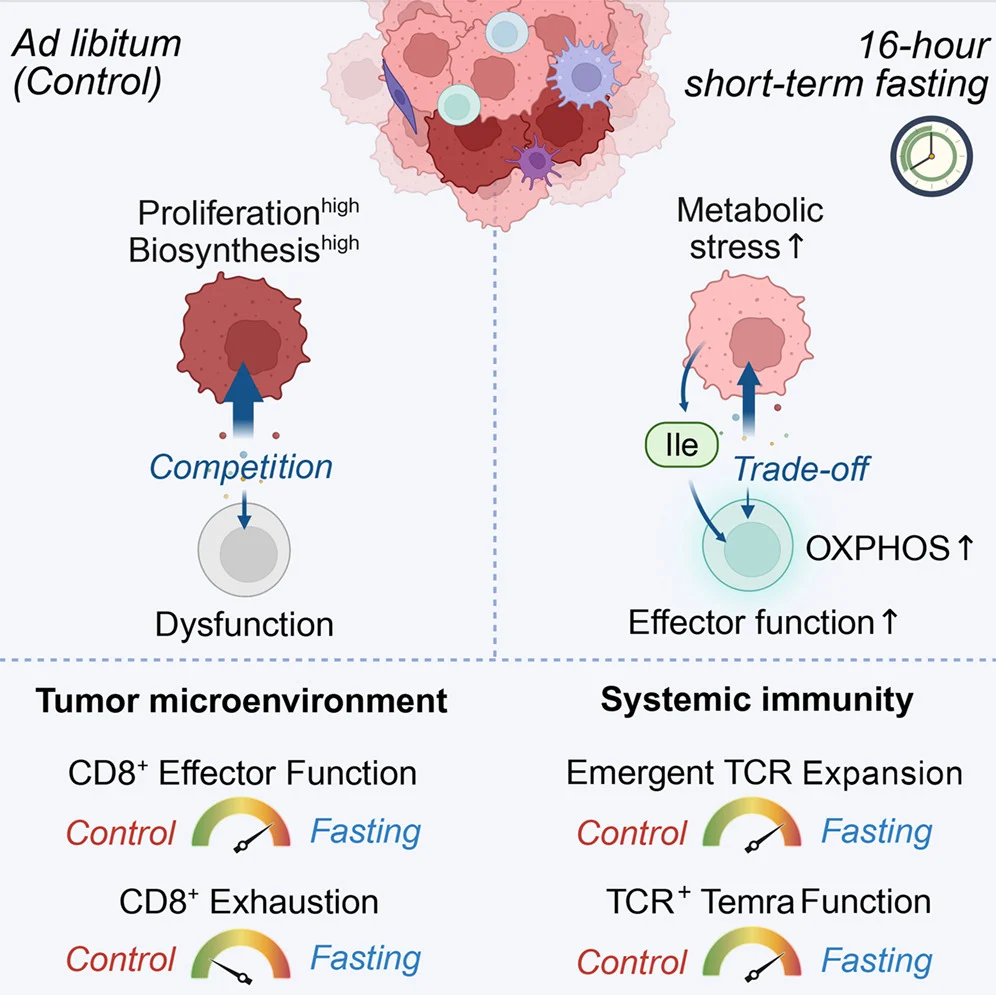
In this study, investigators explored whether a brief and clinically feasible 16-hour fasting regimen could transiently reprogram the tumor microenvironment in a way that selectively favors immune-cell function without the toxicity associated with prolonged fasting protocols. Using murine tumor models, single-cell sequencing, metabolomics, and prospective human cohorts, the study demonstrated that short-term fasting significantly enhanced anti-tumor immunity and improved responses to immunotherapy. Mechanistically, fasting induced accumulation of intratumoral isoleucine, a branched-chain amino acid that became a critical metabolic substrate supporting CD8+ T-cell cytotoxicity and epigenetic activation.
Study Design and Experimental Strategy
The investigators designed a transient overnight fasting protocol beginning at 8 p.m. and continuing until 12 p.m. the following day, creating a total fasting duration of 16 hours. This schedule intentionally mirrored fasting practices already familiar in perioperative clinical settings, making the strategy substantially more translatable than previously studied multi-day fasting interventions.
The study integrated:
- Murine melanoma and colorectal tumor models
- Single-cell RNA sequencing
- Tumor interstitial fluid metabolomics
- T-cell receptor sequencing
- Human prospective clinical cohorts receiving immunotherapy
to comprehensively characterize how fasting reshapes tumor metabolism and anti-tumor immune responses.
Short-Term Fasting Reprogrammed the Tumor Immune Microenvironment
Single-cell analyses demonstrated that fasting profoundly altered the functional state of intratumoral immune cells. Rather than globally changing immune-cell composition, fasting selectively enhanced the cytotoxic capacity of CD8+ T cells while suppressing exhaustion programs and immunosuppressive signaling within the tumor microenvironment. Functional enrichment analyses showed increased activation of anti-tumor immune pathways, alongside reductions in dysfunctional T-cell states and suppressive myeloid-cell activity.
Importantly, the metabolic effects of fasting appeared highly tumor-specific. Human tumor samples collected from patients undergoing surgery after overnight fasting demonstrated stronger CD4+ and CD8+ immune activation networks, increased cytotoxic Temra CD8+ T-cell populations, and reduced exhaustion signatures compared with controls receiving nutritional supplementation. The fasting intervention therefore appeared capable of rapidly shifting the tumor microenvironment toward a more immunologically active state even after only a single overnight fasting cycle.
Isoleucine Emerged as a Central Metabolic Driver of Anti-Tumor Immunity
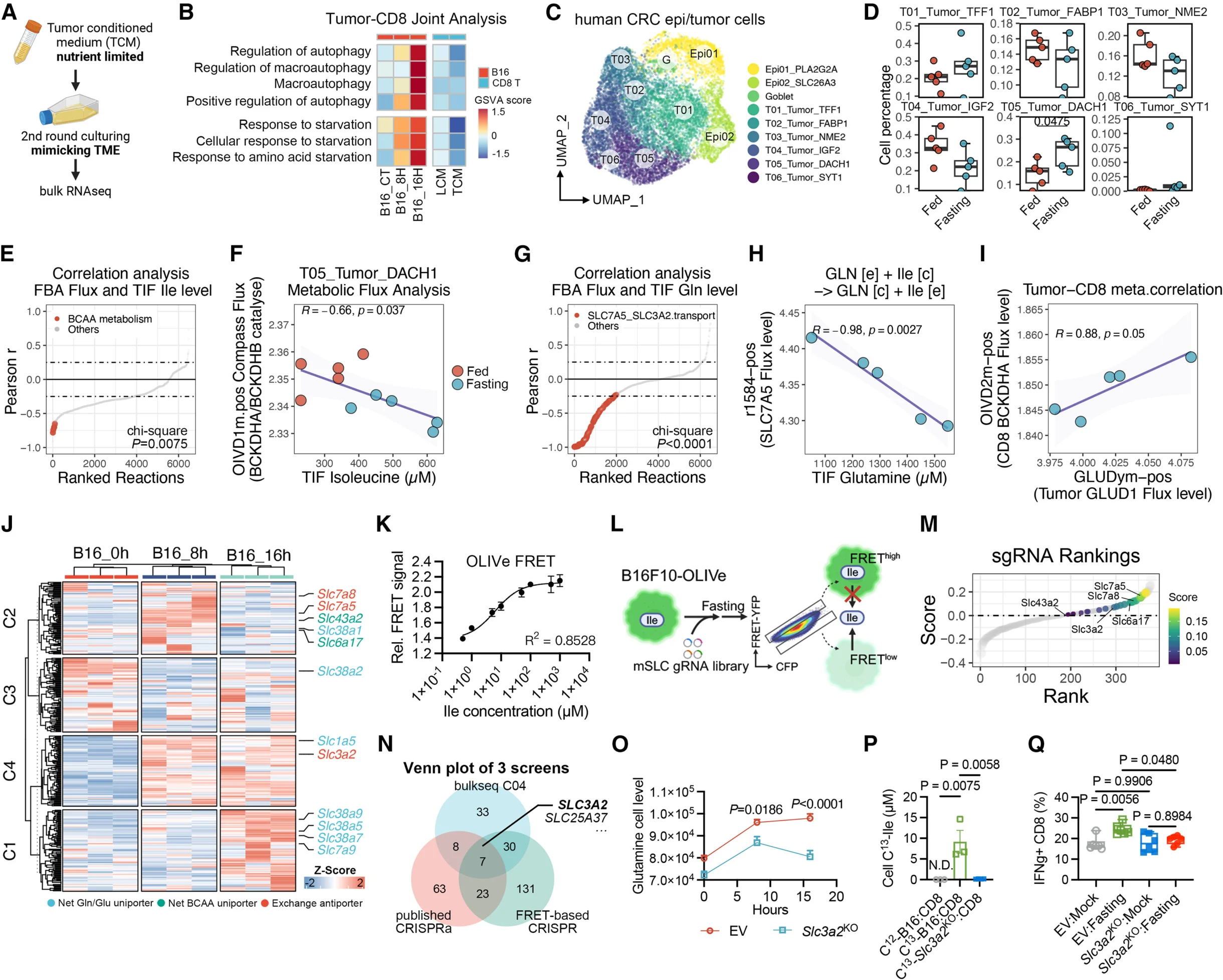
One of the study’s most important findings was the discovery that fasting induced marked accumulation of isoleucine within the tumor microenvironment. Metabolomic analyses revealed that this effect occurred specifically inside tumors, while serum levels remained relatively stable and adjacent healthy tissues showed minimal metabolic change.
Functional experiments demonstrated that isoleucine was indispensable for effective CD8+ T-cell activity. When isoleucine was removed from culture media, CD8+ T-cell proliferation and effector function declined substantially. Conversely, isoleucine supplementation restored T-cell proliferation and significantly increased interferon-γ and granzyme B production under nutrient-deprived conditions resembling the tumor microenvironment.
Mechanistically, isoleucine fueled intracellular acetyl-CoA pools through BCAT2-dependent metabolism, supporting mitochondrial oxidative phosphorylation, histone acetylation, chromatin accessibility, and phospholipid synthesis required for sustained T-cell activation. The study therefore identified a direct metabolic-epigenetic connection between fasting-induced amino acid availability and enhanced cytotoxic T-cell function.
Tumor Cells Underwent a Metabolic Trade-Off During Fasting
The investigators further demonstrated that fasting forced tumor cells into a state of metabolic adaptation. Under nutrient stress, tumor cells increasingly prioritized glutamine acquisition and oxidative phosphorylation pathways required for survival. This adaptation unexpectedly created a metabolic trade-off in which isoleucine was released into the tumor microenvironment through SLC3A2-mediated amino-acid exchange mechanisms.
This process effectively generated a transient metabolic window favoring immune-cell activity. While tumor cells struggled to maintain survival pathways under fasting conditions, CD8+ T cells utilized the newly available isoleucine to restore their effector functions. Disruption of the SLC3A2 transporter eliminated fasting-induced isoleucine accumulation and abrogated the enhancement of CD8+ T-cell activity, confirming the mechanistic importance of this metabolic exchange system.
Short-Term Fasting Enhanced Responses to Immunotherapy
The translational significance of these findings became especially apparent when fasting was combined with immune checkpoint blockade. In murine melanoma and colorectal cancer models, fasting synchronized with anti-PD1 therapy significantly enhanced tumor control and reduced terminally exhausted PD1+TIM3+ CD8+ T-cell populations. Fasting also enhanced antigen-presentation pathways and reduced immunosuppressive signaling within the tumor microenvironment.
The investigators subsequently evaluated this approach in a prospective cohort of patients with pMMR/MSS colorectal cancer receiving chemotherapy plus anti-PD1 therapy. Single-cell sequencing and T-cell receptor tracking demonstrated that fasting promoted expansion of tumor-reactive Temra and resident-memory T-cell populations, enhanced TCR clonal expansion, and reduced exhaustion-associated transcriptional programs. Tumor-reactive T-cell clones in fasting-treated patients exhibited higher expression of cytotoxic markers such as IFNG and GZMB, together with lower expression of inhibitory checkpoint-associated genes including PDCD1 and HAVCR2.
Key Results
- A 16-hour fasting regimen was sufficient to induce tumor-specific metabolic reprogramming associated with enhanced anti-tumor immunity and improved immune-cell functionality within the tumor microenvironment.
- Short-term fasting enhanced major functional characteristics of anti-tumor CD8+ T cells, including increased cytotoxic activity, greater clonal expansion, and improved effector-memory differentiation, while simultaneously suppressing T-cell exhaustion programs.
- Fasting induced significant accumulation of intratumoral isoleucine with minimal changes in systemic serum levels, highlighting a highly localized metabolic effect within the tumor microenvironment.
- In the prospective human immunotherapy cohort, 6 of 7 patients achieved partial responses, and fasting-treated patients demonstrated greater tumor regression together with enhanced expansion of tumor-reactive T-cell populations.
- Combination of fasting with anti-PD1 therapy significantly improved tumor control in murine melanoma and colorectal cancer models and was associated with reduced populations of terminally exhausted PD1+TIM3+ CD8+ T cells.
Read Full Article Here


